
Concept explainers
(a)
Interpretation: The number of rings and number of pi bonds in A is to be determined. And one possible structure is to be drawn.
Concept introduction: Degree of unsaturation is used to determine the total number of rings and pi bonds present in compound by just looking at the molecular formula. It does not specify the total number of rings and total number of pi bonds individually.
Answer to Problem 12.35P
The number of pi bonds and number of rings in A is one. The possible structure is given in Figure 1.
Explanation of Solution
For compound A:
Before hydrogenation, the molecular formula is
The maximum number of
The maximum number of
The number of
Substitute the values of maximum number of
The degree of unsaturation is calculated by the formula,
Hence, the degree of unsaturation before hydrogenation is two.
After hydrogenation, the molecular formula is
The maximum number of
The maximum number of
The number
Substitute the values of maximum number of
The degree of unsaturation is calculated by the formula,
Hence, the degree of unsaturation after hydrogenation is one.
The number of pi bonds in A is calculated by the formula,
Substitute the values of degree of unsaturation before hydrogenation and degree of unsaturation after hydrogenation in the above formula.
Hence, the number of pi bonds is one.
Number of rings is calculated by the formula,
Substitute the values of degree of unsaturation and number of pi bonds in the above formula.
Hence, the number of rings is one.
The possible structure for A is,
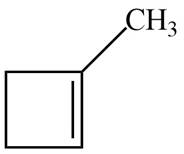
Figure 1
The number of pi bonds and number of rings in A is one. The possible structure is given in Figure 1.
(b)
Interpretation: The number of rings and number of pi bonds in B is to be determined. And one possible structure is to be drawn.
Concept introduction: Degree of unsaturation is used to determine the total number of rings and pi bonds present in compound by just looking at the molecular formula. It does not specify the total number of rings and total number of pi bonds individually.
Answer to Problem 12.35P
The number of pi bonds and number of rings in B is one and two respectively. The possible structure is given in Figure 2.
Explanation of Solution
For compound B:
Before hydrogenation, the molecular formula is
The maximum number of
The maximum number of
The number of
Substitute the values of maximum number of
The degree of unsaturation is calculated by the formula,
Hence, the degree of unsaturation before hydrogenation is three.
After hydrogenation, the molecular formula is
The maximum number of
The maximum number of
The number
Substitute the values of maximum number of
The degree of unsaturation is calculated by the formula,
Hence, the degree of unsaturation after hydrogenation is two.
The number of pi bonds in A is calculated by the formula,
Substitute the values of degree of unsaturation before hydrogenation and degree of unsaturation after hydrogenation in the above formula.
Hence, the number of pi bonds is one.
Number of rings is calculated by the formula,
Substitute the values of degree of unsaturation and number of pi bonds in the above formula.
Hence, the number of rings is two.
The possible structure for B is,
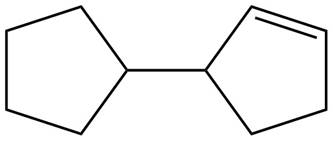
Figure 2
The number of pi bonds and number of rings in B is one and two respectively. The possible structure is given in Figure 2.
(c)
Interpretation: The number of rings and number of pi bonds in B is to be determined. And one possible structure is to be drawn.
Concept introduction: Degree of unsaturation is used to determine the total number of rings and pi bonds present in compound by just looking at the molecular formula. It does not specify the total number of rings and total number of pi bonds individually.
Answer to Problem 12.35P
The number of pi bonds and number of rings in C is four and one respectively. The possible structure is given in Figure 3.
Explanation of Solution
For compound C:
Before hydrogenation, the molecular formula is
The maximum number of
The maximum number of
The number of
Substitute the values of maximum number of
The degree of unsaturation is calculated by the formula,
Hence, the degree of unsaturation before hydrogenation is five.
After hydrogenation, the molecular formula is
The maximum number of
The maximum number of
The number
Substitute the values of maximum number of
The degree of unsaturation is calculated by the formula,
Hence, the degree of unsaturation after hydrogenation is one.
The number of pi bonds in A is calculated by the formula,
Substitute the values of degree of unsaturation before hydrogenation and degree of unsaturation after hydrogenation in the above formula.
Hence, the number of pi bonds is four.
Number of rings is calculated by the formula,
Substitute the values of degree of unsaturation and number of pi bonds in the above formula.
Hence, the number of rings is one.
The possible structure for C is,
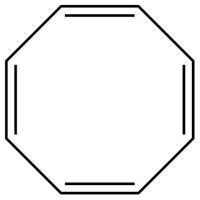
Figure 3
The number of pi bonds and number of rings in C is four and one respectively. The possible structure is given in Figure 3.
Want to see more full solutions like this?
Chapter 12 Solutions
ORGANIC CHEMISTRY
- :0: :0: Select to Add Arrows :0: (CH3)2NH :0: ■ Select to Add Arrows :0: :0: (CH3)2NH ■ Select to Add Arrowsarrow_forwardDraw the product of the following H action sequence. Ignore any inorganic byproducts formed. 1. (CH3CH2)2CuLi, THF 2. CH3Br Q Atoms, Bonds and Rings H Charges ㅁarrow_forwardPlease help me with this the problem is so confusingarrow_forward
- 14 Question (1 point) Disiamylborane adds to a triple bond to give an alkenylborane. Upon oxidation with OH, H2O2, the alkenylborane will form an enol that tautomerizes to an aldehyde. In the first box below, draw the mechanism arrows for the reaction of disiamylborane with the alkyne, and in the last box draw the structure of the aldehyde. 4th attempt Feedback i > 3rd attempt OH, H2O2 i See Periodic Table See Hintarrow_forwardanswer with mechanisms and steps. handwritten please!arrow_forwardHello I need some help with Smartwork. For drawing structure B, I know the correct answer is CH₃B₂, but when I try to type it in, it keeps giving me CH₄BH₃ instead. Do you know how I should write it properly? Should I use a bond or something else?arrow_forward
