
(a)
Interpretation: The products for the given reaction have to be identified.
Concept Introduction:
Ketals:
Ketals are used to protect the carbonyl group of the
In the following example, the carbonyl group of the acetone has been protected as ketal by using ethylene glycol.
Example:
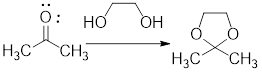
In acidic condition, a ketone reacts with two molecules of alcohol or a molecule of
General scheme:
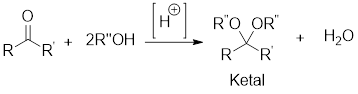
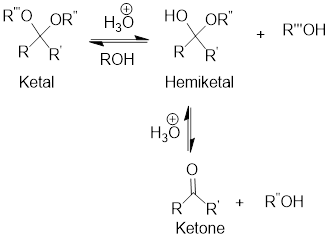
Hydrolysis of Ketal:
Hydrolysis of ketal gives back the corresponding alcohol and the corresponding ketone from which the ketal has been formed. The hydrolysis takes place only in acidic conditions. So, the hydrolysis of ketal is an acid-catalyzed hydrolysis reaction.
General scheme:
(b)
Interpretation: The products for the given reaction have to be found.
Concept Introduction:
Ketals:
Ketals are used to protect the carbonyl group of the ketone.
In the following example, the carbonyl group of the acetone has been protected as ketal by using ethylene glycol.
Example:

In acidic condition, a ketone reacts with two molecules of alcohol or a molecule of diol to form ketal or cyclic ketal respectively.
General scheme:
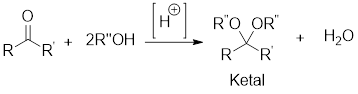
Hydrolysis of Ketal:
Hydrolysis of ketal gives back the corresponding alcohol and the corresponding ketone from which the ketal has been formed. The hydrolysis takes place only in acidic conditions. So, the hydrolysis of ketal is an acid-catalyzed hydrolysis reaction.
General scheme:

Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
EP ESSENTIAL ORG.CHEM.-MOD.MASTERING
- Indicate the reagent needed to go from cyclopentane-CH2-CHO to cyclopentane-CH2-CH=CH-C6H5.arrow_forwardesc Write the systematic name of each organic molecule: structure CH3 CH3-C=CH2 CH3-CH2-C-CH2-CH3 CH-CH3 CH3 ☐ ☐ ☐ CI-CH-CH=CH2 Explanation Check F1 F2 name 80 F3 F4 F5 F6 A 7 ! 2 # 3 4 % 5 6 & 7 Q W E R Y FT 2025 Mcarrow_forwardTwo reactants X and Z are required to convert the compound CH3-CH2-CH2Br to the compound CH3-CH2-CH=P(C6H5)3. State reactants X and Z.arrow_forward
- 2. Write a reasonable mechanism that converts the reactants into the products. Avoid issues A-U from the previous page. You can use any number of steps (it does not have to be a one-step mechanism). Do not use any other chemicals (solvents, acids, bases, etc.) in your mechanism. 2 2 H ΗΘarrow_forwardFor the following reaction, the partial pressures were determined for the reaction components as shownbelow. Is the reaction at equilibrium? If not, in which direction will it proceed?I2 (g) + Cl2 (g) ⇋ 2 ICl (g) Kp = 81.9 partial pressures: I2 = 0.114 atm; Cl2 = 0.102 atm; ICl = 0.355 atmarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. H3O+ + Oarrow_forward
- Write the law of mass action for the following quilibrium 2N2O5(g) --> 4 NO2(g) + O2(g).arrow_forwardState the products (formulas) of the reaction of acetophenone with iodine and NaOH.arrow_forwardCh. 4- Precipitation Reactions Worksheet Write balanced, complete ionic, and net ionic equations for the following reactions that mav produce precipitates. Use NR to indicate no reaction Ave 1\ +3 =6 Fe + V-2 Na S04 13. Write the balanced equation for the reaction of iron (III) phosphate with sodium sulfate to make iron (III) sulfate and sodium phosphate. 2FePO4 + M, Soy a) If you perform this reaction with 25 grams of iron (III) phosphate and an excess of sodium sulfate, how many grams of iron (III) sulfate can you make? 21 Fe 2 3x 1 Na 3 25g Fe Ingle 150,829 Indes 2 nol 3 1335 349.89 35.90 Ihol & Sanz Fez Bak heck 3x1 50ab) If 18.5 grams of iron (III) sulfate are actually made when you do this reaction, what is your Poy percent yield? 118.5 259-1-100 51.4% (0.74)x100610 335 If you do this reaction with 15 grams of sodium sulfate and get a 65.0% yield, how many grams of sodium phosphate will you make? 10.59 14. Ammonia is produced from the reaction of nitrogen and hydrogen according…arrow_forward
- == Functional Groups Identifying and drawing hemiacetals and acetals In the drawing area below, create an acetal with 1 isopropoxy group, 1 hydroxyl group, and a total of 10 carbon atoms. Explanation Click and drag to start drawing a structure. Check G +arrow_forwardState the products (formulas) of the reaction of acetophenone with iodine and NaOH.arrow_forwardExplanation Check Draw the skeletal ("line") structure of 5-hydroxy-4-methyl-2-pentanone. Click and drag to start drawing a structure. Х © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cer ☐ : Carrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





