
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.35AP
The sec-butyl cation can react as both a Brønsted-Lowry acid (a proton donor) and a Lewis acid (an electron pair acceptor) in the presence of a water–sulfuric acid mixture. In each case, however, the product is different. The two reactions are as follows:
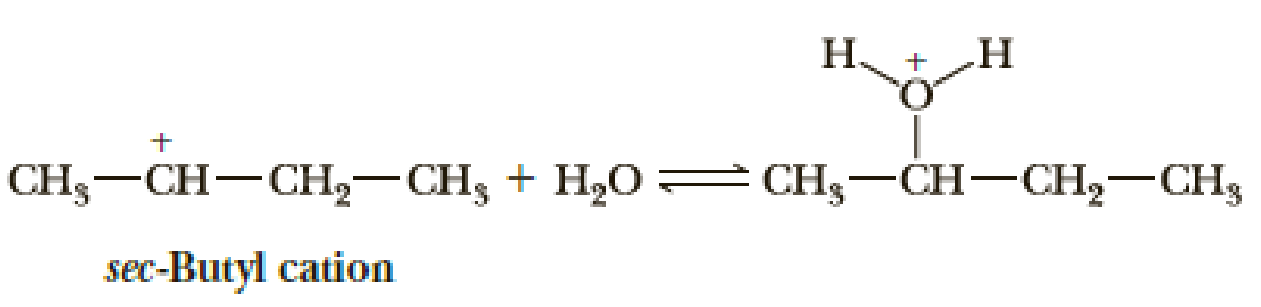
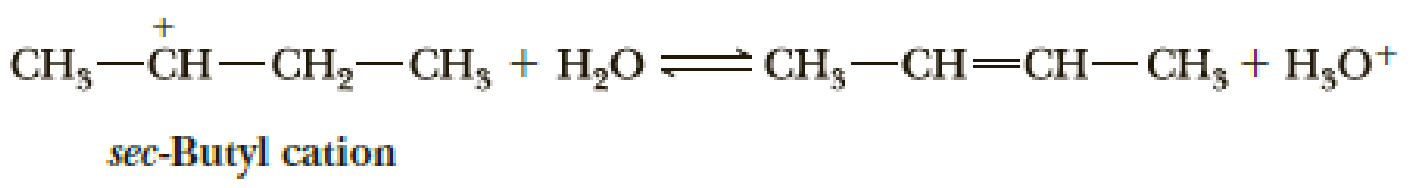
- (a) In which reaction(s) does this cation react as a Lewis acid? In which reaction(s) does it react as a Brønsted-Lowry acid?
- (b) Write Lewis structures for reactants and products and show by the use of curved arrows how each reaction occurs.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Using the following two half-reactions, determine the pH range in which $NO_2^-\ (aq)$ cannot be found as the predominant chemical species in water.* $NO_3^-(aq)+10H^+(aq)+8e^-\rightarrow NH_4^+(aq)+3H_2O(l),\ pE^{\circ}=14.88$* $NO_2^-(aq)+8H^+(aq)+6e^-\rightarrow NH_4^+(aq)+2H_2O(l),\ pE^{\circ}=15.08$
Indicate characteristics of oxodec acid.
What is the final product when hexanedioic acid reacts with 1º PCl5 and 2º NH3.
Chapter 4 Solutions
Organic Chemistry
Ch. 4.2 - For each conjugate acid-base pair, identify the...Ch. 4.2 - Write these reactions as proton-transfer...Ch. 4.2 - Following is a structural formula for guanidine,...Ch. 4.2 - Write an equation to show the proton transfer...Ch. 4.3 - For each value of Ka, calculate the corresponding...Ch. 4.4 - Predict the position of equilibrium and calculate...Ch. 4.5 - Calculate Keq for a reaction with G0 = 17.1 kJ/mol...Ch. 4.6 - Acid-Base Equilibria Many factors contribute to...Ch. 4.6 - What is the relative trend in acidity and pKa of...Ch. 4.7 - Write an equation for the reaction between each...
Ch. 4 - For each conjugate acid-base pair, identify the...Ch. 4 - Complete a net ionic equation for each...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Prob. 4.12PCh. 4 - In acetic acid, CH3COOH, the OH hydrogen is more...Ch. 4 - Which has the larger numerical value? (a) The pKa...Ch. 4 - In each pair, select the stronger acid. (a)...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - If the G for a reaction is 4.5 kcal/mol at 298 K,...Ch. 4 - Calculate the Keq for the following reactions from...Ch. 4 - Prob. 4.20PCh. 4 - Answer true or false to the following statements...Ch. 4 - In each of the following three reaction coordinate...Ch. 4 - The acid-base chemistry reaction of barium...Ch. 4 - Unless under pressure, carbonic acid (H2CO3) in...Ch. 4 - Prob. 4.25PCh. 4 - Acetic acid, CH3COOH, is a weak organic acid, pKa...Ch. 4 - Benzoic acid, C6H5COOH (pKa 4.19), is only...Ch. 4 - Prob. 4.28PCh. 4 - One way to determine the predominant species at...Ch. 4 - Will acetylene react with sodium hydride according...Ch. 4 - Prob. 4.31PCh. 4 - For each equation, label the Lewis acid and the...Ch. 4 - Complete the equation for the reaction between...Ch. 4 - Each of these reactions can be written as a Lewis...Ch. 4 - The sec-butyl cation can react as both a...Ch. 4 - Prob. 4.36APCh. 4 - Prob. 4.37APCh. 4 - Prob. 4.38APCh. 4 - Explain why the hydronium ion, H3O+, is the...Ch. 4 - What is the strongest base that can exist in...Ch. 4 - Prob. 4.42APCh. 4 - Prob. 4.43APCh. 4 - Methyl isocyanate, CH3N=C=O, is used in the...Ch. 4 - Offer an explanation for the following...Ch. 4 - Prob. 4.46APCh. 4 - Alcohols (Chapter 10) are weak organic acids, pKa...Ch. 4 - As we shall see in Chapter 19, hydrogens on a...Ch. 4 - 2,4-Pentanedione is a considerably stronger acid...Ch. 4 - Write an equation for the acid-base reaction...Ch. 4 - Prob. 4.51APCh. 4 - Prob. 4.52APCh. 4 - Prob. 4.53APCh. 4 - Following is a structural formula for imidazole, a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forwardIn the two chair conformations of glucose, the most stable is the one with all the OH groups in the equatorial position. Is this correct?arrow_forward
- please help me with my homeworkarrow_forwardhelparrow_forwardThe temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forward
- QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY