
Thermodynamics: An Engineering Approach
8th Edition
ISBN: 9780073398174
Author: Yunus A. Cengel Dr., Michael A. Boles
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1.11, Problem 112RP
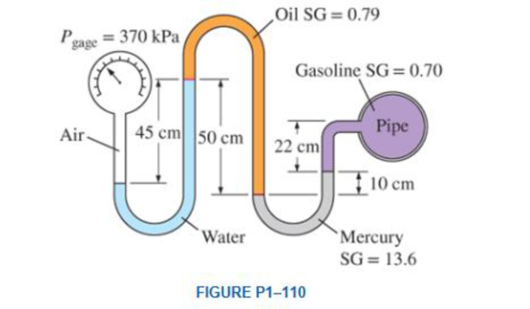
A gasoline line is connected to a pressure gage through a double-U manometer, as shown in Fig. 1–110. If the reading of the pressure gage is 370 kPa, determine the gage pressure of the gasoline line.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
turbomachienery
auto controls
auto controls
Chapter 1 Solutions
Thermodynamics: An Engineering Approach
Ch. 1.11 - What is the difference between the classical and...Ch. 1.11 - The value of the gravitational acceleration g...Ch. 1.11 - One of the most amusing things a person can...Ch. 1.11 - An office worker claims that a cup of cold coffee...Ch. 1.11 - 1–5C What is the difference between kg-mass and...Ch. 1.11 - Explain why the light-year has the dimension of...Ch. 1.11 - What is the net force acting on a car cruising at...Ch. 1.11 - 1–8 At 45° latitude, the gravitational...Ch. 1.11 - What is the weight, in N, of an object with a mass...Ch. 1.11 - A 3-kg plastic tank that has a volume of 0.2 m3 is...
Ch. 1.11 - Prob. 11PCh. 1.11 - Prob. 12PCh. 1.11 - Solve Prob. 113 using appropriate software. Print...Ch. 1.11 - A 4-kW resistance heater in a water heater runs...Ch. 1.11 - A 150-lbm astronaut took his bathroom scale (a...Ch. 1.11 - The gas tank of a car is filled with a nozzle that...Ch. 1.11 - Prob. 17PCh. 1.11 - A large fraction of the thermal energy generated...Ch. 1.11 - Prob. 19PCh. 1.11 - 1–20C A can or soft drink at room temperature is...Ch. 1.11 - What is the difference between intensive and...Ch. 1.11 - Is the number of moles of a substance contained in...Ch. 1.11 - Is the state of the air in an isolated room...Ch. 1.11 - The specific weight of a system is defined as the...Ch. 1.11 - What is a quasi-equilibrium process? What is its...Ch. 1.11 - Define the isothermal, isobaric, and isochoric...Ch. 1.11 - Prob. 27PCh. 1.11 - Prob. 28PCh. 1.11 - 1–29C What is specific gravity? How is it related...Ch. 1.11 - 1–31C What are the ordinary and absolute...Ch. 1.11 - Prob. 32PCh. 1.11 - Prob. 33PCh. 1.11 - Prob. 34PCh. 1.11 - Prob. 35PCh. 1.11 - Prob. 36PCh. 1.11 - Prob. 37PCh. 1.11 - Prob. 38PCh. 1.11 - The temperature of a system drops by 45F during a...Ch. 1.11 - Explain why some people experience nose bleeding...Ch. 1.11 - A health magazine reported that physicians...Ch. 1.11 - Someone claims that the absolute pressure in a...Ch. 1.11 - 1–43C Express Pascal’s law, and give a real-world...Ch. 1.11 - Consider two identical fans, one at sea level and...Ch. 1.11 - A vacuum gage connected to a chambee reads 35 kPa...Ch. 1.11 - Prob. 46PCh. 1.11 - 1–47E The pressure in a water line is 1500 kPa....Ch. 1.11 - 1–48E If the pressure inside a rubber balloon is...Ch. 1.11 - A manometer is used to measure the air pressure in...Ch. 1.11 - 1–50 The water in a tank is pressurized by air,...Ch. 1.11 - 1–51 Determine the atmospheric pressure at a...Ch. 1.11 - A 200-pound man has a total foot imprint area of...Ch. 1.11 - The gage pressure in a liquid at a depth of 3 m is...Ch. 1.11 - The absolute pressure in water at a depth of 9 m...Ch. 1.11 - 1–55E Determine the pressure exerted on the...Ch. 1.11 - 1–56 Consider a 70-kg woman who has a total foot...Ch. 1.11 - Prob. 57PCh. 1.11 - The barometer of a mountain hiker reads 750 mbars...Ch. 1.11 - The basic barometer can be used to measure the...Ch. 1.11 - Prob. 61PCh. 1.11 - A gas is contained in a vertical, frictionless...Ch. 1.11 - Reconsider Prob. 158. Using appropriate software,...Ch. 1.11 - Both a gage and a manometer are attached to a gas...Ch. 1.11 - Reconsider Prob. 161. Using appropriate software,...Ch. 1.11 - A manometer containing oil ( = 850 kg/m3) is...Ch. 1.11 - A mercury manometer ( = 13.600 kg/m3) is connected...Ch. 1.11 - Repeat Prob. 165 for a differential mercury height...Ch. 1.11 - The pressure in a natural gas pipeline is measured...Ch. 1.11 - Repeat Prob. 167E by replacing air with oil with a...Ch. 1.11 - Blood pressure is usually measure by wrapping a...Ch. 1.11 - The maximum blood pressure in the upper arm of a...Ch. 1.11 - Prob. 73PCh. 1.11 - Consider a U-tube whose arms are open to the...Ch. 1.11 - Consider a double-fluid manometer attached to an...Ch. 1.11 - Prob. 76PCh. 1.11 - Prob. 77PCh. 1.11 - Calculate the absolute pressure. P1, of the...Ch. 1.11 - Consider the manometer in Fig. 173. If the...Ch. 1.11 - Consider the manometer in Fig. 173. If the...Ch. 1.11 - Consider the system shown in Fig. 177. If a change...Ch. 1.11 - What is the value of the engineering software...Ch. 1.11 - Determine a positive real root of this equation...Ch. 1.11 - Solve this system of three equations with three...Ch. 1.11 - Solve this system of three equations with three...Ch. 1.11 - The reactive force developed by a jet engine to...Ch. 1.11 - A man goes to a traditional market to buy a steak...Ch. 1.11 - What is the weight of a 1-kg substance in N, kN,...Ch. 1.11 - A hydraulic lift is to be used to lift a 1900-kg...Ch. 1.11 - Prob. 92RPCh. 1.11 - Prob. 93RPCh. 1.11 - Prob. 94RPCh. 1.11 - Prob. 95RPCh. 1.11 - Prob. 96RPCh. 1.11 - It is well known that cold air feels much colder...Ch. 1.11 - Reconsider Prob. 1116E. Using appropriate...Ch. 1.11 - A vertical pistoncylinder device contains a gas at...Ch. 1.11 - An air-conditioning system requires a 35-m-long...Ch. 1.11 - The average body temperature of a person rises by...Ch. 1.11 - Balloons are often filled with helium gas because...Ch. 1.11 - Reconsider Prob. 1101. Using appropriate software,...Ch. 1.11 - Determine the maximum amount of load, in kg, the...Ch. 1.11 - The lower half of a 6-m-high cylindrical container...Ch. 1.11 - A vertical, frictionless pistoncylinder device...Ch. 1.11 - A pressure cooker cooks a lot faster than an...Ch. 1.11 - Prob. 108RPCh. 1.11 - Consider a U-tube whose arms are open to the...Ch. 1.11 - Prob. 110RPCh. 1.11 - A water pipe is connected to a double-U manometer...Ch. 1.11 - A gasoline line is connected to a pressure gage...Ch. 1.11 - Repeat Prob. 1110 for a pressure gage reading of...Ch. 1.11 - The average atmosphere pressure on earth is...Ch. 1.11 - Prob. 115RPCh. 1.11 - Prob. 116RPCh. 1.11 - Consider the flow of air through a wind turbine...Ch. 1.11 - The drag force exerted on a car by air depends on...Ch. 1.11 - An apple loses 3.6 kJ of heat as it cools per C...Ch. 1.11 - Consider a fish swimming 5 m below the free...Ch. 1.11 - The atmospheric pressures at the top and the...Ch. 1.11 - Consider a 2.5-m-deep swimming pool. The pressure...Ch. 1.11 - During a heating process, the temperature of an...Ch. 1.11 - At sea level, the weight of 1 kg mass in SI units...
Additional Engineering Textbook Solutions
Find more solutions based on key concepts
Comprehension Check 7-14
The power absorbed by a resistor can be given by P = I2R, where P is power in units of...
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
How is the hydrodynamic entry length defined for flow in a pipe? Is the entry length longer in laminar or turbu...
Fluid Mechanics: Fundamentals and Applications
Porter’s competitive forces model: The model is used to provide a general view about the firms, the competitors...
Management Information Systems: Managing The Digital Firm (16th Edition)
Why is the study of database technology important?
Database Concepts (8th Edition)
CONCEPT QUESTIONS
15.CQ3 The ball rolls without slipping on the fixed surface as shown. What is the direction ...
Vector Mechanics for Engineers: Statics and Dynamics
What types of coolant are used in vehicles?
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- 1 Pleasearrow_forwardA spring cylinder system measures the pressure. Determine which spring can measure pressure between 0-1 MPa with a large excursion. The plate has a diameter of 20 mm. Also determine the displacement of each 0.1 MPa step.Spring power F=c x fF=Springpower(N)c=Spring constant (N/mm)f=Suspension (mm) How do I come up with right answer?arrow_forwardA lift with a counterweight is attached to the ceiling. The attachment is with 6 stainless and oiled screws. What screw size is required? What tightening torque? - The lift weighs 500 kg and can carry 800 kg. - Counterweight weight 600 kg - Durability class 12.8 = 960 MPa- Safety factor ns=5+-Sr/Fm= 0.29Gr =0.55arrow_forward
- Knowing that a force P of magnitude 750 N is applied to the pedal shown, determine (a) the diameter of the pin at C for which the average shearing stress in the pin is 40 MPa, (b) the corresponding bearing stress in the pedal at C, (c) the corresponding bearing stress in each support bracket at C. 75 mm 300 mm- mm A B P 125 mm 5 mm C Darrow_forwardAssume the B frame differs from the N frame through a 90 degree rotation about the second N base vector. The corresponding DCM description is: 1 2 3 4 5 6 9 # adjust the return matrix values as needed def result(): dcm = [0, 0, 0, 0, 0, 0, 0, 0, 0] return dcmarrow_forwardFind the reaction at A and B The other response I got was not too accurate,I need expert solved answer, don't use Artificial intelligence or screen shot it solvingarrow_forward
- A six cylinder petrol engine has a compression ratio of 5:1. The clearance volume of each cylinder is 110CC. It operates on the four-stroke constant volume cycle and the indicated efficiency ratio referred to air standard efficiency is 0.56. At the speed of 2400 rpm. 44000KJ/kg. Determine the consumes 10kg of fuel per hour. The calorific value of fuel average indicated mean effective pressure.arrow_forwardThe members of a truss are connected to the gusset plate as shown in (Figure 1). The forces are concurrent at point O. Take = 90° and T₁ = 7.5 kN. Part A Determine the magnitude of F for equilibrium. Express your answer to three significant figures and include the appropriate units. F= 7.03 Submit ? kN Previous Answers Request Answer × Incorrect; Try Again; 21 attempts remaining ▾ Part B Determine the magnitude of T2 for equilibrium. Express your answer to three significant figures and include the appropriate units. Figure T₂ = 7.03 C T2 |? KN Submit Previous Answers Request Answer × Incorrect; Try Again; 23 attempts remaining Provide Feedbackarrow_forwardConsider the following acid-base reaction: Fe3+(aq) +3H2O -Fe(OH)3 (s) + 3H* ← A. Using thermodynamics, calculate the equilibrium constant K at 25°C (The AG° of formation of Fe(OH)3(s) is -699 kJ/mol). B. Using the value of K you calculated in part a, if a solution contains 10-4 M Fe3+ and has a pH of 7.5, will Fe(OH)3(s) precipitate? Show all calculations necessary to justify your answer. Note that the reaction as written is for precipitation, not dissolution like Ksp-arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
First Law of Thermodynamics, Basic Introduction - Internal Energy, Heat and Work - Chemistry; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=NyOYW07-L5g;License: Standard youtube license