
Concept explainers
Draw the products formed when
a.
b.
c.
d.
e.
f.
g.
h.
![Chapter 11, Problem 11.36P, 11.33 Draw the products formed when is treated with each reagent.
[2] ;](http://dev-ingestion-image-output.s3-website-us-east-1.amazonaws.com/9780078021558/Chapter-11/images/21558-11-11.33p-question-digital_image001.jpg) ;
;
(a)
Interpretation: The product that is formed by the reaction of
Concept introduction: The addition of an electrophile to an alkyne is followed by Markovnikoff’s rule and anti-stereoselectivity.
The addition of a halogen to an alkyne chain leads to the formation of corresponding alkene or alkane. The reaction which includes the addition of bromine atoms to the alkyne chain is known as bromination. Bromination can be done by using the reagents like
Answer to Problem 11.36P
The product that is formed by the reaction of
Explanation of Solution
The reaction of
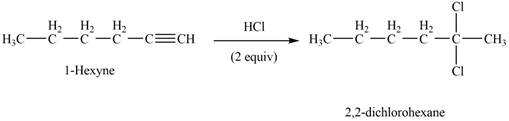
Figure 1
The reaction of
The product that is formed by the reaction of
(b)
Interpretation: The product that is formed by the reaction of
Concept introduction: The addition of an electrophile to an alkyne is followed by Markovnikoff’s rule and anti-stereoselectivity.
The addition of a halogen to an alkyne chain leads to the formation of corresponding alkene or alkane. The reaction which includes the addition of bromine atoms to the alkyne chain is known as bromination. Bromination can be done by using the reagents like
Answer to Problem 11.36P
The product that is formed by the reaction of
Explanation of Solution
The reaction of
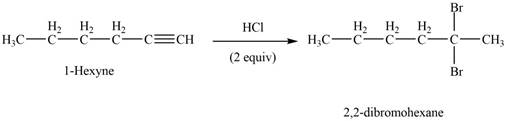
Figure 2
The reaction of
The product that is formed by the reaction of
(c)
Interpretation: The product that is formed by the reaction of
Concept introduction: The addition of an electrophile to an alkyne is followed by Markovnikoff’s rule and anti-stereoselectivity.
The addition of a halogen to an alkyne chain leads to the formation of corresponding alkene or alkane. The reaction which includes the addition of bromine atoms to the alkyne chain is known as bromination. Bromination can be done by using the reagents like
Answer to Problem 11.36P
The product that is formed by the reaction of
Explanation of Solution
The reaction of
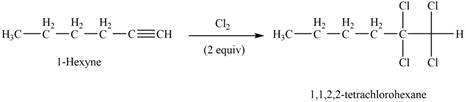
Figure 3
The reaction of
The product that is formed by the reaction of
(d)
Interpretation: The product that is formed by the reaction of
Concept introduction: A terminal alkyne reacts with
The chemical equilibrium that exists between a keto form of a compound and an enol form of the same compound is known as keto-enol tautomerism. Tautomers refer to these keto and enol forms.
Answer to Problem 11.36P
The product that is formed by the reaction of
Explanation of Solution
The reaction of
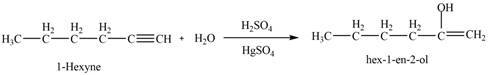
Figure 4
The reaction of
Then, the enol form,
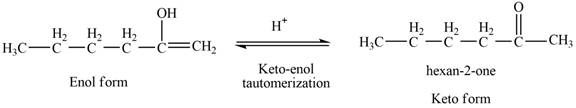
Figure 5
Thus, the keto-enol tautomerization of
The product that is formed by the reaction of
(e)
Interpretation: The product that is formed by the reaction of
Concept introduction: A stepwise procedure of transforming an alkyne into a carbonyl group is known hydroboration-oxidation reaction. In a hydroboration-oxidation reaction, a terminal alkyne reacts with
Answer to Problem 11.36P
The product that is formed by the reaction of
Explanation of Solution
The reaction of
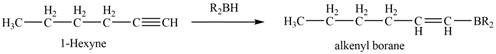
Figure 6
The reaction of
Then, the reaction of alkenylborane with
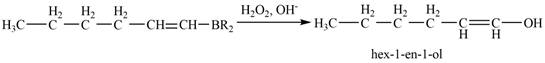
Figure 7
Thus, the reaction of alkenylborane with
In the last step,
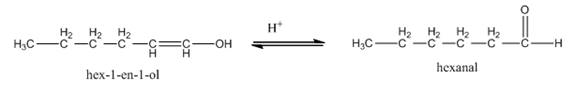
Figure 8
Thus, the tautomerization of
The product that is formed by the reaction of
(f)
Interpretation: The product that is formed by the reaction of
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Answer to Problem 11.36P
The product that is formed by the reaction of
Explanation of Solution
The reaction of
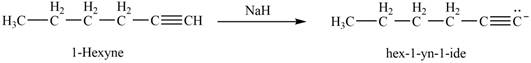
Figure 9
The reaction of
(a) The product that is formed by the reaction of
(g)
Interpretation: The product that is formed by the reaction of
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Answer to Problem 11.36P
The product that is formed by the reaction of
Explanation of Solution
The reaction of
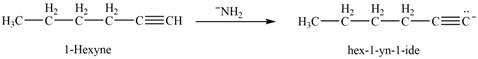
Figure 10
The reaction of
Then, the reaction of
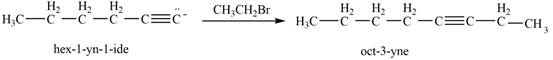
Figure 11
Thus, in the above reaction,
The product that is formed by the reaction of
(h)
Interpretation: The product that is formed by the reaction of ![]() ;
;
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Answer to Problem 11.36P
The product that is formed by the reaction of ![]() ;
;
Explanation of Solution
The reaction of
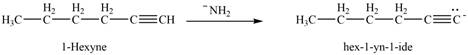
Figure 12
The reaction of
Then, the reaction of
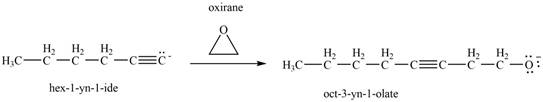
Figure 13
Thus, in the above reaction,
Then, the reaction of
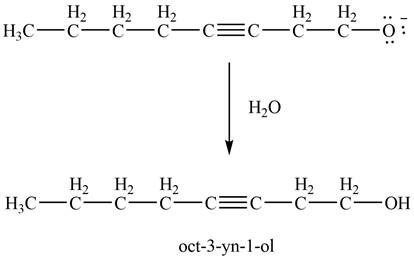
Figure 14
Thus, in the above reaction,
The product that is formed by the reaction of ![]() ;
;
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Biology: Concepts and Investigations
Biology: Life on Earth with Physiology (11th Edition)
Physical Universe
Microbiology with Diseases by Body System (5th Edition)
Loose Leaf For Integrated Principles Of Zoology
- Indicate one aspect that benefits and another that makes it difficult to use the hydroquinone electrode to measure pH.arrow_forwardAt an electrified interface according to the Gouy-Chapman model, what types of interactions do NOT occur between the ions and the solvent according to this theory?arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. Hint: In this case you must choose the best answer to demonstrate the stereochemistry of H2 addition. 1.03 2. (CH3)2S BIZ CH₂OH 2. DMS KMnO4, NaOH ΖΗ Pd or Pt (catalyst) HBr 20 1 HBr ROOR (peroxide) HO H-SO HC 12 11 10 BH, THE 2. H2O2, NaOH Brz cold HI 19 18 17 16 MCPBA 15 14 13 A Br H₂O BH3⚫THF Brz EtOH Pd or Ni (catalyst) D₂ (deuterium) 1. Os04 2. H2O2 CH3CO3H (peroxyacid) 1. MCPBA 2. H₂O* H B + H H H "H C H H Darrow_forward
- Explain how Beer’s Law can be used to determine the concentration in a selected food sample. Provide examples.arrow_forwardExplain the importance of having a sampling plan with respect to food analysis. Explain the importance of having a sampling plan with respect to food analysis. Provide examples.arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. cold KMnO4, NaOH 2. DMS 1. 03 CH3OH Br2 1. 03 2. (CH3)2S H₂ Pd or Pt (catalyst) HBr 18 19 20 1 HBr ROOR (peroxide) H₂O H₂SO4 HCI HI 17 16 6 15 MCPBA 1. BH3 THF 2. H₂O2, NaOH 1. OsO4 2. H₂O₂ 110 CH3CO₂H (peroxyacid) 1. MCPBA 2. H₂O* Br2 H₂O BH3 THF B12 EtOH Pd or Ni (catalyst) D₂ (deuterium) Bra A B C D H OH H OH OH H OH α α α OH H OH OH фон d H "Harrow_forward
- Briefly indicate the models that describe the structure of the interface: Helmholtz-Perrin, Gouy-Chapman, Stern and Grahame models.arrow_forwardElectrochemistry. Briefly describe the Gibbs model and the Gibbs absorption equation.arrow_forwardThermodynamic analysis of electrified interfaces.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





