
Organic Chemistry-Package(Custom)
4th Edition
ISBN: 9781259141089
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10, Problem 10.21P
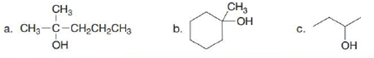
What two
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How to determine the product with mechanism showed
how to determine the product for this reaction in the footo
b.
H3C
Br
-CEN
H3C
Chapter 10 Solutions
Organic Chemistry-Package(Custom)
Ch. 10 - Prob. 10.1PCh. 10 - Problem 10.2 How many degrees of unsaturation are...Ch. 10 - Give an example of a compound with molecular...Ch. 10 - Give the IUPAC name for each alkene.Ch. 10 - Give the IUPAC name for each polyfunctional...Ch. 10 - Prob. 10.6PCh. 10 - Prob. 10.7PCh. 10 - Prob. 10.8PCh. 10 - Prob. 10.9PCh. 10 - Problem 10.10 Rank the following isomers in order...
Ch. 10 - Linolenic acidTable 10.2 and stearidonic acid are...Ch. 10 - Prob. 10.12PCh. 10 - Prob. 10.13PCh. 10 - Prob. 10.14PCh. 10 - Prob. 10.15PCh. 10 - Prob. 10.16PCh. 10 - Prob. 10.17PCh. 10 - Prob. 10.18PCh. 10 - Prob. 10.19PCh. 10 - Which compounds A-D in Figure 10.12 are formed by...Ch. 10 - What two alkenes give rise to each alcohol as the...Ch. 10 - Prob. 10.22PCh. 10 - Problem 10.23 Draw the products of each reaction,...Ch. 10 - Problem 10.24 Draw all stereoisomers formed in...Ch. 10 - Prob. 10.25PCh. 10 - Prob. 10.26PCh. 10 - Borane is sold for laboratory use as a complex...Ch. 10 - What alkylborane is formed from hydroboration of...Ch. 10 - Draw the products formed when each alkene is...Ch. 10 - What alkene can be used to prepare each alcohol as...Ch. 10 - Prob. 10.31PCh. 10 - Draw the products of each reaction using the two...Ch. 10 - Problem 10.31 Devise a synthesis of each compound...Ch. 10 - Give the IUPAC name for each compound. a.b.Ch. 10 - a Label the carbon-carbon double bond in A as E or...Ch. 10 - Prob. 10.36PCh. 10 - Calculate the number of degrees of unsaturation f...Ch. 10 - Prob. 10.38PCh. 10 - The fertility drug clomiphene trade name Clomid is...Ch. 10 - Give the IUPAC name for each compound. a....Ch. 10 - Give the structure corresponding to each name. a....Ch. 10 - 10.40 (a) Draw all possible stereoisomers of, and...Ch. 10 - Prob. 10.43PCh. 10 - 10.42 Now that you have learned how to name...Ch. 10 - Prob. 10.45PCh. 10 - Prob. 10.46PCh. 10 - Prob. 10.47PCh. 10 - Prob. 10.48PCh. 10 - By using the bond dissociation energies in...Ch. 10 - Prob. 10.50PCh. 10 - Repeat Problem 10.50 with CH32C=CH2 as the...Ch. 10 - What alkene can be used to prepare each alkyl...Ch. 10 - Prob. 10.53PCh. 10 - Draw the constitutional isomer formed in each...Ch. 10 - Prob. 10.55PCh. 10 - Draw all stereoisomers formed in each reaction....Ch. 10 - Prob. 10.57PCh. 10 - Prob. 10.58PCh. 10 - Prob. 10.59PCh. 10 - Draw a stepwise mechanism for the following...Ch. 10 - Draw a stepwise mechanism for each reaction. a.b.Ch. 10 - Draw a stepwise mechanism that shows how all three...Ch. 10 - Less stable alkenes can be isomerized to more...Ch. 10 - Prob. 10.64PCh. 10 - Prob. 10.65PCh. 10 - Bromoetherification, the addition of the elements...Ch. 10 - Devise a synthesis of each product from the given...Ch. 10 - Prob. 10.68PCh. 10 - Prob. 10.69PCh. 10 - Prob. 10.70PCh. 10 - 10.66 Explain why A is a stable compound but B is...Ch. 10 - Prob. 10.72PCh. 10 - Prob. 10.73PCh. 10 - 10.69 Lactones, cyclic esters such as compound A,...Ch. 10 - 10.70 Draw a stepwise mechanism for the following...Ch. 10 - 10.71 Like other electrophiles, carbocations add...Ch. 10 - 10.72 Draw a stepwise mechanism for the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY