
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9, Problem 9.6P
Rank the following compounds in order of increasing boiling point.
a.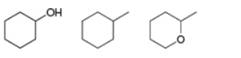
b.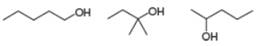
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the major products of this organic reaction:
1. LDA (-78°C)
?
2. Br
Some notes:
• Draw only the major product, or products. You can draw them in any arrangement you like.
.
• Be sure to use wedge and dash bonds where necessary, for example to distinguish between major products that are enantiomers.
• If there are no products, just check the box under the drawing area.
No reaction.
Click and drag to start drawing a structure.
X
Please draw the structures
Draw the missing intermediates 1 and 2, plus the final product 3, of this synthesis:
0
1. Eto
1. Eto-
1
2
2. MeBr
2. EtBr
H3O+
A
3
You can draw the three structures in any arrangement you like.
Explanation
Check
Click and drag to start drawing a structure.
Chapter 9 Solutions
Organic Chemistry
Ch. 9 - Problem 9.1 Label each ether and alcohol in...Ch. 9 - Give the IUPAC name for each compound.Ch. 9 - Problem 9.3 Give the structure corresponding to...Ch. 9 - Name each of the following ethers.Ch. 9 - Name each epoxide.
a. (two ways) b. c. (two...Ch. 9 - Problem 9.6 Rank the following compounds in order...Ch. 9 - Which mechanism is favored by the use of crown...Ch. 9 - Problem 9.8 Draw the organic product of each...Ch. 9 - Prob. 9.9PCh. 9 - Problem 9.10 Draw the products of each reaction.
...
Ch. 9 - Problem 9.11 Draw the products formed when each...Ch. 9 - Prob. 9.12PCh. 9 - Problem 9.13 Draw the structure of each...Ch. 9 - What other alkene is also formed along with Y in...Ch. 9 - Prob. 9.15PCh. 9 - Explain why two substitution products are formed...Ch. 9 - Draw the products of each reaction. a. b. c.Ch. 9 - Problem 9.18 Draw the products of each reaction,...Ch. 9 - Problem 9.19 What is the major product formed...Ch. 9 - Prob. 9.20PCh. 9 - Prob. 9.21PCh. 9 - Problem 9.22 Draw the organic products formed in...Ch. 9 - Problem 9.23 Draw two steps to convert into each...Ch. 9 - Prob. 9.24PCh. 9 - Problem 9.25 Draw the products of each reaction,...Ch. 9 - Draw the products formed when (S)-butan-2-ol is...Ch. 9 - Draw the product formed when (CH3)2CHOH is treated...Ch. 9 - What alkyl halides are formed when each ether is...Ch. 9 - Explain why the treatment of anisole with HBr...Ch. 9 - Name each thiol.
a. b.
Ch. 9 - Draw the product of each reaction. ac b.d.Ch. 9 - Give the IUPAC name for each sulfide.
a. b.
Ch. 9 - Draw the product of each reaction.
a. b.
Ch. 9 - Prob. 9.34PCh. 9 - The cis and trans isomers of 2, 3-dimethyloxirane...Ch. 9 - Problem 9.36 Draw the product of each...Ch. 9 - 9.37 Name each compound depicted in the...Ch. 9 - Answer each question using the ball-and-stick...Ch. 9 - Prob. 9.39PCh. 9 - 9.40 Give IUPAC name for each...Ch. 9 - Prob. 9.41PCh. 9 - Prob. 9.42PCh. 9 - Prob. 9.43PCh. 9 - 9.44 Why is the boiling point of higher than...Ch. 9 - 9.45 Draw the organic product(s) formed when is...Ch. 9 - 9.46 What alkenes are formed when each alcohol is...Ch. 9 - Prob. 9.47PCh. 9 - 9.48 Draw the products of each reaction and...Ch. 9 - 9.49 Draw the product of the following reaction,...Ch. 9 - Prob. 9.50PCh. 9 - Prob. 9.51PCh. 9 - 9.52 Draw a stepwise mechanism for the following...Ch. 9 - 9.53 Although alcohol V gives a single alkene W...Ch. 9 - Prob. 9.54PCh. 9 - Prob. 9.55PCh. 9 - Prob. 9.56PCh. 9 - 9.57 Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 9.58PCh. 9 - 9.59 Draw two different routes to each of the...Ch. 9 - Prob. 9.60PCh. 9 -
9.61 Draw the products formed when each ether is...Ch. 9 - 9.62 Draw a stepwise mechanism for each...Ch. 9 - Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 9.64PCh. 9 - Draw the products of each reaction. a.c. b.d.Ch. 9 - When each halohydrin is treated with, a product of...Ch. 9 - Prob. 9.67PCh. 9 - Prob. 9.68PCh. 9 - Prob. 9.69PCh. 9 - Prob. 9.70PCh. 9 - Prepare each compound from cyclopentanol. More...Ch. 9 - 9.72 Identify the reagents (a–h) needed to carry...Ch. 9 - Prob. 9.73PCh. 9 - 9.74 Treatment of with affords compound A and ....Ch. 9 - Prob. 9.75PCh. 9 - Prob. 9.76PCh. 9 - 9.77 Draw a stepwise, detailed mechanism for the...Ch. 9 - 9.78 Dehydration of with affords
as a minor...Ch. 9 - Prob. 9.79PCh. 9 - 9.80 Draw a stepwise mechanism for the following...Ch. 9 -
9.81 Aziridines are heterocycles that contain an...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. Rub your hands together vigorously. What happens? Discuss the energy transfers and transformations that take...
College Physics: A Strategic Approach (3rd Edition)
Gregor Mendel never saw a gene, yet he concluded that some inherited factors were responsible for the patterns ...
Campbell Essential Biology (7th Edition)
To test your knowledge, discuss the following topics with a study partner or in writing ideally from memory. Th...
HUMAN ANATOMY
What were the major microbiological interests of Martinus Beijerinck and Sergei Winogradsky? It can be said tha...
Brock Biology of Microorganisms (15th Edition)
More than one choice may apply. Using the terms listed below, fill in the blank with the proper term. anterior ...
Essentials of Human Anatomy & Physiology (12th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the missing intermediate 1 and final product 2 of this synthesis: 1. MeO- H3O+ 1 2 2. PrBr Δ You can draw the two structures in any arrangement you like. Click and drag to start drawing a structure.arrow_forwardWhat is the differences between: Glyceride and phosphoglyceride Wax and Fat Soap and Fatty acid HDL and LDL cholesterol Phospho lipids and sphingosine What are the types of lipids? What are the main lipid components of membrane structures? How could lipids play important rules as signaling molecules and building units? The structure variety of lipids makes them to play significant rules in our body, conclude breifly on this statement.arrow_forwardWhat is the differences between DNA and RNA for the following: - structure - function - type What is the meaning of: - replication - transcription - translation show the base pair connection(hydrogen bond) in DNA and RNAarrow_forward
- What is the IP for a amino acid- give an example what are the types of amino acids What are the structures of proteins The N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Try-Val-His Sar-Arg-Val His-Pro-Ala Val- Tyr- Val Arg-Val-Tyr What is the structure of saralasin?arrow_forwardWhat is the IP for a amino acid- give an example what are the types of amino acids What are the structures of proteins The N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Try-Val-His Sar-Arg-Val His-Pro-Ala Val- Tyr- Val Arg-Val-Tyr What is the structure of saralasin?arrow_forward> aw the missing intermediates 1 and 2, plus the final product 3, of this synthesis: 1. Eto 1. EtO¯ H3O+ 1 2 2. PrBr 2. PrBr Δ You can draw the three structures in any arrangement you like. 3 Click and drag to start drawing a structure. Explanation Check 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacarrow_forward
- There are various factors that affect an equilibrium. Give 3 of these factors and explain using examples andequations how an equilibrium is affected by these factors. Please remember that this is a communication question so that you are communicating your understanding of the factors that affect and equilibrium.arrow_forwardEEZE LETCHUP ID Draw the most likely conjugate base resulting from this acid-base reaction. Include all lone pairs. Ignore inorganic byproducts. Drawing く NaOCH2CH3 :0: :0: 狗arrow_forwardAnswerarrow_forward
- 2. Provide a clear arrow-pushing mechanism for the following reactions. Do not skip proton transfers, do not combine steps, and make sure your arrows are clear enough to be interpreted without ambiguity. a. CH3 Ph OEt هد Ph CH3 Hint: the species on the left is an ynolate, which behaves a lot like an enolate.arrow_forwardb. CH3 H3C CH3 CH3 H3C an unexpected product, containing a single 9- membered ring the expected product, containing two fused rings H3C-I (H3C)2CuLi an enolatearrow_forwardb. H3C CH3 1. 2. H3O+ H3C MgBr H3Carrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY