
(a)
Interpretation:
The product of the reaction of
Concept introduction:
Answer to Problem 11.46AP
The product of the reaction of
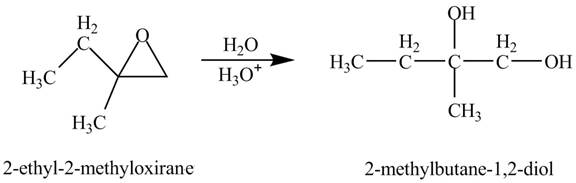
Explanation of Solution
The compound
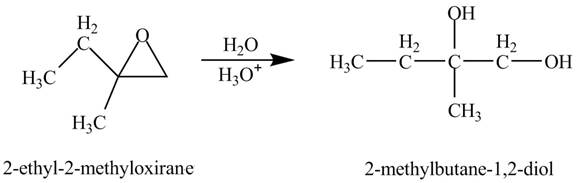
Figure 1
The product of the reaction of
(b)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring opening reactions which are base-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are basic, then the reaction will occur at the less substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
Explanation of Solution
The compound

Figure 2
The product of the reaction of
(c)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are base-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are basic, then the reaction will occur at the less substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
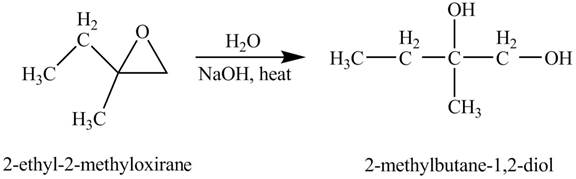
Explanation of Solution
The compound
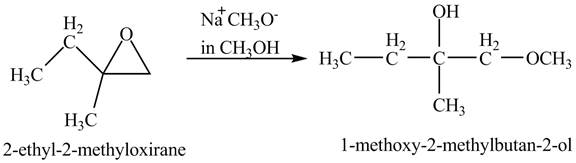
Figure 3
The product of the reaction of
(d)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
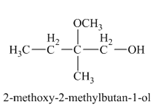
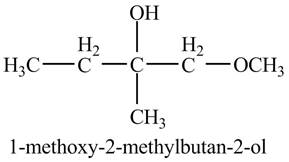
Explanation of Solution
The compound
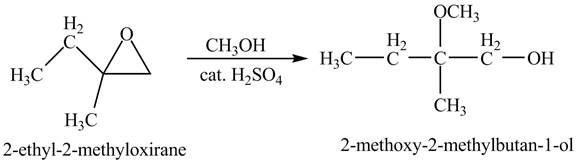
Figure 4
The product of the reaction of
(e)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
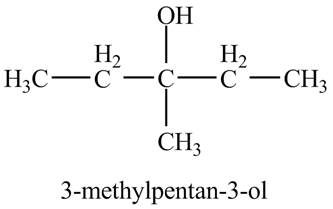
Explanation of Solution
The compound
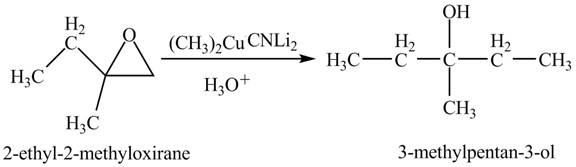
Figure 5
The product of the reaction of
(f)
Interpretation:
The product of the reaction of the product of part (c) with
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of the product of part (c) with
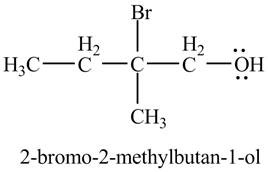
Explanation of Solution
In the presence of
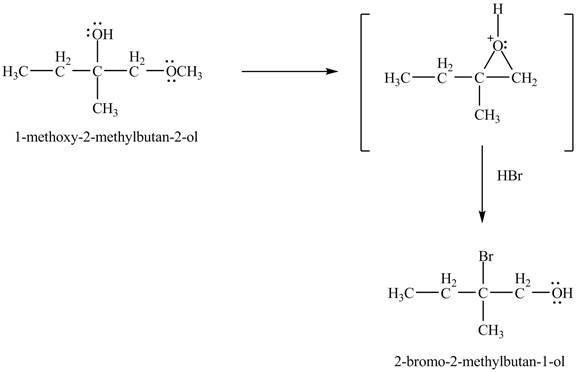
Figure 6
The product of the reaction of the given compound with
(g)
Interpretation:
The product of the reaction of the product of part (d) with
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of the product of part (d) with
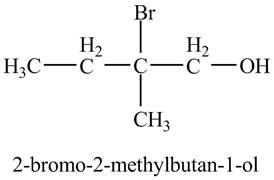
Explanation of Solution
In the presence of
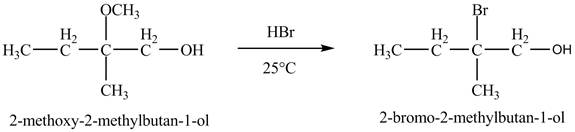
Figure 7
The product of the reaction of the given compound with
(h)
Interpretation:
The product of the reaction of the product of part (c) with
Concept introduction:
The metal hydride reagents are good reducing agents such as
Answer to Problem 11.46AP
The product of the reaction of the product of part (c) with
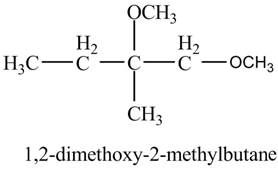
Explanation of Solution
The base
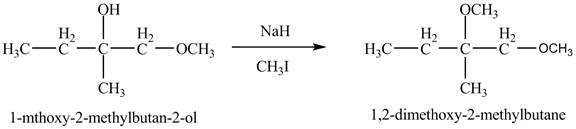
Figure 8
The product of the reaction of the given compound with
(i)
Interpretation:
The product of the reaction of the product of part (d) with
Concept introduction:
The metal hydride reagents are good reducing agents such as
Answer to Problem 11.46AP
The product of the reaction of the product of part (d) with
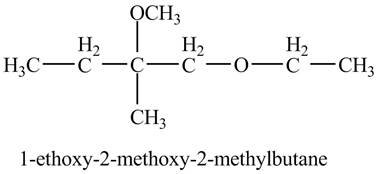
Explanation of Solution
The base
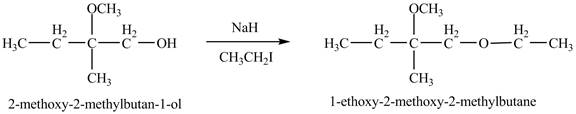
Figure 9
The product of the reaction of the given compound with
(j)
Interpretation:
The product of the reaction of the product of part (a) with periodic acid is to be predicted.
Concept introduction:
The periodic acid acts as a strong oxidizing agent. The periodic acid reacts with a vicinal diol to form two
Answer to Problem 11.46AP
The products of the reaction of the product of part (a) with periodic acid are shown below.
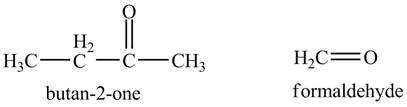
Explanation of Solution
The given compound is vicinal diol. It reacts with periodic acid to form two aldehydes. The carbon-carbon bond between the carbon atoms attached to two adjacent hydroxyl groups gets breaks. The corresponding chemical reaction is shown below.
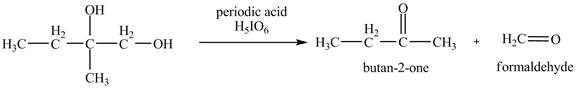
Figure 10
The products of the reaction of the given compound with periodic acid are shown in Figure 10.
(k)
Interpretation:
The product of the reaction of the product of part (f) with
Concept introduction:
Grignard reagents are
Answer to Problem 11.46AP
The product of the reaction of the product of part (f) with
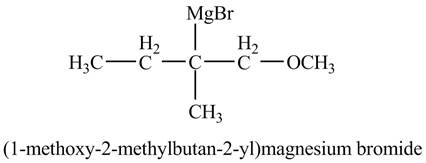
Explanation of Solution
The compound
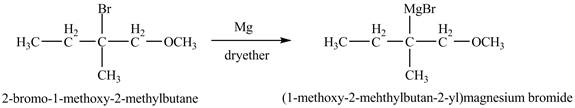
Figure 11
The product of the reaction of the given compound with
(l)
Interpretation:
The product of the reaction of the product of part (k) with ethylene oxide followed by addition of
Concept introduction:
Grignard reagents are organometallic compounds which are prepared using alkyl halides in the presence of magnesium metal in dry ether. These reagents act as strong nucleophiles and bases. Grignard reagents react with carbonyl compounds to form alcohol.
Answer to Problem 11.46AP
The product of the reaction of the product of part (k) with ethylene oxide followed by addition of
Explanation of Solution
Grignard reagent can act as a nucleophile. In the presence of an acid, it can attack the more substituted carbon atom of the epoxy ring. The Grignard reagent reacts with the ethylene oxide followed by protonolysis to form alcohol. The corresponding chemical reaction is shown below.
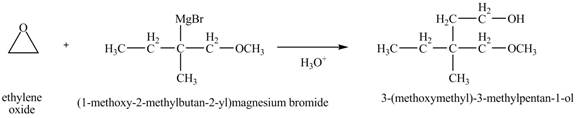
Figure 12
The product of the reaction of the given compound with ethylene oxide followed by addition of
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry Study Guide and Solutions
- 7. The three sets of replicate results below were accumulated for the analysis of the same sample. Pool these data to obtain the most efficient estimate of the mean analyte content and the standard deviation. Lead content/ppm: Set 1 Set 2 Set 3 1. 9.76 9.87 9.85 2. 9.42 9.64 9.91 3. 9.53 9.71 9.42 9.81 9.49arrow_forwardDraw the Zaitsev product famed when 2,3-dimethylpentan-3-of undergoes an El dehydration. CH₂ E1 OH H₁PO₁ Select Draw Templates More QQQ +H₂Oarrow_forwardComplete the clean-pushing mechanism for the given ether synthesia from propanol in concentrated sulfurica140°C by adding any mining aloms, bands, charges, nonbonding electron pairs, and curved arrows. Draw hydrogen bonded to cayan, when applicable. ore 11,0 HPC Step 1: Draw curved arrows Step 2: Complete the intend carved Q2Q 56 QQQ Step 3: Complete the intermediate and add curved Step 4: Modify the structures to draw the QQQ QQQarrow_forward
- 6. In an experiment the following replicate set of volume measurements (cm3) was recorded: (25.35, 25.80, 25.28, 25.50, 25.45, 25.43) A. Calculate the mean of the raw data. B. Using the rejection quotient (Q-test) reject any questionable results. C. Recalculate the mean and compare it with the value obtained in 2(a).arrow_forwardA student proposes the transformation below in one step of an organic synthesis. There may be one or more reactants missing from the left-hand side, but there are no products missing from the right-hand side. There may also be catalysts, small inorganic reagents, and other important reaction conditions missing from the arrow. • Is the student's transformation possible? If not, check the box under the drawing area. • If the student's transformation is possible, then complete the reaction by adding any missing reactants to the left-hand side, and adding required catalysts, inorganic reagents, or other important reaction conditions above and below the arrow. • You do not need to balance the reaction, but be sure every important organic reactant or product is shown. + T G OH де OH This transformation can't be done in one step.arrow_forwardMacmillan Leaming Draw the major organic product of the reaction. 1. CH3CH2MgBr 2. H+ - G Select Draw Templates More H о QQarrow_forward
- Draw the condensed structure of 3-hydroxy-2-butanone. Click anywhere to draw the first atom of your structure.arrow_forwardGive the expected major product of reaction of 2,2-dimethylcyclopropane with each of the following reagents. 2. Reaction with dilute H₂SO, in methanol. Select Draw Templates More CHC Erase QQQ c. Reaction with dilute aqueous HBr. Select Drew Templates More Era c QQQ b. Reaction with NaOCH, in methanol. Select Draw Templates More d. Reaction with concentrated HBr. Select Draw Templates More En a QQQ e. Reaction with CH, Mg1, then H*, H₂O 1. Reaction with CH,Li, then H', H₂Oarrow_forwardWrite the systematic name of each organic molecule: structure O OH OH name X ☐arrow_forward
- Macmillan Learning One of the molecules shown can be made using the Williamson ether synthesis. Identify the ether and draw the starting materials. А со C Strategy: Review the reagents, mechanism and steps of the Williamson ether synthesis. Determine which of the molecules can be made using the steps. Then analyze the two possible disconnection strategies and deduce the starting materials. Identify the superior route. Step 6: Put it all together. Complete the two-step synthesis by selecting the reagents and starting materials. C 1. 2. Answer Bank NaH NaOH NaOCH, снен, сен, он Сиси, Сне (СН), СОН (Сн, Свarrow_forwardWrite the systematic name of each organic molecule: structure CH3 O CH3-CH-CH-C-CH3 OH HV. CH3-C-CH-CH2-CH3 OH CH3 O HO—CH, CH–CH—C CH3 OH 오-오 name X G ☐arrow_forwardHI Organic Functional Groups Predicting the reactants or products of esterification What is the missing reactant in this organic reaction? HO OH H +回 + H₂O 60013 Naomi V Specifically, in the drawing area below draw the skeletal ("line") structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. No answer Click and drag to start drawing a structure. Explanation Check 1 2 #3 $ 4 2025 % ala5 'a :☐ G & 67 8 Ar K enter Accessible 9 Q W E R TY U 1 tab , S H J Karrow_forward
