
Concept explainers
(a)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(a)
Explanation of Solution
Given molecule,
Increase in
(b)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(b)
Explanation of Solution
Given molecule,
Increase in
(c)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(c)
Explanation of Solution
Given molecule,
Increase in
(d)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(d)
Explanation of Solution
Given molecule,
Increase in
(e)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(e)
Explanation of Solution
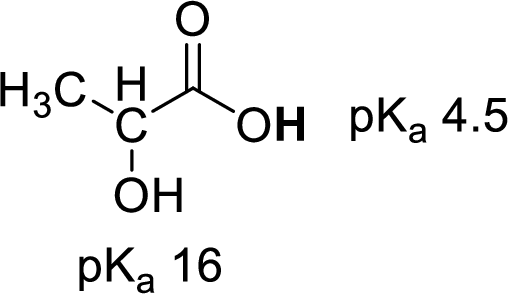
Given molecule,
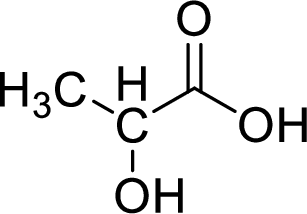
Increase in
(f)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(f)
Explanation of Solution
Given molecule,
Increase in
(g)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(g)
Explanation of Solution
Given molecule,
Increase in
(h)
Interpretation:
The most acidic hydrogen in given molecule has to be labelled and justify it using
Concept Introduction:
Position of equilibrium in acid-base reaction:
In an acid-base reaction, the position of equilibrium always favors reaction of the stronger acid and stronger base to form the weaker acid and weaker base. Therefore, the major species present at equilibrium in an acid-base reaction are weaker acid and weaker base. The reaction equilibrium shifts to a direction where weaker acid and weaker base is formed. Acids with greater
(h)
Explanation of Solution
Given molecule,
Increase in
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry
- Indicate the product formed in each reaction. If the product exhibits tautomerism, draw the tautomeric structure. a) о + CH3-NH-NH2 CO2C2H5 b) + CoH5-NH-NH2 OC2H5arrow_forwardIndicate the formula of the compound, that is the result of the N- alquilación (nucleofílic substitution), in which an additional lateral chain was formed (NH-CH2-COOMe). F3C. CF3 NH NH2 Br о OMe K2CO3, DABCO, DMFarrow_forwardSynthesis of 1-metilbenzotriazole from 1,2-diaminobenceno.arrow_forward
- Synthesis of 1-metilbenzotriazole.arrow_forwardIndicate the formula of the compound, that is the result of the N- alquilación (nucleofílic substitution), in which an additional lateral chain was formed (NH-CH2-COOMe). F3C. CF3 NH NH2 Br о OMe K2CO3, DABCO, DMFarrow_forwardIdentify the mechanism through which the following reaction will proceed and draw the major product. Part 1 of 2 Br KOH EtOH Through which mechanism will the reaction proceed? Select the single best answer. E1 E2 neither Part: 1/2 Part 2 of 2 Draw the major product formed as a result of the reaction. Click and drag to start drawing a structure. Xarrow_forward
- What is single-point calibration? Provide an example.arrow_forwardDraw the major product formed via an E1 pathway.arrow_forwardPart 9 of 9 Consider the products for the reaction. Identify the major and minor products. HO Cl The E stereoisomer is the major product and the Z stereoisomer is the minor product ▼ S major product minor productarrow_forward
- Consider the reactants below. Answer the following questions about the reaction mechanism and products. HO Clarrow_forwardjulietteyep@gmail.com X YSCU Grades for Juliette L Turner: Orc X 199 A ALEKS - Juliette Turner - Modul X A ALEKS - Juliette Turner - Modul x G butane newman projection - Gox + www-awa.aleks.com/alekscgi/x/Isl.exe/10_u-IgNslkr7j8P3jH-IBxzaplnN4HsoQggFsejpgqKoyrQrB2dKVAN-BcZvcye0LYa6eXZ8d4vVr8Nc1GZqko5mtw-d1MkNcNzzwZsLf2Tu9_V817y?10Bw7QYjlb il Scribbr citation APA SCU email Student Portal | Main Ryker-Learning WCU-PHARM D MySCU YSCU Canvas- SCU Module 4: Homework (Ch 9-10) Question 28 of 30 (1 point) | Question Attempt: 1 of Unlimited H₂SO heat OH The mechanism of this reaction involves two carbocation intermediates, A and B. Part 1 of 2 KHSO 4 rearrangement A heat B H₂O 2 OH Draw the structure of A. Check Search #t m Save For Later Juliet Submit Assignm 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessarrow_forwardThe electrons flow from the electron-rich atoms of the nucleophile to the electrons poor atoms of the alkyl halide. Identify the electron rich in the nucleophile. Enter the element symbol only, do not include any changes.arrow_forward
