
Chemistry & Chemical Reactivity
9th Edition
ISBN: 9781133949640
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 48GQ
The structure of amphetamine, a stimulant, is shown below. (Replacing one H atom on the NH2, or amino, group with CH3 gives methamphetamine a particularly dangerous drug commonly known as “speed.”)
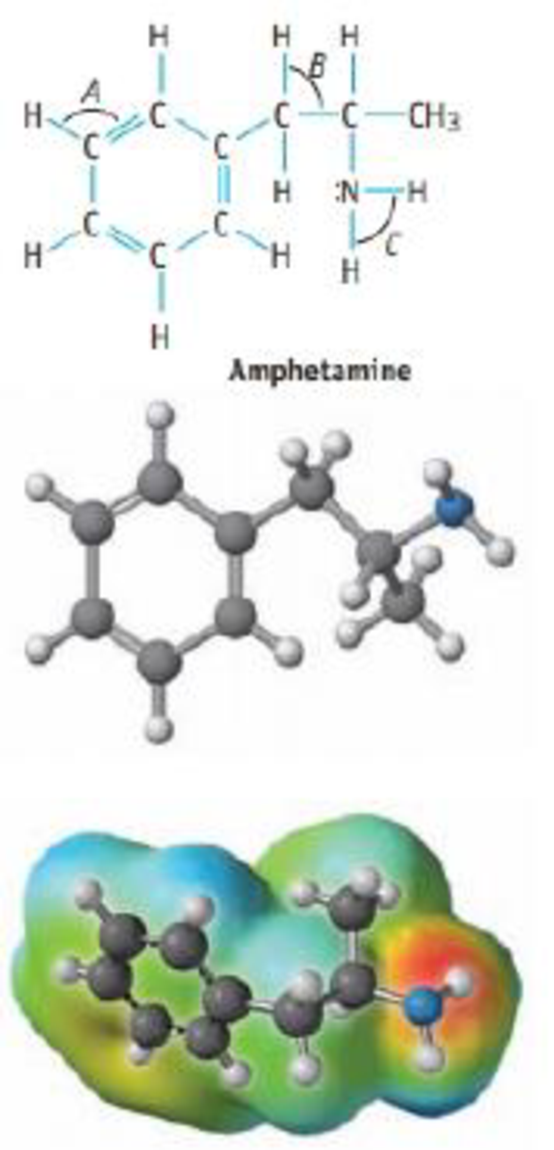
(a) What are the hybrid orbitals used by the C atoms of the C6 ring. by the C atoms of the side chain, and by the N atom?
(b) Give approximate values for the bond angles A, B, and C.
(c) How many σ bonds and π bonds are in the molerule?
(d) Is the molecule polar or nonpolar?
(e) Amphetamine reacts readily with a proton (H+) in aqueous solution. Where does this proton attach to the molecule? Explain how the electrostatic potential map predicts this site of protonation.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method:
Regular Tomato Sauce
Salt Reduced Tomato Sauce
340.0mmol/L
262.7mmol/L
QUESTION: For both groups (Regular & Salt Reduced tomato sauce) of data provide answers to the following calculations below:
1. Standard Deviation (Sx)
2. T Values (t0.05,4)
3. 95% Confidence Interval (mmol/L)
4. [Na+] (mg/100 mL)
5. 95% Confidence Interval (mg/100 mL)
If we have leucine (2-amino-4-methylpentanoic acid), alanine (2-aminopropanoic acid) and phenylalanine (2-amino-3-phenylpropanoic acid), indicate the tripeptides that can be formed (use the abbreviated symbols Leu., Ala and Phe).
Briefly state why trifluoroacetic acid is more acidic than acetic acid.
Chapter 9 Solutions
Chemistry & Chemical Reactivity
Ch. 9.2 - Use valence bond theory to describe the bonding in...Ch. 9.2 - Identify the hybridization of each underlined atom...Ch. 9.2 - Use valence bond theory to describe the bonding in...Ch. 9.2 - What is the hybridization of the S atom (the...Ch. 9.2 - 2. Which of the following is incorrect?
The...Ch. 9.2 - Prob. 3RCCh. 9.2 - Prob. 4RCCh. 9.3 - What is the electron configuration of the H2+ ion?...Ch. 9.3 - Could the anion Li2 exist? What is the ions bond...Ch. 9.3 - The cations O2+ and N2+ are formed when molecules...
Ch. 9.3 - What is the NO bond order in nitrogen monoxide,...Ch. 9.3 - Prob. 2RCCh. 9.3 - Prob. 3RCCh. 9.3 - 4. Among the known dioxygen species (O2+, O2, O2−...Ch. 9.3 - What is the empirical formula of Tynan purple?Ch. 9.3 - Butter yellow absorbs light with a wavelength of...Ch. 9.3 - Prob. 3CSCh. 9.A - Photoelectron spectroscopy is s1milar to the...Ch. 9.A - What is the energy of a photon with a wavelength...Ch. 9.A - Using the accompanying figure, state which...Ch. 9.A - The kinetic energy of an electron ejected from the...Ch. 9.A - The N2+ ions that are formed when electrons with...Ch. 9 - Draw the Lewis structure for chloroform, CHCl3....Ch. 9 - Draw the Lewis structure for NF3. What are its...Ch. 9 - Draw the Lewis structure for hydroxylamine, H2NOH....Ch. 9 - Draw the Lewis structure for 1,...Ch. 9 - Draw the Lewis structure for carbonyl fluoride,...Ch. 9 - Draw the Lewis structure for acetamide, CH3CONH2....Ch. 9 - Specify the electron-pair and molecular geometry...Ch. 9 - Specify the electron-pair and molecular geometry...Ch. 9 - Prob. 9PSCh. 9 - What is the hybrid orbital set used by each of the...Ch. 9 - Draw the Lewis structures of the acid HPO2F2 and...Ch. 9 - Draw the Lewis structures of the arid HSO3F and...Ch. 9 - What is the hybridization of the carbon atom in...Ch. 9 - What is the hybridization of the carbon atoms in...Ch. 9 - What is the electron-pair and molecular geometry...Ch. 9 - Prob. 17PSCh. 9 - For each compound below, decide whether cis and...Ch. 9 - Molecular Orbital Theory (See Examples 9.49.6.)...Ch. 9 - Give the electron configurations for the ions Li2+...Ch. 9 - Platinum hexafluoride is an extremely strong...Ch. 9 - When potassium and oxygen react, one of the...Ch. 9 - Among the following, which has the shortest bond...Ch. 9 - Consider the following list of small molecules and...Ch. 9 - Prob. 27PSCh. 9 - The nitrosyl ion. NO+, has an interesting...Ch. 9 - These questions are not designated as to type or...Ch. 9 - What is the OSO angle and the hybrid orbital set...Ch. 9 - Sketch the resonance structures for the nitrite...Ch. 9 - Sketch the resonance structures for the nitrate...Ch. 9 - Sketch the resonance structures for the N2O...Ch. 9 - Compare the structure and bonding in CO2 and CO32...Ch. 9 - Numerous molecules are detected in deep space....Ch. 9 - Acrolein, a component of photochemical smog, has a...Ch. 9 - The organic compound below is a member of a class...Ch. 9 - The compound sketched below is acetylsalicylic...Ch. 9 - Phosphoserine is a less-common amino acid. (a)...Ch. 9 - Lactic acid is a natural compound found in sour...Ch. 9 - Cinnamaldehyde ocaus naturally in cinnamon oil....Ch. 9 - The ion Si2 was reported in a laboratory...Ch. 9 - The simple valence bond picture of O2 does not...Ch. 9 - Nitrogen, N2, can ionize to form N2+ or add an...Ch. 9 - Which of the homonuclear, diatomic molecules of...Ch. 9 - Which of the following molecules or ions are...Ch. 9 - Prob. 47GQCh. 9 - The structure of amphetamine, a stimulant, is...Ch. 9 - Menthol is used in soaps, perfumes, and foods. It...Ch. 9 - Prob. 50GQCh. 9 - Suppose you carry out the following reaction of...Ch. 9 - Ethylene oxide is an intermediate in the...Ch. 9 - The sulfamate ion, H2NSO3, can be thought of as...Ch. 9 - The compound whose structure is shown here is...Ch. 9 - Prob. 55ILCh. 9 - Carbon dioxide (CO2), dinitrogen monoxide (N2O),...Ch. 9 - Draw the two resonance structures that describe...Ch. 9 - Draw a Lewis structure for diimide, HNNH. Then,...Ch. 9 - Prob. 59SCQCh. 9 - Consider the three fluorides BF4, SiF4, and SF4....Ch. 9 - When two amino acids react with each other, they...Ch. 9 - What is the connection between bond order, bond...Ch. 9 - When is it desirable to use MO theory rather than...Ch. 9 - Show how valence bond theory and molecular orbital...Ch. 9 - Three of the four molecular orbitals for...Ch. 9 - Lets look more closely at the process of...Ch. 9 - Borax has the molecular formula Na2B4O5(OH)4. The...Ch. 9 - A model of the organic compound allene is shown...Ch. 9 - Prob. 69SCQCh. 9 - Prob. 70SCQCh. 9 - Bromine forms a number of oxides of varying...Ch. 9 - Prob. 72SCQCh. 9 - Urea reacts with malonic acid to produce...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain why acid chlorides are more reactive than amides in reactions with nucleophiles.arrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 101.7 mL of a 0.3500M solution of piperidine (C5H10NH) with a 0.05700M solution of HClO4. The pK of piperidine is 2.89. Calculate the pH of the base solution after the chemist has added 682.9 mL of the HClO solution to it. 4 Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HClO solution added. 4 Round your answer to 2 decimal places. pH = .11 00. 18 Ararrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0 262.7 QUESTION: For both groups of data provide answers to the calculations attached in the imagearrow_forward
- 7. Concentration and uncertainty in the estimate of concentration (class data) Class mean for sample (Regular) |[Cl-] (mmol/L) class mean Sn za/2 95% Confidence Interval (mmol/L) [Na+] (mg/100 mL) 95% Confidence Interval (mg/100 mL)arrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forwardGive reason(s) for six from the followings [using equations if possible] a. Addition of sodium carbonate to sulfanilic acid in the Methyl Orange preparation. b. What happened if the diazotization reaction gets warmed up by mistake. c. Addition of sodium nitrite in acidified solution in MO preparation through the diazotization d. Using sodium dithionite dihydrate in the second step for Luminol preparation. e. In nitroaniline preparation, addition of the acid mixture (nitric acid and sulfuric acid) to the product of step I. f. What is the main reason of the acylation step in nitroaniline preparation g. Heating under reflux. h. Fusion of an organic compound with sodium. HAND WRITTEN PLEASEarrow_forward
- edict the major products of the following organic reaction: u A + ? CN Some important notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. Explanation Check Click and drag to start drawing a structure. Х © 2025 McGraw Hill LLC. All Rights Reserved. Te LMUNDARYarrow_forwardSketch the intermediates for A,B,C & D.arrow_forwardCan the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? O ? A . If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. . If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. ㅇ 80 F5 F6 A 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cente FIGarrow_forward
- In methyl orange preparation, if the reaction started with 0.5 mole of sulfanilic acid to form the diazonium salt of this compound and then it converted to methyl orange [0.2 mole]. If the efficiency of the second step was 50%, Calculate: A. Equation(s) of Methyl Orange synthesis: Diazotization and coupling reactions. B. How much diazonium salt was formed in this reaction? C. The efficiency percentage of the diazotization reaction D. Efficiency percentage of the whole reaction.arrow_forwardHand written equations pleasearrow_forwardHand written equations pleasearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY