
Concept explainers
(a)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In
In organic chemistry, reduction reaction is referred to the number
Alcohols do undergo
Primary alcohol:
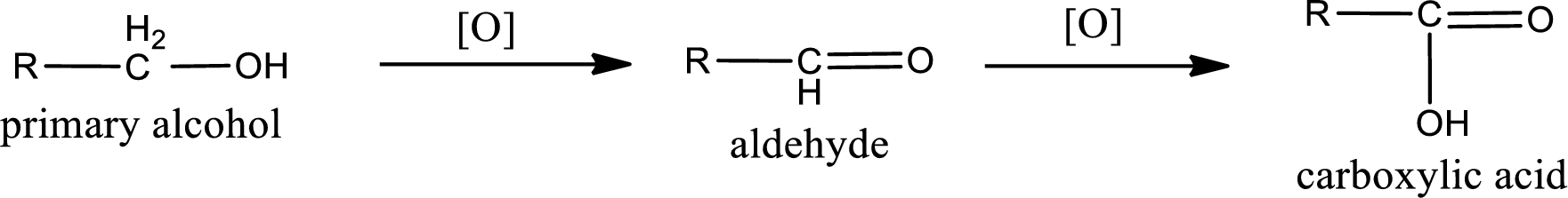
Secondary alcohol:
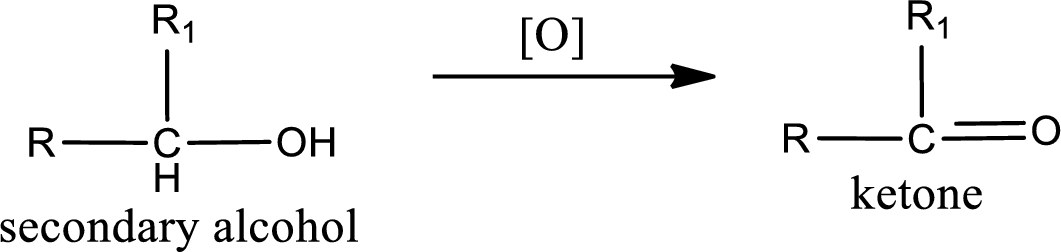
(a)
Answer to Problem 14.67EP
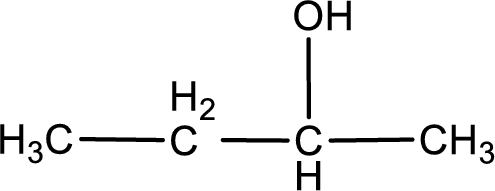
The structure of alcohol is,
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
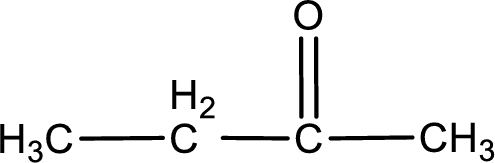
The given compound is a ketone. This has to be formed from a secondary alcohol only on oxidation. The structure of the secondary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
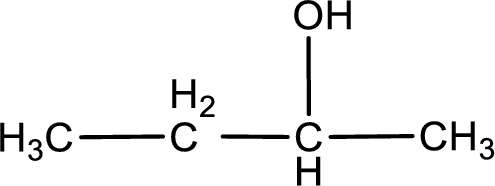
Structure of alcohol is drawn from which the given product is obtained by oxidation.
(b)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
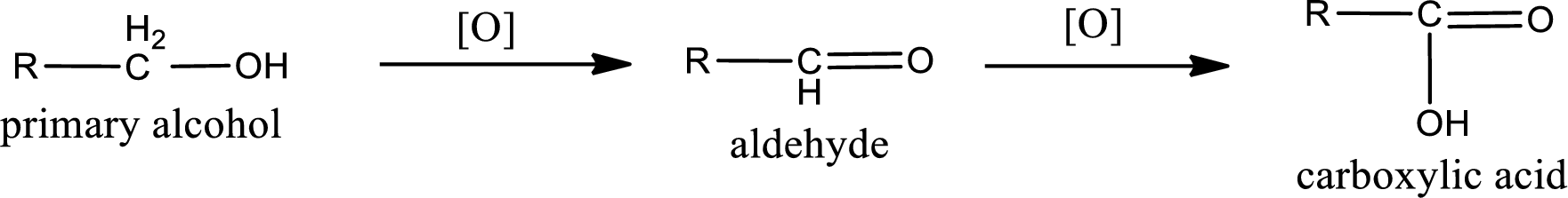
Alcohols do undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and this id further oxidized to give carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Primary alcohol:
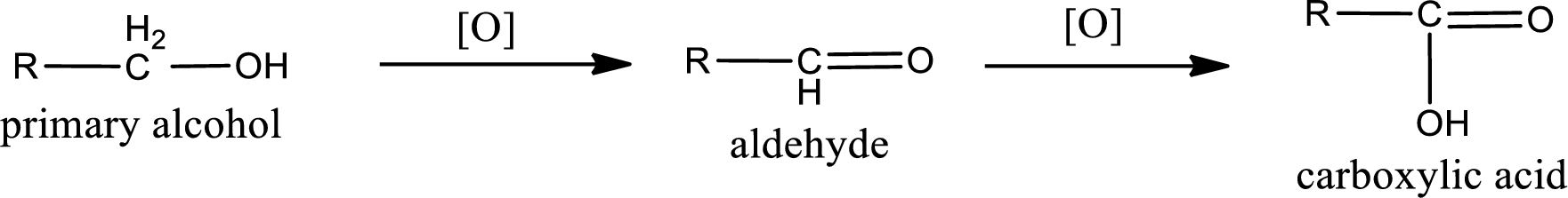
Secondary alcohol:
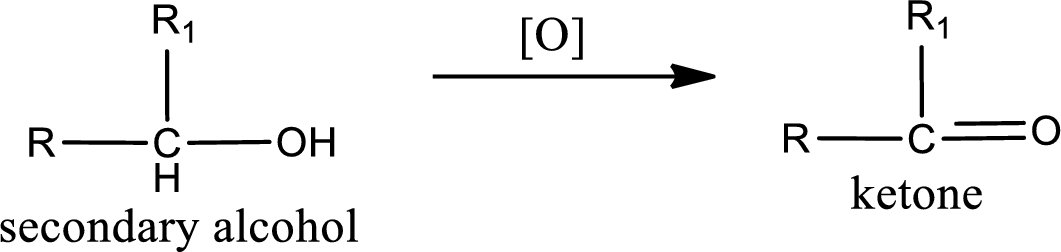
(b)
Answer to Problem 14.67EP
The structure of alcohol is,
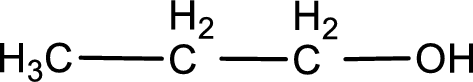
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
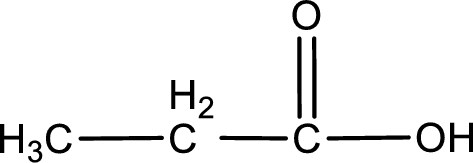
The given compound is a carboxylic acid. This has to be formed from aldehyde by oxidation. The aldehyde must have formed from primary alcohol by oxidation. The structure of the primary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
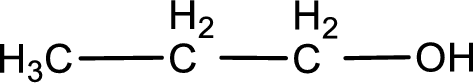
Structure of alcohol is drawn from which the given product is obtained by oxidation.
(c)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
Alcohols do undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and this id further oxidized to give carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Primary alcohol:
Secondary alcohol:
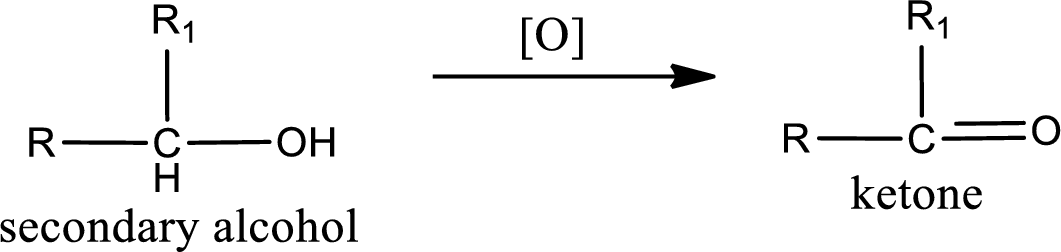
(c)
Answer to Problem 14.67EP
The structure of alcohol is,
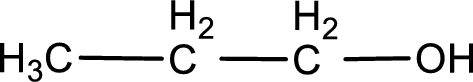
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
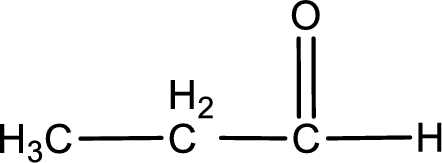
The given compound is a aldehyde. The aldehyde must have formed from primary alcohol by oxidation. The structure of the primary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
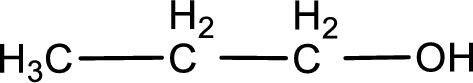
Structure of alcohol is drawn from which the given product is obtained by oxidation.
(d)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
Alcohols do undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and this id further oxidized to give carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Primary alcohol:
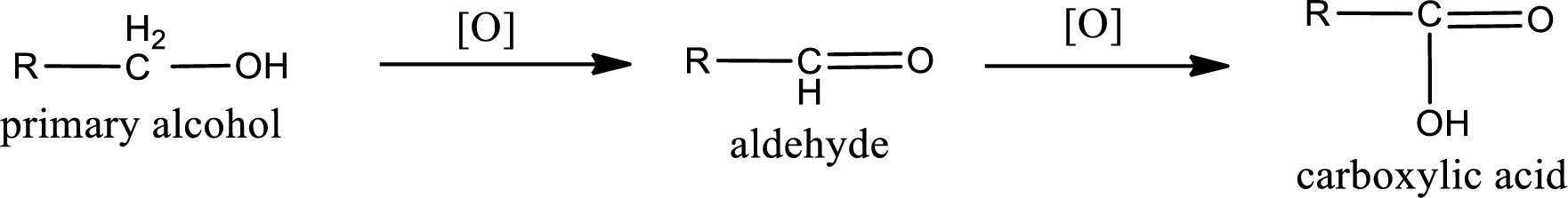
Secondary alcohol:
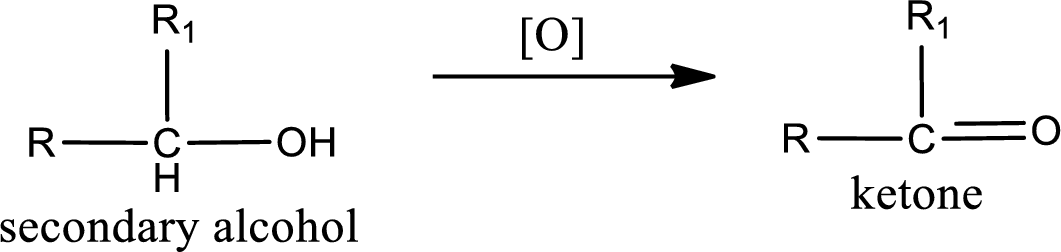
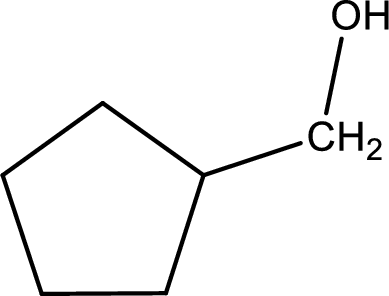
(d)
Answer to Problem 14.67EP
The structure of alcohol is,
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
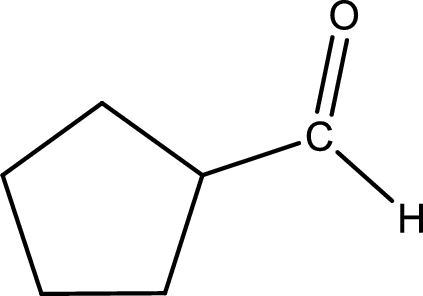
The given compound is a aldehyde. The aldehyde must have formed from primary alcohol by oxidation. The structure of the primary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
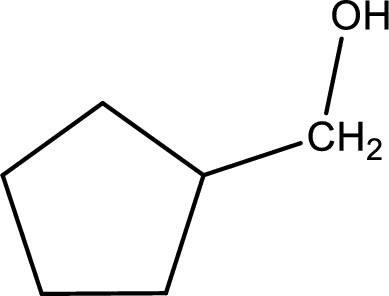
Structure of alcohol is drawn from which the given product is obtained by oxidation.
Want to see more full solutions like this?
Chapter 14 Solutions
General, Organic, and Biological Chemistry Seventh Edition
- For the condensation reaction between Alamine and histamine, please help me write the amididation reaction mechanism. Then write the three letter code for the product of the reaction, then write the one letter code for the product of the reaction. arrow_forwardHow to draw the reaction mechasnism belowarrow_forwardName the following molecules with IUpacarrow_forward
- What is the molecular orbital for cyclopropenyl anion and is it aromatic, antiaromatic or nonaromatic?arrow_forwardUsing the chart describe the change from cystine to tyrosine and its impact on the protein. Using the chart describe the change from histidine to aspartic acid and its impact on the protein.arrow_forwardHow to get the predicted product of this reaction belowarrow_forward
- Please help me fill out the chart then using the chart describe the change from cystine to tyrosine and its impact on the protein. Then using the chart describe the change from histidine to aspartic acid.arrow_forwardWrite the Esterification reaction mechanism for acetic acid, and one propanol to make propanol ethanoate (molecule that gives peas its odor in flavor)arrow_forwardProvide solutionsarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





