
EBK ORGANIC CHEMISTRY
8th Edition
ISBN: 8220102744127
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 98P
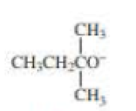
When 2-bromo-2,3-dimethylbutane reacts with a strong base, two
- a. Which of the bases (A, B, C, or D) would form the highest percentage of the highest percentage of the 1-alkene?
- b. Which would give the highest percentage of the 2-alkene?
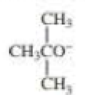
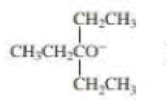
- A. CH3CH2O–
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When 2-bromo-2,3-dimethylbutane reacts with a strong base, two alkenes (2,3-dimethyl-1-butene and 2,3-dimethyl-2-butene) are formed.
a. Which of the bases (A, B, C, or D) would form the highest percentage of the 1-alkene?
b. Which would give the highest percentage of the 2-alkene?
F.
When 3-bromo-2,4-dimethylpentane is treated with sodium hydroxide, only one alkene is formed.
A.
B.
How many different alkenes will be produced when the following substrate is treated with a strong base?
2-Chloro-2-methylpentane
Number of different alkenes:
How many different alkenes will be produced when the following substrate is treated with a strong base?
3-Chloro-3-methylhexane
Number of different alkenes:
Draw the product.
Draw Your Solution
C.
How many different alkenes will be produced when the following substrate is treated with a strong base?
2-Chloropentane
Number of different alkenes:
eTextbook and Media
Explain why this reaction has only one regiochemical outcome.
O There are only two beta protons. Abstraction of either proton lead to the same product.
O There are four beta protons. Abstraction of any proton leads to the same product.
There are two beta protons, but only one is anti to the leaving group.
There is only one beta proton so abstraction leads to one product.…
7) You want to synthesize 3-methyl-2-pentene from 2-chloro-3-methylpentane. Which reagent
would you use?
a. HCI, heat
b. NH:(aq), 25°C
c. CH:CO2NA, CH:CO2H, heat
d. CH3CH2ONA, CH3CH2OH, heat
e. CН:CH2ОН, heat
Chapter 9 Solutions
EBK ORGANIC CHEMISTRY
Ch. 9.1 - Prob. 3PCh. 9.1 - Does increasing the energy barrier for an SN2...Ch. 9.1 - Rank the following alkyl bromides from most...Ch. 9.1 - Prob. 8PCh. 9.2 - Prob. 9PCh. 9.2 - Prob. 10PCh. 9.2 - Prob. 11PCh. 9.2 - Prob. 12PCh. 9.2 - Which substitution reaction lakes place more...Ch. 9.2 - Prob. 15P
Ch. 9.2 - Prob. 17PCh. 9.3 - Prob. 18PCh. 9.4 - Prob. 19PCh. 9.5 - Draw the configuration(s) of the substitution...Ch. 9.5 - Which of the following reactions take place more...Ch. 9.7 - Prob. 22PCh. 9.7 - Prob. 23PCh. 9.7 - Prob. 24PCh. 9.7 - Prob. 25PCh. 9.8 - Four alkenes are formed from the E1 reaction of...Ch. 9.8 - If 2-fluoropentane could undergo an E1 reaction,...Ch. 9.8 - Prob. 28PCh. 9.8 - Propose a mechanism for the following reaction:Ch. 9.9 - Prob. 30PCh. 9.9 - What percentage of the reaction described in...Ch. 9.10 - Prob. 33PCh. 9.10 - Prob. 35PCh. 9.11 - Why do cis-1-bromo-2-ethylcyclohexane and...Ch. 9.11 - Which isomer reacts more rapidly in an E2...Ch. 9.11 - Prob. 38PCh. 9.12 - Prob. 39PCh. 9.12 - Prob. 40PCh. 9.12 - Prob. 41PCh. 9.12 - Explain why only a substitution product and no...Ch. 9.12 - a. Explain why 1-bromo-2,2-dimethylpropane has...Ch. 9.13 - Prob. 44PCh. 9.13 - Prob. 45PCh. 9.13 - What products will be obtained from the El...Ch. 9.13 - Prob. 47PCh. 9.13 - Prob. 48PCh. 9.13 - Prob. 49PCh. 9.13 - Why is a cumulated diene not formed in the...Ch. 9.13 - What product is obtained when the following...Ch. 9.13 - What products are formed from the following...Ch. 9.14 - Prob. 54PCh. 9.14 - Prob. 55PCh. 9.14 - Prob. 56PCh. 9.14 - Prob. 58PCh. 9.14 - Under which of the following reaction conditions...Ch. 9.15 - A small amount of another organic product is...Ch. 9.15 - What is the best way to prepare the following...Ch. 9.15 - Prob. 62PCh. 9.15 - What products (including stereoisomers, if...Ch. 9.16 - After a proton is removed from the OH group, which...Ch. 9.16 - Prob. 65PCh. 9.17 - Prob. 66PCh. 9 - Prob. 1PCh. 9 - Methoxychlor is an insecticide that was intended...Ch. 9 - Prob. 67PCh. 9 - Prob. 68PCh. 9 - Prob. 69PCh. 9 - Prob. 70PCh. 9 - Prob. 71PCh. 9 - Prob. 72PCh. 9 - Starting with cyclohexene, how can the following...Ch. 9 - Prob. 74PCh. 9 - The pKa of acetic acid in water is 4.76. What...Ch. 9 - Prob. 76PCh. 9 - Prob. 77PCh. 9 - Prob. 78PCh. 9 - Prob. 79PCh. 9 - Prob. 80PCh. 9 - Prob. 81PCh. 9 - Prob. 82PCh. 9 - Draw the major product obtained when each of the...Ch. 9 - Prob. 84PCh. 9 - a. Indicate how each of the following factors...Ch. 9 - Prob. 86PCh. 9 - A chemist wanted to synthesize the...Ch. 9 - Prob. 88PCh. 9 - Prob. 89PCh. 9 - Prob. 90PCh. 9 - Prob. 91PCh. 9 - Starting with an alkyl halide, how could the...Ch. 9 - Indicate which species in each pair gives a higher...Ch. 9 - Prob. 94PCh. 9 - Rank the following from most reactive to least...Ch. 9 - For each of the following alkyl halides, indicate...Ch. 9 - Prob. 97PCh. 9 - When 2-bromo-2,3-dimethylbutane reacts with a...Ch. 9 - Prob. 100PCh. 9 - When the following compound undergoes solvolysis...Ch. 9 - cis-1-Bromo-4-tert-butylcyclohexane and...Ch. 9 - Draw the substitution and elimination products.Ch. 9 - tert-Butyl chloride undergoes solvolysis in both...Ch. 9 - Prob. 105PCh. 9 - Prob. 106PCh. 9 - In which solventethanol or diethyl etherwould the...Ch. 9 - Prob. 108PCh. 9 - Two bromoethers are obtained from the reaction of...Ch. 9 - Prob. 110PCh. 9 - Prob. 111PCh. 9 - Prob. 112PCh. 9 - Which of the following hexachlorocyclohexanes is...Ch. 9 - Explain why the rate of the reaction of...Ch. 9 - Prob. 115PCh. 9 - Two elimination products are obtained from the...Ch. 9 - Draw the structures or the product of the obtained...Ch. 9 - How could you prepare the following compounds from...Ch. 9 - cis-4-Bromocyclohexanol and...Ch. 9 - Prob. 120PCh. 9 - Propose a mechanism for the following reaction:Ch. 9 - Prob. 122PCh. 9 - Prob. 123PCh. 9 - Prob. 124PCh. 9 - Prob. 125PCh. 9 - Predict the product for the following reaction and...Ch. 9 - Prob. 127PCh. 9 - Prob. 128PCh. 9 - When equivalent amounts of methyl bromide nod...Ch. 9 - Prob. 130PCh. 9 - The reaction of chloromethane with hydroxide ion...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What stereoisomers are formed in the following reactions? Which stereoisomer is the major product? a. the acid-catalyzed dehydration of 1-pentanol to 2-pentene b. the acid-catalyzed dehydration of 3,4-dimethyl-3-hexanol to 3,4-dimethyl-3-hexenearrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forward2. Name each alkyne. a. CH3CH₂CH₂C=CH b. CH3CH₂CH₂C=CCH3 3. Predict the product of the following reactions. a. CH = CCH3 + HCI b. CH CCH₂CH3 + O2arrow_forward
- Pick the reactant or solvent in each part that gives the faster elimination reaction.a. reaction of -OH with 1-chloro-1-methylcyclohexane or 1-chloro-3-methylcyclohexaneb. reaction of H2O with CH3CH(Cl)CH2CH3 or (CH3)2C(Cl)CH2CH3c. reaction of (CH3)3CCl with -OH in H2O or DMSOarrow_forwardSelect the best reagent expected to convert 3-heptyne to cis-3-heptene. A. NaNH2, NH3 B. Na, NH3 C. H2, Lindlar’s catalyst D. Both A and C E. Both B and Carrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent.a. HClb. CH3COClc. (CH3CO)2Od. excess CH3Ie. (CH3)2C = Of. CH3COCl, AlCl3g. CH3CO2Hh. NaNO2, HCli. Part (b), then CH3COCl, AlCl3j. CH3CHO, NaBH3CNarrow_forward
- 37. What will be the major product of the reaction of 1 mol of propyne with each of the following reagents? a. HBr (1 mol) b. HBr (2 mol) c. Br2 (1 mol)/CH,Cl2 d. Br2 (2 mol)/CH2C12 e. aqueous H2SO4, HgSO4 f. borane in THF followed by H,O2/HO¯/H2O g. excess H2, Pd/C h. H,/Lindlar catalyst i. sodium in liquid ammonia at -78 °C j. sodium amide k. product of part j followed by 1-chloropropanearrow_forwardDraw the organic products formed when cyclopentene is treated with each reagent. With some reagents, no reaction occurs. a.H2 + Pd-C b.H2 + Lindlar catalyst c.Na, NH3 d.CH3CO3H e.[1] CH3CO3H; [2] H2O, HO− f.[1]OsO4 + NMO; [2] NaHSO3, H2O g.KMnO4, H2O, HO− h.[1] LiAlH4; [2] H2O i. [1] O3; [2] CH3SCH3 j.(CH3)3COOH, Ti[OCH(CH3)2]4, (−)-DET k.mCPBA l.Product in (k); then [1] LiAlH4; [2] H2Oarrow_forwardDraw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to give each product.arrow_forward
- 76% 4. Classify each structure below as a nucleophile or electrophile (or both) and briefly explain your choice. a. N31- (the azide ion) C. phenol b. H3O+ (the hydronium ion)arrow_forwardBefore you answer this question, please read the following important principle: If a carbocation is formed in the slow step of a reaction, the reactivity of the reactant will be greater if that carbocation is more stable. The question: Which of the following alkenes reacts fastest with HCI? I HỌC=CHCH,CH,CH, IV CH₂ CH₂CH₂ Select one: OA. I O B. II O C. IV O D. III H H II CH3 H₂C-C-CH=CH₂ H III H₂C. H H CH₂CHarrow_forwardDraw the product or products that will be obtained from the reaction of cis-2-butene and trans-2-butene with each of the following reagents. If a product can exist as stereoisomers, show which stereoisomers are formed. 1. HCl. 4.Br2 in CH2Cl2 7. H2O + H2SO4 2. BH3/THF, followed by HO-, H2O2, H2O 5. Br2 + H2O 8. CH3OH + H2SO4 3. a peroxyacid 6. H2 + Pd/Carrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY