
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
2nd Edition
ISBN: 9780077633707
Author: Janice Smith
Publisher: Mcgraw-hill Higher Education (us)
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.32UKC
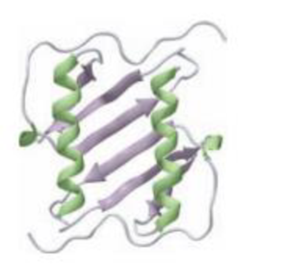
Label the regions of secondary structure in the following protein ribbon diagram.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Provide the structure, circle or draw, of the monomeric unit found in the biological polymeric
materials given below.
HO
OH
amylose
OH
OH
행
3
HO
cellulose
OH
OH
OH
Ho
HO
What units (if any) does K have? Does K depend upon how the concentration is expressed (e.g. molarity, ppm, ppb, etc.)? in calculating the response factor
Don't used hand raiting and don't used Ai solution
Chapter 16 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
Ch. 16.2 - In addition to the amino and carboxyl groups, what...Ch. 16.2 - How do the OH groups in Ser, Thr, and Tyr differ?Ch. 16.2 - Draw both enantiomers of each amino acid in...Ch. 16.2 - Which of the following amino acids is naturally...Ch. 16.3 - Draw the structure of the amino acid valine at...Ch. 16.3 - Identify the amino acid shown with all uncharged...Ch. 16.3 - Draw the positively charged, neutral, and...Ch. 16.4 - Identify the N-terminal and C-terminal amino acid...Ch. 16.4 - (a) Identify the N-terminal amino acid in the...Ch. 16.4 - Identify the individual amino acids in each...
Ch. 16.4 - Prob. 16.11PCh. 16.5 - Prob. 16.12PCh. 16.6 - Prob. 16.13PCh. 16.6 - Draw the structures of each pair of amino acids...Ch. 16.6 - The fibroin proteins found in silk fibers consist...Ch. 16.7 - Prob. 16.16PCh. 16.7 - Prob. 16.17PCh. 16.8 - Prob. 16.18PCh. 16.8 - Prob. 16.19PCh. 16.8 - Prob. 16.20PCh. 16.9 - Prob. 16.21PCh. 16.9 - Prob. 16.22PCh. 16.9 - The nerve gas sarin acts as a poison by covalently...Ch. 16.10 - Prob. 16.24PCh. 16 - Prob. 16.25UKCCh. 16 - Prob. 16.26UKCCh. 16 - For each amino acid: [1] draw the L enantiomer in...Ch. 16 - For each amino acid: [1] give the name; [2] give...Ch. 16 - For the given tripeptide: (a) identify the amino...Ch. 16 - For the given tripeptide: (a) identify the amino...Ch. 16 - Label the regions of secondary structure in the...Ch. 16 - Label the regions of secondary structure in the...Ch. 16 - What type of interactions occur at each of the...Ch. 16 - Using the given representations for an enzyme and...Ch. 16 - Naturally occurring amino acids are L--amino...Ch. 16 - Why do neutral amino acids exist as zwitterions...Ch. 16 - The amino acid alanine is a solid at room...Ch. 16 - Why is phenylalanine water soluble but...Ch. 16 - Draw the structure of a naturally occurring amino...Ch. 16 - Draw the structure of a naturally occurring amino...Ch. 16 - For each amino acid: [1] draw the l enantiomer in...Ch. 16 - For each amino acid: [1] draw the l enantiomer in...Ch. 16 - Draw both enantiomers of each amino acid and label...Ch. 16 - Which of the following Fischer projections...Ch. 16 - For each amino acid: [1] give the name; [2] give...Ch. 16 - For each amino acid: [1] give the name; [2] give...Ch. 16 - Draw the amino acid leucine at each pH: (a) 6; (b)...Ch. 16 - Draw the amino acid isoleucine at each pH: (a) 6;...Ch. 16 - Draw the structure of the neutral, positively...Ch. 16 - Draw the structure of the neutral, positively...Ch. 16 - For each tripeptide: [1] identify the N-terminal...Ch. 16 - For each tripeptide: [1] identify the N-terminal...Ch. 16 - For the given tripeptide: (a) identify the amino...Ch. 16 - For the given tripeptide: (a) identify the amino...Ch. 16 - Locate the peptide bond in the dipeptide shown in...Ch. 16 - Label the N-terminal and C-terminal amino acids in...Ch. 16 - Draw the structures of the amino acids formed when...Ch. 16 - Draw the structures of the amino acids formed when...Ch. 16 - What amino acids are formed by hydrolysis of the...Ch. 16 - Give the three-letter abbreviations for the amino...Ch. 16 - What is the difference between the primary and...Ch. 16 - What is the difference between the tertiary and...Ch. 16 - What type of intermolecular forces exist between...Ch. 16 - Which of the following pairs of amino acids can...Ch. 16 - List two amino acids that would probably be...Ch. 16 - List two amino acids that would probably be...Ch. 16 - Compare -keratin and hemoglobin with regards to...Ch. 16 - Compare collagen and myoglobin with regards to...Ch. 16 - When a protein is denatured, how is its primary,...Ch. 16 - Hydrogen bonding stabilizes both the secondary and...Ch. 16 - Describe the function or biological activity of...Ch. 16 - Describe the function or biological activity of...Ch. 16 - Use the given representations for an enzyme,...Ch. 16 - Use the given representations for an enzyme and...Ch. 16 - How are enzyme inhibitors used to treat high blood...Ch. 16 - How are enzyme inhibitors used to treat HIV? Give...Ch. 16 - What structural feature in -keratin makes...Ch. 16 - Why does the -keratin in hair contain many...Ch. 16 - Why must vegetarian diets be carefully balanced?Ch. 16 - Why does cooking meat make it easier to digest?Ch. 16 - Sometimes an incision is cauterized (burned) to...Ch. 16 - Prob. 16.82APCh. 16 - How is sickle cell disease related to hemoglobin...Ch. 16 - The silk produced by a silkworm is a protein with...Ch. 16 - Explain the difference in the mechanism of action...Ch. 16 - How are blood enzyme levels used to diagnose...Ch. 16 - Explain why two amino acids aspartic acid and...Ch. 16 - Prob. 16.88CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used Ai solution and don't used hand raitingarrow_forwardOA. For the structure shown, rank the bond lengths (labeled a, b and c) from shortest to longest. Place your answer in the box. Only the answer in the box will be graded. (2 points) H -CH3 THe b Нarrow_forwardDon't used hand raitingarrow_forward
- Quizzes - Gen Organic & Biological Che... ☆ myd21.lcc.edu + O G screenshot on mac - Google Search savings hulu youtube google disney+ HBO zlib Homework Hel...s | bartleby cell bio book Yuzu Reader: Chemistry G periodic table - Google Search b Home | bartleby 0:33:26 remaining CHEM 120 Chapter 5_Quiz 3 Page 1: 1 > 2 > 3 > 6 ¦ 5 > 4 > 7 ¦ 1 1 10 8 ¦ 9 a ¦ -- Quiz Information silicon-27 A doctor gives a patient 0.01 mC i of beta radiation. How many beta particles would the patient receive in I minute? (1 Ci = 3.7 x 10 10 d/s) Question 5 (1 point) Saved Listen 2.22 x 107 222 x 108 3.7 x 108 2.22 x 108 none of the above Question 6 (1 point) Listen The recommended dosage of 1-131 for a test is 4.2 μCi per kg of body mass. How many millicuries should be given to a 55 kg patient? (1 mCi = 1000 μСi)? 230 mCiarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
- Q3: Arrange each group of compounds from fastest SN2 reaction rate to slowest SN2 reaction rate. CI Cl H3C-Cl CI a) A B C D Br Br b) A B C Br H3C-Br Darrow_forwardQ4: Rank the relative nucleophilicity of halide ions in water solution and DMF solution, respectively. F CI Br | Q5: Determine which of the substrates will and will not react with NaSCH3 in an SN2 reaction to have a reasonable yield of product. NH2 Br Br Br .OH Brarrow_forwardClassify each molecule as optically active or inactive. Determine the configuration at each H соон Chirality center OH 애 He OH H3C Ноос H H COOH A K B.arrow_forward
- Q1: Rank the relative nucleophilicity of the following species in ethanol. CH3O¯, CH3OH, CH3COO, CH3COOH, CH3S Q2: Group these solvents into either protic solvents or aprotic solvents. Acetonitrile (CH3CN), H₂O, Acetic acid (CH3COOH), Acetone (CH3COCH3), CH3CH2OH, DMSO (CH3SOCH3), DMF (HCON(CH3)2), CH3OHarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward10. The main product of the following reaction is [1.1:4',1"-terphenyl]-2'-yl(1h-pyrazol-4- yl)methanone Ph N-H Pharrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY