
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
7th Edition
ISBN: 9780100853188
Author: STOKER
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.75EP
For each of the
a. 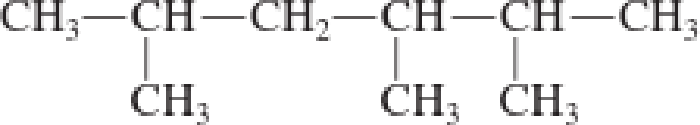
b. 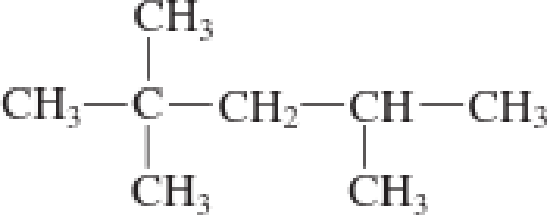
c. 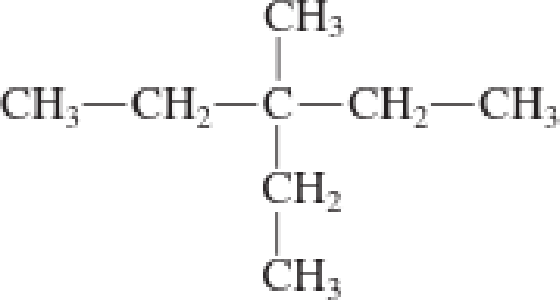
d. 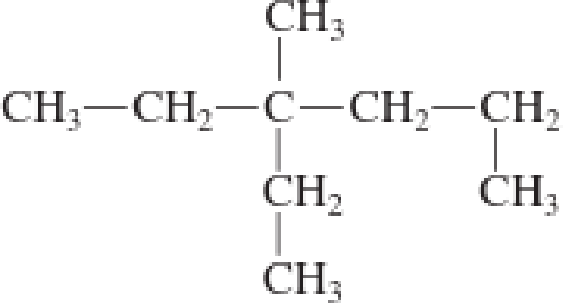
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
None
In the phase diagram of steel (two components Fe and C), region A is the gamma austenite solid and region B contains the gamma solid and liquid. Indicate the degrees of freedom that the fields A and B have,
For a condensed binary system in equilibrium at constant pressure, indicate the maximum number of phases that can exist.
Chapter 12 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
Ch. 12.1 - Prob. 1QQCh. 12.1 - Prob. 2QQCh. 12.2 - Prob. 1QQCh. 12.2 - Prob. 2QQCh. 12.3 - Prob. 1QQCh. 12.3 - Prob. 2QQCh. 12.4 - Prob. 1QQCh. 12.4 - Prob. 2QQCh. 12.4 - Prob. 3QQCh. 12.5 - Prob. 1QQ
Ch. 12.5 - Prob. 2QQCh. 12.5 - Prob. 3QQCh. 12.6 - Prob. 1QQCh. 12.6 - Prob. 2QQCh. 12.6 - Prob. 3QQCh. 12.6 - Prob. 4QQCh. 12.7 - Prob. 1QQCh. 12.7 - Prob. 2QQCh. 12.8 - Prob. 1QQCh. 12.8 - Prob. 2QQCh. 12.8 - Prob. 3QQCh. 12.8 - Prob. 4QQCh. 12.8 - Prob. 5QQCh. 12.8 - Prob. 6QQCh. 12.8 - Prob. 7QQCh. 12.9 - Prob. 1QQCh. 12.9 - Prob. 2QQCh. 12.10 - Prob. 1QQCh. 12.10 - Prob. 2QQCh. 12.11 - Prob. 1QQCh. 12.11 - Prob. 2QQCh. 12.11 - Prob. 3QQCh. 12.12 - Prob. 1QQCh. 12.12 - Prob. 2QQCh. 12.12 - Prob. 3QQCh. 12.13 - Prob. 1QQCh. 12.13 - Prob. 2QQCh. 12.13 - Prob. 3QQCh. 12.14 - Prob. 1QQCh. 12.14 - Prob. 2QQCh. 12.14 - Prob. 3QQCh. 12.15 - Prob. 1QQCh. 12.15 - Prob. 2QQCh. 12.16 - Prob. 1QQCh. 12.16 - Prob. 2QQCh. 12.16 - Prob. 3QQCh. 12.17 - Prob. 1QQCh. 12.17 - Prob. 2QQCh. 12.17 - Prob. 3QQCh. 12.17 - Prob. 4QQCh. 12.18 - Prob. 1QQCh. 12.18 - Prob. 2QQCh. 12.18 - Prob. 3QQCh. 12.18 - Prob. 4QQCh. 12 - Prob. 12.1EPCh. 12 - Prob. 12.2EPCh. 12 - Prob. 12.3EPCh. 12 - Prob. 12.4EPCh. 12 - Indicate whether each of the following situations...Ch. 12 - Indicate whether each of the following situations...Ch. 12 - Prob. 12.7EPCh. 12 - Prob. 12.8EPCh. 12 - What is the difference between a saturated...Ch. 12 - Prob. 12.10EPCh. 12 - Prob. 12.11EPCh. 12 - Prob. 12.12EPCh. 12 - Prob. 12.13EPCh. 12 - Prob. 12.14EPCh. 12 - Prob. 12.15EPCh. 12 - Prob. 12.16EPCh. 12 - Prob. 12.17EPCh. 12 - Prob. 12.18EPCh. 12 - Convert the expanded structural formulas in...Ch. 12 - Prob. 12.20EPCh. 12 - Prob. 12.21EPCh. 12 - Prob. 12.22EPCh. 12 - Prob. 12.23EPCh. 12 - Prob. 12.24EPCh. 12 - Prob. 12.25EPCh. 12 - Prob. 12.26EPCh. 12 - Indicate whether each of the following would be...Ch. 12 - Indicate whether each of the following would be...Ch. 12 - Prob. 12.29EPCh. 12 - Prob. 12.30EPCh. 12 - Prob. 12.31EPCh. 12 - Prob. 12.32EPCh. 12 - Prob. 12.33EPCh. 12 - How many of the numerous seven-carbon alkane...Ch. 12 - Prob. 12.35EPCh. 12 - For each of the following pairs of structures,...Ch. 12 - Prob. 12.37EPCh. 12 - Prob. 12.38EPCh. 12 - Prob. 12.39EPCh. 12 - Prob. 12.40EPCh. 12 - Prob. 12.41EPCh. 12 - What is the name of the IUPAC prefix associated...Ch. 12 - What is the IUPAC name for each of the following...Ch. 12 - What is the IUPAC name for each of the following...Ch. 12 - Prob. 12.45EPCh. 12 - What is the chemical formula for each of the...Ch. 12 - Prob. 12.47EPCh. 12 - Prob. 12.48EPCh. 12 - Prob. 12.49EPCh. 12 - Prob. 12.50EPCh. 12 - Prob. 12.51EPCh. 12 - Prob. 12.52EPCh. 12 - Draw a condensed structural formula for each of...Ch. 12 - Draw a condensed structural formula for each of...Ch. 12 - Prob. 12.55EPCh. 12 - Prob. 12.56EPCh. 12 - Explain why the name given for each of the...Ch. 12 - Prob. 12.58EPCh. 12 - Indicate whether or not the two alkanes in each of...Ch. 12 - Prob. 12.60EPCh. 12 - How many of the 18 C8 alkane constitutional...Ch. 12 - Prob. 12.62EPCh. 12 - Prob. 12.63EPCh. 12 - Prob. 12.64EPCh. 12 - Prob. 12.65EPCh. 12 - Prob. 12.66EPCh. 12 - Do the line-angle structural formulas in each of...Ch. 12 - Do the line-angle structural formulas in each of...Ch. 12 - Convert each of the condensed structural formulas...Ch. 12 - Convert each of the condensed structural formulas...Ch. 12 - Assign an IUPAC name to each of the compounds in...Ch. 12 - Prob. 12.72EPCh. 12 - Prob. 12.73EPCh. 12 - Prob. 12.74EPCh. 12 - For each of the alkane structures in Problem...Ch. 12 - Prob. 12.76EPCh. 12 - Prob. 12.77EPCh. 12 - Prob. 12.78EPCh. 12 - Prob. 12.79EPCh. 12 - Prob. 12.80EPCh. 12 - Prob. 12.81EPCh. 12 - Prob. 12.82EPCh. 12 - Draw condensed structural formulas for the...Ch. 12 - Draw condensed structural formulas for the...Ch. 12 - To which carbon atoms in a hexane molecule can...Ch. 12 - Prob. 12.86EPCh. 12 - Prob. 12.87EPCh. 12 - Prob. 12.88EPCh. 12 - Prob. 12.89EPCh. 12 - Prob. 12.90EPCh. 12 - Prob. 12.91EPCh. 12 - Prob. 12.92EPCh. 12 - Prob. 12.93EPCh. 12 - Using the general formula for a cycloalkane,...Ch. 12 - Prob. 12.95EPCh. 12 - Prob. 12.96EPCh. 12 - Prob. 12.97EPCh. 12 - Prob. 12.98EPCh. 12 - How many secondary carbon atoms are present in...Ch. 12 - Prob. 12.100EPCh. 12 - Prob. 12.101EPCh. 12 - Assign an IUPAC name to each of the following...Ch. 12 - Prob. 12.103EPCh. 12 - What is wrong with each of the following attempts...Ch. 12 - Draw line-angle structural formulas for the...Ch. 12 - Draw line-angle structural formulas for the...Ch. 12 - Prob. 12.107EPCh. 12 - Prob. 12.108EPCh. 12 - Prob. 12.109EPCh. 12 - Prob. 12.110EPCh. 12 - Determine the number of constitutional isomers...Ch. 12 - Determine the number of constitutional isomers...Ch. 12 - Prob. 12.113EPCh. 12 - Determine whether cistrans isomerism is possible...Ch. 12 - Prob. 12.115EPCh. 12 - Prob. 12.116EPCh. 12 - Prob. 12.117EPCh. 12 - Indicate whether the members of each of the...Ch. 12 - Prob. 12.119EPCh. 12 - Prob. 12.120EPCh. 12 - Prob. 12.121EPCh. 12 - Prob. 12.122EPCh. 12 - Which member in each of the following pairs of...Ch. 12 - Prob. 12.124EPCh. 12 - Prob. 12.125EPCh. 12 - Prob. 12.126EPCh. 12 - Answer the following questions about the...Ch. 12 - Prob. 12.128EPCh. 12 - Prob. 12.129EPCh. 12 - Prob. 12.130EPCh. 12 - Prob. 12.131EPCh. 12 - Prob. 12.132EPCh. 12 - Prob. 12.133EPCh. 12 - Write structural formulas for all the possible...Ch. 12 - Assign an IUPAC name to each of the following...Ch. 12 - Assign an IUPAC name to each of the following...Ch. 12 - Prob. 12.137EPCh. 12 - Prob. 12.138EPCh. 12 - Prob. 12.139EPCh. 12 - Draw structural formulas for the following...Ch. 12 - Prob. 12.141EPCh. 12 - Prob. 12.142EPCh. 12 - Prob. 12.143EPCh. 12 - Prob. 12.144EPCh. 12 - Prob. 12.145EPCh. 12 - Prob. 12.146EPCh. 12 - Prob. 12.147EPCh. 12 - Prob. 12.148EP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Part V. Label ad match the carbons in compounds Jane and Diane w/ the corresponding peak no. in the Spectra (Note: use the given peak no. To label the carbons, other peak no are intentionally omitted) 7 4 2 -0.13 -0.12 -0.11 -0.10 -0.08 8 CI Jane 1 -0.09 5 210 200 190 180 170 160 150 140 130 120 110 100 -8 90 f1 (ppm) 11 8 172.4 172.0 f1 (ppr HO CI NH Diane 7 3 11 80 80 -80 -R 70 60 60 2 5 -8 50 40 8. 170 160 150 140 130 120 110 100 90 -0 80 70 20 f1 (ppm) 15 30 -20 20 -60 60 -0.07 -0.06 -0.05 -0.04 -0.03 -0.02 -0.01 -0.00 -0.01 10 -0.17 16 15 56 16 -0.16 -0.15 -0.14 -0.13 -0.12 -0.11 -0.10 -0.09 -0.08 -0.07 -0.06 -0.05 -0.04 17.8 17.6 17.4 17.2 17.0 f1 (ppm) -0.03 -0.02 550 106 40 30 20 20 -0.01 -0.00 F-0.01 10 0arrow_forwardConsider the reaction of 2-methylpropane with a halogen. With which halogen will the product be almost exclusively 2-halo-2-methylpropane? 1. F2 2. Cl2 3. Br2 4. I2arrow_forwardNonearrow_forward
- Nonearrow_forwardn Feb 3 A T + 4. (2 pts) Draw the structure of the major component of the Limonene isolated. Explain how you confirmed the structure. 5. (2 pts) Draw the fragment corresponding to the base peak in the Mass spectrum of Limonene. 6. (1 pts) Predict the 1H NMR spectral data of R-Limonene. Proton NMR: 5.3 pon multiplet (H Ringarrow_forwardPart VI. Ca H 10 O is the molecular formula of compound Tom and gives the in the table below. Give a possible structure for compound Tom. 13C Signals summarized C1 C2 C3 C4 C5 C6 C7 13C shift (ppm) 23.5 27.0 33.0 35.8 127 162 205 DEPT-90 + DEPT-135 + +arrow_forward
- 2. Using the following data to calculate the value of AvapH o of water at 298K. AvapH o of water at 373K is 40.7 kJ/mol; molar heat capacity of liquid water at constant pressure is 75.2J mol-1 K-1 and molar heat capacity of water vapor at constant pressure is 33.6 J mol-1 K-1.arrow_forwardPart VII. Below are the 'HNMR 13 3 C-NMR, COSY 2D- NMR, and HSQC 20-NMR (Similar with HETCOR but axes are reversed) spectra of an organic compound with molecular formula C6H13 O. Assign chemical shift values to the H and c atoms of the compound. Find the structure. Show complete solutions. Predicted 1H NMR Spectrum ли 4.7 4.6 4.5 4.4 4.3 4.2 4.1 4.0 3.9 3.8 3.7 3.6 3.5 3.4 3.3 3.2 3.1 3.0 2.9 2.8 2.7 2.6 2.5 2.4 2.3 2.2 2.1 2.0 1.9 1.8 1.7 1.6 1.5 1.4 1.3 1.2 1.1 1.0 0.9 0.8 f1 (ppm)arrow_forward3. Draw the expanded structural formula, the condensed structural formula, and the skeletal structural formula for 2-pentene. expanded structure: Condensed structure: Skeletal formula: 4. Draw the expanded structural formula, the condensed structural formula, and the skeletal structural formula for 2-methyl-3-heptene. expanded structure: Condensed structure: Skeletal formula: following structurearrow_forward
- Part IV. Propose a plausible Structure w/ the following descriptions: a) A 5-carbon hydrocarbon w/ a single peak in its proton decoupled the DEPT-135 Spectrum shows a negative peak C-NMR spectrum where b) what cyclohexane dione isomer gives the largest no. Of 13C NMR signals? c) C5H120 (5-carbon alcohol) w/ most deshielded carbon absent in any of its DEPT Spectivaarrow_forward13C NMR is good for: a) determining the molecular weight of the compound b) identifying certain functional groups. c) determining the carbon skeleton, for example methyl vs ethyl vs propyl groups d) determining how many different kinds of carbon are in the moleculearrow_forward6 D 2. (1 pt) Limonene can be isolated by performing steam distillation of orange peel. Could you have performed this experiment using hexane instead of water? Explain. 3. (2 pts) Using GCMS results, analyze and discuss the purity of the Limonene obtained from the steam distillation of orange peel.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License