
Chemistry: The Molecular Nature of Matter
7th Edition
ISBN: 9781118516461
Author: Neil D. Jespersen, Alison Hyslop
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 78RQ
Which of the two molecules below is the stronger Brønsted-Lowry acid? Why?
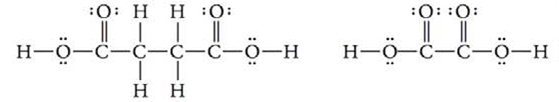
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which is the stronger acid in each of the following pairs?
Which of the following is a Brønsted-Lowry base?
Ο ΝH3
О ССц
О НF
O I2
None of the above are Brønsted-Lowry bases.
Please arrange the following in order of decreasing acid strength.
Chapter 15 Solutions
Chemistry: The Molecular Nature of Matter
Ch. 15 - Which of the following are conjugate acid-base...Ch. 15 - Write the formula of the conjugate base for each...Ch. 15 - Sodium cyanide solution, when poured into excess...Ch. 15 - One kind of baking powder contains sodium...Ch. 15 - Which of the following are amphoteric and which...Ch. 15 - The anion of sodium monohydrogen phosphate,...Ch. 15 -
Given that is a stronger acid than what is the...Ch. 15 - Given that HClO is a weaker acid than determine...Ch. 15 - Order the following groups of acids from the...Ch. 15 - Using only the periodic cable, choose the stronger...
Ch. 15 - Prob. 11PECh. 15 - Explain why one acid is weaker than the other in...Ch. 15 - In each pair, explain why one is a stronger acid...Ch. 15 - In each pair, explain why one is a weaker acid...Ch. 15 - How would you expect the acidities of the...Ch. 15 - List these acids in terms of increasing acidity:...Ch. 15 - Identify the Lewis acid and Lewis base in each...Ch. 15 - Is the fluoride ion more likely to behave as a...Ch. 15 - Brnsted-Lowry Acids and Bases How is a...Ch. 15 - Brnsted-Lowry Acids and Bases How are the formulas...Ch. 15 - Brnsted-Lowry Acids and Bases Is H2SO4 the...Ch. 15 - Brnsted-Lowry Acids and Bases What is meant by the...Ch. 15 - Brnsted-Lowry Acids and Bases Define the term...Ch. 15 - Strengths of Bronsted-Lowry Acids and Bases
15.6...Ch. 15 - Strengths of Brønsted-Lowry Acids and Bases
15.7...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases The...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases Acetic...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases Nitric...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases HCIO4...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases Formic...Ch. 15 - Periodic Trends in the Strength of Acids Explain...Ch. 15 - Periodic Trends in the Strength of Acids What are...Ch. 15 - Periodic Trends in the Strength of Acids Within...Ch. 15 - Periodic Trends in the Strength of Acids Explain...Ch. 15 - Periodic Trends in the Strength of Acids Within...Ch. 15 - Periodic Trends in the Strength of Acids Explain...Ch. 15 - Periodic Trends in the Strength of Acids Astatine,...Ch. 15 - Periodic Trends in the Strength of Acids
15.21...Ch. 15 - Periodic Trends in the Strength of Acids
15.22...Ch. 15 - Periodic Trends in the Strength of Acids Which of...Ch. 15 - Periodic Trends in the Strength of Acids Which of...Ch. 15 - Lewis Acids and Bases Define Lewis acid and Lewis...Ch. 15 - Lewis Acids and Bases In terms of atomic orbitals,...Ch. 15 - Lewis Acids and Bases
15.27 Explain why the...Ch. 15 - Lewis Acids and Bases Methylamine has the formula...Ch. 15 - Use Lewis structures to show the Lewis acid-base...Ch. 15 - Lewis Acids and Bases
15.30 Explain why the oxide...Ch. 15 - Lewis Acids and Bases The molecule SbF5 is able to...Ch. 15 - Lewis Acids and Bases In the reaction of calcium...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Prob. 35RQCh. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Prob. 40RQCh. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Prob. 43RQCh. 15 - Advanced Ceramics and Acid-Base Chemistry What is...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry What is...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry
15.46...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry How does...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry
15.48...Ch. 15 - Brønsted-Lowry Acids and Bases
15.49 Write the...Ch. 15 - Brønsted-Lowry Acids and Bases
15.50 Write the...Ch. 15 - Brønsted-Lowry Acids and Bases
15.51 Write the...Ch. 15 - Brnsted-Lowry Acids and Bases Write the formula...Ch. 15 - Brønsted-Lowry Acids and Bases
15.53 Identify the...Ch. 15 - Brønsted-Lowry Acids and Bases
15.54 Identify the...Ch. 15 - Periodic Trends in the Strengths of Acids Choose...Ch. 15 - Periodic Trends in the Strengths of Acids Choose...Ch. 15 - Choose the stronger acid and give your reason:...Ch. 15 - Choose the stronger acid and give your reason:...Ch. 15 - Choose the stronger acid:...Ch. 15 - Choose the stronger acid:...Ch. 15 - Lewis Acids and Bases Use Lewis symbols co diagram...Ch. 15 - Lewis Acids and Bases Use Lewis symbols to diagram...Ch. 15 - *15.63 Beryllium chloride, , exists in the solid...Ch. 15 - Aluminum chloride, AlCl3, forms molecules with...Ch. 15 - Use Lewis structures to diagram the reaction...Ch. 15 - Use Lewis structures to diagram the reaction...Ch. 15 - Use Lewis structures to show how the following...Ch. 15 - *15.68 Use Lewis structures to show how the...Ch. 15 - Acid-Base Properties of Elements and Their...Ch. 15 - Acid-Base Properties of Elements and Their Oxides...Ch. 15 - Prob. 71RQCh. 15 - Prob. 72RQCh. 15 - What is the formula of the conjugate acid of...Ch. 15 - *15.74 Using liquid ammonia as a solvent, sodium...Ch. 15 - In liquid SO2asasolvent,SOCl2reactswithNa2SO3 in a...Ch. 15 - *15.76 The following space-filling model depicts...Ch. 15 - Which of the following compounds is the stronger...Ch. 15 - Which of the two molecules below is the stronger...Ch. 15 - 15.79 Write equations that illustrate the...Ch. 15 - Hydrogen peroxide is a stronger Brnsted-Lowry acid...Ch. 15 - Sodium hydroxide, NaOH, is basic. Aluminum...Ch. 15 - Hydrazine, N2H4, is a weaker Brnsted-Lowry base...Ch. 15 - Identify the two Brnsted-Lowry acids and two bases...Ch. 15 - In the reaction in the preceding exercise, the...Ch. 15 - How would you expect the degree of ionization of...Ch. 15 - Prob. 86RQCh. 15 - A mixture is prepared containing 0.10 M of each of...Ch. 15 - 15.88 Are all Arrhenius acids Brønsted-Lowry...Ch. 15 - How could you determine whether HBr is a stronger...Ch. 15 - 15.90 Alcohols are organic compounds that have an...Ch. 15 - Acid rain, acid mine runoff, and acid leaching of...Ch. 15 - 15.92 Using just Figure 7.30, find the five most...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Distinguish between microevolution, speciation, and macroevolution.
Campbell Essential Biology (7th Edition)
Police Captain Jeffers has suffered a myocardial infarction. a. Explain to his (nonmedically oriented) family w...
Human Physiology: An Integrated Approach (8th Edition)
When limonene (Section 23.3) is heated strongly, it yields 2 mol of isoprene. What kind of reaction is involved...
Organic Chemistry
6.1 State the number of electrons that be must be lost by atoms of each of the following to achieve a stable el...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Choose the best answer to each of the following. Explain your reasoning. Which of a planets fundamental propert...
Cosmic Perspective Fundamentals
Why is an endospore called a resting structure? Of what advantage is an endospore to a bacterial cell?
Microbiology: An Introduction
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The sec-butyl cation can react as both a Brnsted-Lowry acid (a proton donor) and a Lewis acid (an electron pair acceptor) in the presence of a watersulfuric acid mixture. In each case, however, the product is different. The two reactions are as follows: (a) In which reaction(s) does this cation react as a Lewis acid? In which reaction(s) does it react as a Brnsted-Lowry acid? (b) Write Lewis structures for reactants and products and show by the use of curved arrows how each reaction occurs.arrow_forwardHypofluorous acid, HOF, is known, but fluorous acid, HOFO, is not. Which acid would you expect to be stronger?arrow_forwardHydrazine, N2H4 (having the structure H2NNH2), and its derivatives have been used as rocket fuels. Draw the Lewis electron-dot formula for the hydrazine molecule. Describe the geometries expected about the nitrogen atoms in this molecule. Why would you expect hydrazine to be basic? Which substance, NH3 or N2H4, would you expect to be more basic? Why? Write the chemical equation in which hydrazine reacts with hydrochloric acids to form the salt N2H5Cl. Consider the positive ion of this salt. How does its basic character compare with that of NH3 and N2H4? Explain.arrow_forward
- Order each of the following pairs by acid strength, giving the weaker acid first. Explain your answer. a HNO3, HNO2 b HCO3, H2CO3 c H2S, H2Te d HCl, H2S e H3PO4, H3AsO4arrow_forwardThe following reactions illustrate Brnsted acid-base behavior. Complete each equation. a.HI(aq)+?H3O+(aq)+I(aq) b.NH3(l)+?NH4++NH2 c.H2C2O4(aq)+H2O(l)?+HC2O4(aq) d.H2N2O2(aq)+H2O(l)H3O+(aq)+? e.?+H2O(l)H3O+(aq)+CO32(aq)arrow_forwardThe structure of ethylene diamine is illustrated in study question 76. Is this compound a Brnsted acid, a Brnsted base, a Lewis acid or a Lewis base, or some combination of these.arrow_forward
- In the following net ionic reaction, identify each species as either a Brnsted-Lowry acid or a Brnsted -Lowry base: CH3COO(aq)+HS(aq)CH3COOH(aq)+S2(aq). Identify the conjugate of each reactant and state whether it is a conjugate acid or a conjugate base.arrow_forwardBoric acid, H3303, is not a Bronsted-Lowry acid but a Lewis acid.. (a) Write an equation for its reaction with water. (b) Predict the shape of the anion thus formed. (c) What is the hybridization 0n the boron consistent with the shape you have predicted?arrow_forwardWhat are the major species in solution after NaHSO4 is dissolved in water? What happens to the pH of the solution as more NaHSO4 is added? Why? Would the results vary if baking soda (NaHCO3) were used instead?arrow_forward
- Several acids and their respective equilibrium constants are: Which is the strongest acid? Which is the weakest acid? Which acid has the weakest conjugate base? Which acid has the strongest conjugate base?arrow_forwardWhich of the following conditions indicate an acidic solution? pH = 3.04 (H+| > 1.0 X IO’7 A/ pOH = 4.51 |OH-J = 3.21 X 10"12 Marrow_forwardHydrazine (N2H4) is like CO32 in that it is a polyprotic base (Kb1 = 85 107 and Kb2 = 8.9 1016). The two conjugate acids are N2H5+ and N2H62+. What is the expected pH of a 0.025 M solution of N2H4? (a) 3.83 (b) 8.32 (c) 10.16arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY