
Concept explainers
(a)
Interpretation:
Condensed structural formula for the given name that has molecular formula of
Concept Introduction:
A common nomenclature of naming acetal and hemiacetal is done by finding the product of hydrolysis of the hemiacetal or acetal. The name of the carbonyl compound is added at the end to describe the name of such compound.
The general structure of hemiacetal is,
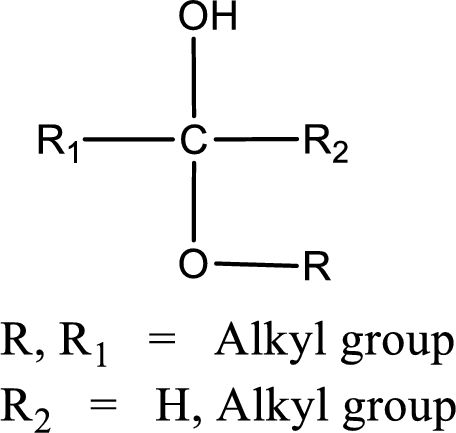
The general structure of acetal is,
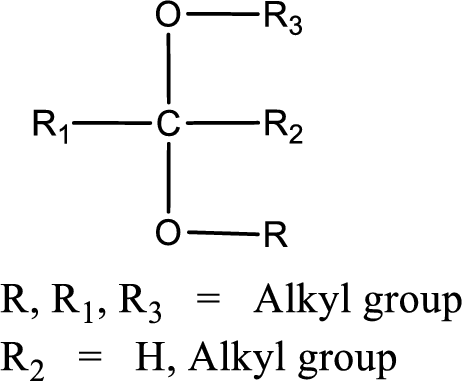
(b)
Interpretation:
Condensed structural formula for the given name that has molecular formula of
Concept Introduction:
A common nomenclature of naming acetal and hemiacetal is done by finding the product of hydrolysis of the hemiacetal or acetal. The name of the carbonyl compound is added at the end to describe the name of such compound.
The general structure of hemiacetal is,
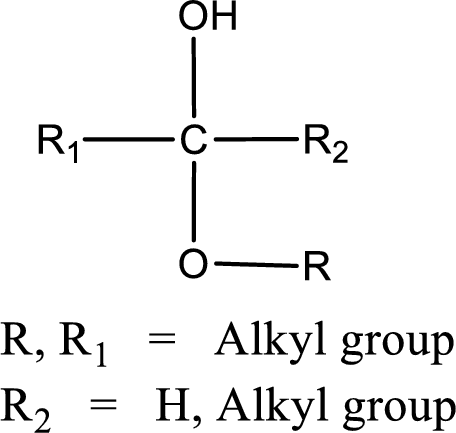
The general structure of acetal is,
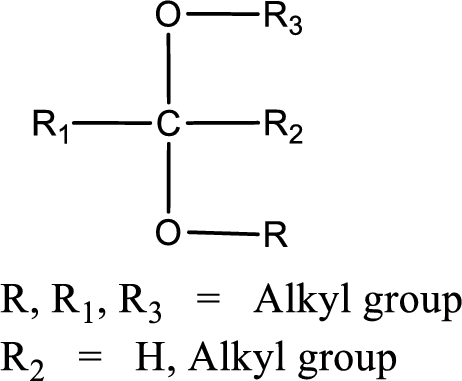
(c)
Interpretation:
Condensed structural formula for the given name that has molecular formula of
Concept Introduction:
A common nomenclature of naming acetal and hemiacetal is done by finding the product of hydrolysis of the hemiacetal or acetal. The name of the carbonyl compound is added at the end to describe the name of such compound.
The general structure of hemiacetal is,
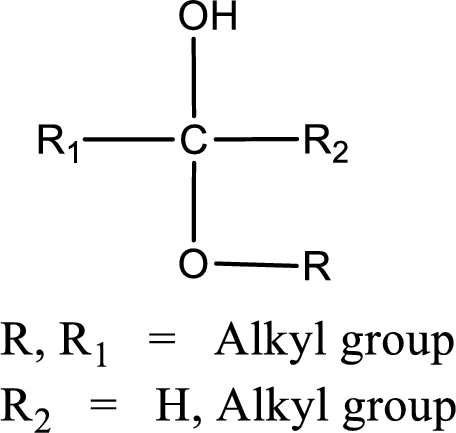
The general structure of acetal is,
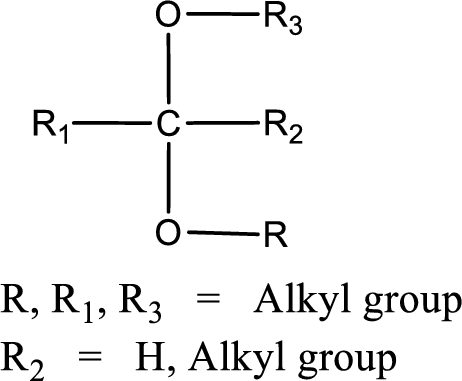
(d)
Interpretation:
Condensed structural formula for the given name that has molecular formula of
Concept Introduction:
A common nomenclature of naming acetal and hemiacetal is done by finding the product of hydrolysis of the hemiacetal or acetal. The name of the carbonyl compound is added at the end to describe the name of such compound.
The general structure of hemiacetal is,
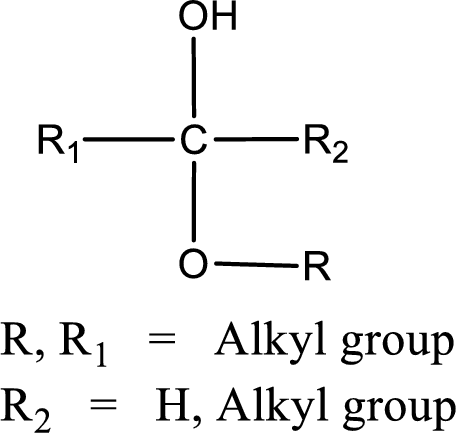
The general structure of acetal is,
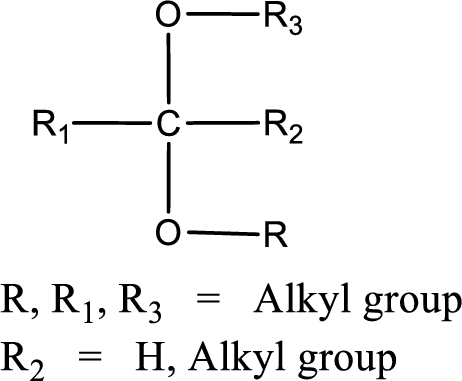
Trending nowThis is a popular solution!

Chapter 15 Solutions
General, Organic, and Biological Chemistry
- For the condensation reaction between Alamine and histamine, please help me write the amididation reaction mechanism. Then write the three letter code for the product of the reaction, then write the one letter code for the product of the reaction. arrow_forwardHow to draw the reaction mechasnism belowarrow_forwardName the following molecules with IUpacarrow_forward
- What is the molecular orbital for cyclopropenyl anion and is it aromatic, antiaromatic or nonaromatic?arrow_forwardUsing the chart describe the change from cystine to tyrosine and its impact on the protein. Using the chart describe the change from histidine to aspartic acid and its impact on the protein.arrow_forwardHow to get the predicted product of this reaction belowarrow_forward
- Please help me fill out the chart then using the chart describe the change from cystine to tyrosine and its impact on the protein. Then using the chart describe the change from histidine to aspartic acid.arrow_forwardWrite the Esterification reaction mechanism for acetic acid, and one propanol to make propanol ethanoate (molecule that gives peas its odor in flavor)arrow_forwardProvide solutionsarrow_forward
- Which of these compounds is Ester formed from the reaction of acetic acid and one propanol arrow_forwardDescribe the four labeled parts of the reaction diagram from the reaction of sucrose, breaking down with and without an enzyme.arrow_forwardHow to determine the product with mechanism showedarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





