
Concept explainers
(a)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(b)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
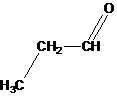
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(c)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(d)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(e)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(f)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
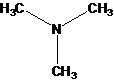
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
Trending nowThis is a popular solution!

Chapter 10 Solutions
Introduction to General, Organic and Biochemistry
- Show the mechanism steps to obtain the lowerenergy intermediate: *see imagearrow_forwardSoap is made by the previous reaction *see image. The main difference between one soap and another soap isthe length (number of carbons) of the carboxylic acid. However, if a soap irritates your skin, they mostlikely used too much lye.Detergents have the same chemical structure as soaps except for the functional group. Detergentshave sulfate (R-SO4H) and phosphate (R-PO4H2) functional groups. Draw the above carboxylic acidcarbon chain but as the two variants of detergents. *see imagearrow_forwardWhat are the reactions or reagents used? *see imagearrow_forward
- The two pKa values of oxalic acid are 1.25 and 3.81. Why are they not the same value? Show the protontransfer as part of your explanation. *see imagearrow_forwardасть Identify all the bonds that gauche interact with C-OMe in the most stable conformation of the above compound.arrow_forwardPredict the reactants used in the formation of the following compounds using Acid-Catalyzed dehydration reactionarrow_forward
- Can I please get help with this?arrow_forward.. Give the major organic product(s) for each of the following reactions or sequences of reactions. Show ll relevant stereochemistry [3 ONLY]. A H Br 1. NaCN 2 NaOH, H₂O, heat 3. H3O+ B. CH₂COOH 19000 1. LiAlH4 THF, heat 2 H₂O* C. CH Br 1. NaCN, acetone 2 H3O+, heat D. Br 1. Mg. ether 3. H₂O+ 2 CO₂ E. CN 1. (CH) CHMgBr, ether 2 H₂O+arrow_forwardAssign this COSY spectrumarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

