
a)
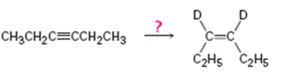
Interpretation:
How to carry out the reaction given which yields deuterium incorporated alkene as the product is to be shown.
Concept introduction:
Deuterium incorporated
To show:
How to carry out the reaction given which yields deuterium incorporated alkene as the product.
b)
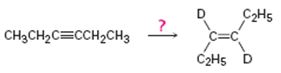
Interpretation:
How to carry out the reaction given which yields deuterium incorporated alkene as the product is to be shown.
Concept introduction:
Deuterium incorporated alkenes can be prepared from alkynes by reduction in the presence of catalysts. Use of deuterium in the presence of Lindlar catalyst yields cis alkenes with the two deuterium atoms arranged on the same side of the double bond while reduction with Li in liquid deuterated ammonia yields trans alkenes with the two deuterium atoms arranged on the opposite sides of the double bond.
To show:
How to carry out the reaction given which yields deuterium incorporated alkene as the product.
c)
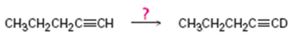
Interpretation:
How to carry out the reaction given which yields deuterium incorporated alkyne as the product is to be shown.
Concept introduction:
Deuterium incorporated alkynes can be prepared first by converting them in to alkynides by treating with NaNH2 in NH3 and then treating the alkynide obtained with D3O+.
To show:
How to carry out the reaction given which yields deuterium incorporated alkyne as the product.
d)
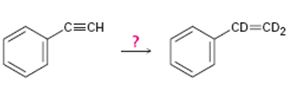
Interpretation:
How to carry out the reaction given which yields deuterium incorporated alkene as the product is to be shown.
Concept introduction:
Deuterium incorporated alkynes can be prepared first by converting them in to alkynides by treating with NaNH2 in NH3 and then treating the alkynide obtained with D3O+. The alkyne thus obtained when treated with deuterium in the presence of Lindlar catalyst yield an alkene with deuterium atom on both carbons.
To show:
How to carry out the reaction given which yields deuterium incorporated alkene as the product.
Trending nowThis is a popular solution!

Chapter 9 Solutions
EBK ORGANIC CHEMISTRY
- When anisole is treated with excess bromine, the reaction gives a product which shows two singlets in 1H NMR. Draw the product.arrow_forward(ii) Draw a reasonable mechanism for the following reaction: CI NaOH heat OH (hint: SNAr Reaction) :arrow_forwardDraw the major product in each of the following reaction:arrow_forward
- Draw the mechanism for the following Friedel-Craft reaction. AlBr3 Brarrow_forward(a) Draw the structures of A and B in the following reaction. (i) NaNH2, NH3(1) A + B (ii) H3O+arrow_forwardFor the reaction 2 N2O5(g) → 4 NO2(g) + O2(g), the following mechanism has been proposed: N2O5 →> NO₂+ NO3_(K1) NO2 + NO3 → N2O5 (k-1) NO2 + NO3 → → NO2 + O2 + NO (K2) NO + N2O5- NO2 + NO2 + NO2 (K3) d[N₂O5] __2k‚k₂[N2O5] Indicate whether the following rate expression is acceptable: dt k₁₁+ k₂arrow_forward
- Consider the following decomposition reaction of N2O5(g): For the reaction 2 N2O5(g) → 4 NO2(g) + O2(g), the following mechanism has been proposed: N2O5 → NO2 + NO3 (K1) NO2 + NO3 → N2O5 (k-1) NO2 + NO3 → NO2 + O2 + NO (K2) NO + N2O5 → NO2 + NO2 + NO2 (K3) Indicate whether the following rate expression is acceptable: d[N2O5] = -k₁[N₂O₂] + K¸₁[NO₂][NO3] - K¸[NO₂]³ dtarrow_forwardIn a reaction of A + B to give C, another compound other than A, B or C may appear in the kinetic equation.arrow_forwardFor the reaction 2 N2O5(g) → 4 NO2(g) + O2(g), the following mechanism has been proposed: N2O5 →> NO₂+ NO3_(K1) NO2 + NO3 → N2O5 (k-1) NO2 + NO3 → → NO2 + O2 + NO (K2) NO + N2O5- NO2 + NO2 + NO2 (K3) d[N₂O5] __2k‚k₂[N2O5] Indicate whether the following rate expression is acceptable: dt k₁₁+ k₂arrow_forward
- Given the reaction R + Q → P, indicate the rate law with respect to R, with respect to P and with respect to P.arrow_forwardSteps and explanations. Also provide, if possible, ways to adress this kind of problems in general.arrow_forwardk₁ Given the reaction A B, indicate k-1 d[A] (A). the rate law with respect to A: (B). the rate law with respect to B: d[B] dt dtarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


