
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
4th Edition
ISBN: 9781265982959
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 52P
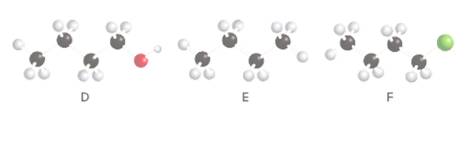
Rank the following compounds in order of increasing boiling point
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
An einstein is the amount of energy needed to dissociate 1 mole of a substance. If we have 0.58 moles, do we need 0.58 einsteins to dissociate that substance?
If the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.Data: Energy of each photon: 0.7835x10-18 J.
If the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.
Chapter 14 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
Ch. 14.1 - Prob. 14.1PCh. 14.2 - Prob. 14.1PPCh. 14.2 - Classify each hydroxyl group in sorbitol as 1°,...Ch. 14.2 - Prob. 14.3PCh. 14.2 - Prob. 14.4PCh. 14.3 - Prob. 14.2PPCh. 14.3 - Give the structure corresponding to each name a....Ch. 14.5 - Draw the products formed when each alcohol is...Ch. 14.5 - Prob. 14.6PCh. 14.5 - Prob. 14.7P
Ch. 14.5 - Prob. 14.4PPCh. 14.6 - Prob. 14.8PCh. 14.7 - Prob. 14.9PCh. 14.7 - Prob. 14.5PPCh. 14.7 - Prob. 14.10PCh. 14.7 - Name each ether. CH3OCH2CH2CH2CH3Ch. 14.7 - Prob. 14.11PCh. 14.8 - (a) Translate the hall and stick model of...Ch. 14.8 - Prob. 14.13PCh. 14.9 - Prob. 14.14PCh. 14.9 - Prob. 14.15PCh. 14.9 - Prob. 14.16PCh. 14.9 - Prob. 14.17PCh. 14.9 - Prob. 14.18PCh. 14.9 - Prob. 14.19PCh. 14.9 - Prob. 14.20PCh. 14.10 - Give the IUPAC name for each thiol.Ch. 14.10 - Prob. 14.21PCh. 14.10 - Prob. 14.22PCh. 14 - Classify each alcohol as 1°, 2°, or 3o a....Ch. 14 - Prob. 24PCh. 14 - Prob. 25PCh. 14 - Classify each halide hi A as 1°, 2°, or 3°. A is a...Ch. 14 - Prob. 27PCh. 14 - Draw the structure of a molecule that fits each...Ch. 14 - Draw the structure of the six constitutional...Ch. 14 - Draw the structure of the four constitutional...Ch. 14 - Draw the structure of l-propanethiol, a compound...Ch. 14 - Prob. 32PCh. 14 - Prob. 33PCh. 14 - Prob. 34PCh. 14 - Prob. 35PCh. 14 - Answer each question about alcohol B. Draw a...Ch. 14 - Prob. 37PCh. 14 - Give the IUAPC name for each alcohol.Ch. 14 - Prob. 39PCh. 14 - Prob. 40PCh. 14 - Prob. 41PCh. 14 - Prob. 42PCh. 14 - Draw the structures and give the IUPAC names for...Ch. 14 - Prob. 44PCh. 14 - Prob. 45PCh. 14 - Prob. 46PCh. 14 - Give the structure corresponding to each IUPAC...Ch. 14 - Give the structure corresponding to each IUPAC...Ch. 14 - Which compound in each pair has the higher boiling...Ch. 14 - Rank the compounds in order of increasing melting...Ch. 14 - Rank the following compounds in order of...Ch. 14 - Rank the following compounds in order of...Ch. 14 - Prob. 53PCh. 14 - Prob. 54PCh. 14 - Prob. 55PCh. 14 - Prob. 56PCh. 14 - Prob. 57PCh. 14 - Prob. 58PCh. 14 - Prob. 59PCh. 14 - Prob. 60PCh. 14 - Prob. 61PCh. 14 - Prob. 62PCh. 14 - Prob. 63PCh. 14 - Prob. 64PCh. 14 - Prob. 65PCh. 14 - Prob. 66PCh. 14 - Prob. 67PCh. 14 - Prob. 68PCh. 14 - Prob. 69PCh. 14 - Prob. 70PCh. 14 - Prob. 71PCh. 14 - Prob. 72PCh. 14 - Prob. 73PCh. 14 - Prob. 74PCh. 14 - Prob. 75PCh. 14 - Prob. 76PCh. 14 - Prob. 77PCh. 14 - Prob. 78PCh. 14 - Prob. 79PCh. 14 - Prob. 80PCh. 14 - With reference to the halogenated organic...Ch. 14 - Prob. 82PCh. 14 - Prob. 83PCh. 14 - Prob. 84PCh. 14 - Write out the chemical reaction that occurs when a...Ch. 14 - Prob. 86PCh. 14 - Prob. 87PCh. 14 - Lactic acid [CH3CH(OH)CO2H] gives sour milk its...Ch. 14 - Prob. 89PCh. 14 - Prob. 90PCh. 14 - Prob. 91PCh. 14 - Prob. 92PCh. 14 - Prob. 93PCh. 14 - Answer the following questions about alcohol B....Ch. 14 - Prob. 95CPCh. 14 - Dehydration of alcohol C forms two products of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The quantum yield of the photochemical decay of HI is 2. Calculating the moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. Calculate the number of Einsteins absorbed per mole knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. How many moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forward
- If the energy absorbed per mole of photons is 450 kJ, the number of Einsteins absorbed per 1 mole.arrow_forwardWhen propionic aldehyde in vapor form at 200 mmHg and 30°C is irradiated with radiation of wavelength 302 nm, the quantum yield with respect to the formation of CO is 0.54. If the intensity of the incident radiation is 1.5x10-3 W, find the rate of formation of CO.arrow_forwardDraw mechanismarrow_forward
- Does Avogadro's number have units?arrow_forwardExplain why the total E in an Einstein depends on the frequency or wavelength of the light.arrow_forwardIf the dissociation energy of one mole of O2 is 5.17 eV, determine the wavelength that must be used to dissociate it with electromagnetic radiation. Indicate how many Einstein's of this radiation are needed to dissociate 1 liter of O2 at 25°C and 1 atm of pressure.Data: 1 eV = 96485 kJ mol-1; R = 0.082 atm L K-1; c = 2.998x108 m s-1; h = 6.626x10-34 J s; NA = 6.022x 1023 mol-1arrow_forward
- Indicate the number of Einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy (wavelength 475 nm).arrow_forwardIndicate the number of einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy?arrow_forwardA unit used in photochemistry is the einstein. If 400 kJ mol-1 of energy has been absorbed, how many einsteins is this equivalent to?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY