
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
4th Edition
ISBN: 9781265982959
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 12.9, Problem 12.22P
Interpretation Introduction
(a)
Interpretation:
The
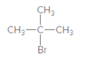
Concept introduction:
An alkyl bromide is obtained by reacting the alkane with bromine in presence of light or heating.
Interpretation Introduction
(b)
Interpretation:
The alkane required to produce the following compound should be determined:
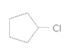
Concept introduction:
A cycloalkyl chloride is obtained by reacting to the cycloalkane with chlorine in presence of light or heating.
Interpretation Introduction
(c)
Interpretation:
The alkane required to produce the following compound should be determined:
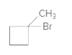
Concept introduction:
A cycloalkyl bromide is obtained by reacting to the cycloalkane with bromine in presence of light or heating.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw Newman projects for each of the following molecules with 3 different rotational
angles from carbon 2 to carbon 3. Rank your structures from lowest to highest energy. What
causes the energy differences? Label the overlap.
a.
b.
Br
OH
C.
Br
Br
Draw the stereoisomers of 3,5-diethylcylopentane. Identify the different relationships
between each molecules (diasteromers, enantiomers, meso compounds, etc.)
Please correct answer and don't use hand rating
Chapter 12 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
Ch. 12.1 - How many hydrogen atoms are present in each...Ch. 12.1 - Which formulas represent acyclic alkanes and which...Ch. 12.2 - Are the compounds in each pair constitutional...Ch. 12.2 - Draw two isomers with molecular formula C6H14 that...Ch. 12.2 - Prob. 12.3PCh. 12.2 - Classify the carbon atoms in each compound as 1°,...Ch. 12.2 - Prob. 12.4PCh. 12.2 - Prob. 12.5PCh. 12.2 - Prob. 12.6PCh. 12.2 - Prob. 12.7P
Ch. 12.2 - Prob. 12.8PCh. 12.2 - Prob. 12.9PCh. 12.4 - Give the IUPAC name for each compound.Ch. 12.4 - Prob. 12.10PCh. 12.4 - Give the IUPAC name for each compound....Ch. 12.4 - Prob. 12.5PPCh. 12.4 - Prob. 12.6PPCh. 12.4 - Prob. 12.12PCh. 12.5 - Prob. 12.13PCh. 12.5 - Prob. 12.14PCh. 12.5 - Give the IUPAC name for each compound.Ch. 12.5 - Prob. 12.15PCh. 12.7 - Answer the following questions about pentane...Ch. 12.7 - Prob. 12.17PCh. 12.7 - Prob. 12.18PCh. 12.8 - Prob. 12.19PCh. 12.9 - Prob. 12.20PCh. 12.9 - Prob. 12.21PCh. 12.9 - Prob. 12.22PCh. 12 - Prob. 23PCh. 12 - Prob. 24PCh. 12 - The waxy coating on the surface of apple skins...Ch. 12 - Prob. 26PCh. 12 - Classify each carbon as 1°, 2°, 3°, or 4°. a....Ch. 12 - Give the structure of an alkane that fits each...Ch. 12 - Prob. 29PCh. 12 - Label each pair of compounds as constitutional...Ch. 12 - Consider compounds A, B, and C. Label each pair of...Ch. 12 - Consider compounds D,E, and F. Label each pair of...Ch. 12 - Prob. 33PCh. 12 - Consider compounds A-E and label each pair of...Ch. 12 - Draw structures that fit the following...Ch. 12 - Draw the five constitutional isomers having...Ch. 12 - Prob. 37PCh. 12 - Prob. 38PCh. 12 - Draw the five constitutional isomers that have...Ch. 12 - Pristane is a high molecular weight alkane found...Ch. 12 - Give the IUPAC name for each of the five...Ch. 12 - Give the IUPAC name for each of the five cyclic...Ch. 12 - Give the IUPAC name for each compoundCh. 12 - Give the IUPAC name for each compound.Ch. 12 - Give the IUPAC name for each compound. c....Ch. 12 - Give the IUPAC name for each compound.Ch. 12 - Give the IUPAC name for each cycloalkane.Ch. 12 - Give the IUPAC name for each ccycloalkane.Ch. 12 - Prob. 49PCh. 12 - Give the structure corresponding to each IUPAC...Ch. 12 - Each of the following IUPAC names is incorrect....Ch. 12 - Each of the following IUPAC names is incorrect....Ch. 12 - Draw three constitutional isomers having molecular...Ch. 12 - Draw four constitutional isomers having molecular...Ch. 12 - Draw a skeletal structure for each compound octane...Ch. 12 - Convert each compound to a skeletal structure CH3(...Ch. 12 - Convert each skeletal structure to a complete...Ch. 12 - Convert each skeletal structure to a complete...Ch. 12 - Which compound in each pair has the higher melting...Ch. 12 - Which compound in each pair has the higher boiling...Ch. 12 - Branching in an alkane chain decreases surface...Ch. 12 - Explain why the boiling points of heptane [CH3( CH...Ch. 12 - Explain why hexane is more soluble in...Ch. 12 - Mineral oil and Vaseline are both mixtures of...Ch. 12 - Write a balanced equation for the combustion of...Ch. 12 - Write a balanced equation for the combustion of...Ch. 12 - Write a balanced equation for the incomplete...Ch. 12 - Prob. 68PCh. 12 - Prob. 69PCh. 12 - Prob. 70PCh. 12 - Prob. 71PCh. 12 - Prob. 72PCh. 12 - Prob. 73PCh. 12 - Prob. 74PCh. 12 - Prob. 75PCh. 12 - Prob. 76PCh. 12 - Prob. 77PCh. 12 - Prob. 78PCh. 12 - Prob. 79PCh. 12 - A major component of animal fat is tristearin, (a)...Ch. 12 - Answer the following questions about the alkane...Ch. 12 - Prob. 82PCh. 12 - Answer the questions in Problem 12.81 for the...Ch. 12 - Prob. 84PCh. 12 - Prob. 85CPCh. 12 - Draw the structure of the 12 constitutional...Ch. 12 - Cyclopentane has a higher boiling point than...Ch. 12 - Prob. 88CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please correct answer and don't use hand rating and don't use Ai solutionarrow_forwardShow work....don't give Ai generated solutionarrow_forwardIs it possible to do the following reduction in one step? If so, add the necessary reagents and catalysts to the reaction arrow. If not, check the box under the drawing area. T G टे 13arrow_forward
- Convert the following structures into a chair representation. Then conduct a chair flip. Cl a. b. C\.... оarrow_forwardAktiv Learning App Cengage Digital Learning Part of Speech Table for Assign x o Mail-Karen Ento-Outlook * + app.aktiv.com Your Aktiv Learning trial expires on 02/06/25 at 01:15 PM Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Problem 17 of 30 Drawing Arrows heat 4 O M B D 5x H H Und Settings H Done :0: H Jararrow_forwardConvert the following chairs into ring representations: a. Brz b.arrow_forward
- Drawing Arrows 1 I I 1 heat 1 51 MO + Drag To Und Settings Done 0 0 Jan 31 3:5arrow_forwardDon't used hand raitingarrow_forwardGramicidin A can adopt more than one structure; NMR spectroscopy has revealed an “end-to-end” dimer form, and x-ray crystallography has revealed an “anti-parallel double- helical” form. Briefly outline and describe an experimentalapproach/strategy to investigate WHICH configuration (“end-to-end dimer” vs “anti-paralleldouble helical”) gramicidin adopts in an actual lipid bilayer.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning