
Concept explainers
Consider the following
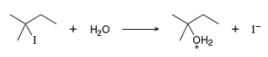
a. Draw a mechanism for this reaction using curved arrows.
b. Draw an energy diagram. Label the axes, the reactants, products,
c. Draw the structure of any transition states.
d. What is the rate equation for this reaction?
e. What happens to the reaction rate in each of the following instances? [1] The leaving group is changed from
(a)
Interpretation: The mechanism of the given reaction is to be drawn by the use of curved arrows.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.58P
The mechanism of the given reaction is shown in Figure 1.
Explanation of Solution
The structure of the given alkyl halide shows that carbon atom, on which bromine is present, is bonded to three other carbon atoms. Hence, the bromine atom is bonded to tertiary carbon atom and the given alkyl halide is
In
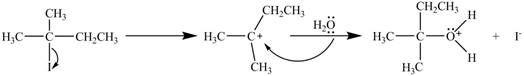
Figure 1
The mechanism of the given reaction is shown in Figure 1.
(b)
Interpretation: The energy diagram is to be drawn. The axes, reactants, products,
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The graphical representation of chemical reaction in which x-axis represents energy of the reaction and y-axis represents the reaction coordinate is called energy profile diagram.
Answer to Problem 7.58P
The energy diagram of the given reaction is shown in Figure 2.
Explanation of Solution
In
The energy of products is equal to the energy of reactants. Therefore, the energy diagram of the given reaction is,
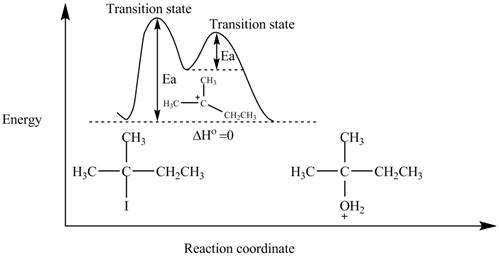
Figure 2
The x-axes of the energy diagram represent the energy of reactants and products and y-axes represent the reaction coordinate.
The energy diagram of the given reaction is shown in Figure 2.
(c)
Interpretation: The structure of the transition state is to be drawn.
Concept introduction: In
Answer to Problem 7.58P
The structure of the transition states is shown in Figure 3.
Explanation of Solution
In
Therefore, the transition states of the given reaction are,
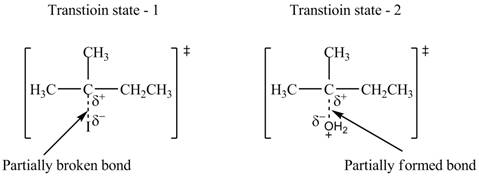
Figure 3
The structure of the transition states is shown in Figure 3.
(d)
Interpretation: The rate equation of the given reaction is to be predicted.
Concept introduction: The rate of
The rate of
Answer to Problem 7.58P
The rate equation of the given reaction is,
Explanation of Solution
The rate of
The rate of
The alkyl halide of given reaction is
Therefore, the rate equation of given reaction is,
The rate equation of the given reaction is,
(e)
Interpretation: The change that occurs to the reaction rate in given instances is to be stated.
Concept introduction: The rate of
The rate of
Answer to Problem 7.58P
The change that occurs to the reaction rate in given instances is,
[1] The rate of the reaction will decrease.
[2] The rate of the reaction will decrease.
[3] The rate of the reaction will decrease.
[4] The rate of the reaction remains unchanged.
[5] The rate of the reaction increases by five times.
Explanation of Solution
[1]
The tertiary halide undergoes nucleophilic substitution by
[2]
The polar protic solvent favors
[3]
The tertiary halide undergoes nucleophilic substitution by
[4]
The rate of
The rate of
According the given statement, the concentration of
Therefore, the rate of the reaction remains unchanged when the concentration of
[5]
The rate of
The rate of
According the given statement, the concentration of
Therefore, the rate of the reaction increases by five times when the concentration
The change that occurs to the reaction rate in given instances is,
[1] The rate of the reaction will decrease.
[2] The rate of the reaction will decrease.
[3] The rate of the reaction will decrease.
[4] The rate of the reaction remains unchanged.
[5] The rate of the reaction increases by five times.
Want to see more full solutions like this?
Chapter 7 Solutions
ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Organic Chemistry
Concepts of Genetics (12th Edition)
Biological Science (6th Edition)
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry (8th Edition)
The Cosmic Perspective (8th Edition)
- Answe Answer A and B pleasearrow_forward3. Refer to the data below to answer the following questions: Isoelectric point Amino Acid Arginine 10.76 Glutamic Acid 3.22 Tryptophan 5.89 A. Define isoelectric point. B. The most basic amino acid is C. The most acidic amino acid is sidizo zoarrow_forward3. A gas mixture contains 50 mol% H2 and 50 mol% He. 1.00-L samples of this gas mixture are mixed with variable volumes of O2 (at 0 °C and 1 atm). A spark is introduced to allow the mixture to undergo complete combustion. The final volume is measured at 0 °C and 1 atm. Which graph best depicts the final volume as a function of the volume of added O2? (A) 2.00 1.75 Final Volume, L 1.50 1.25 1.00 0.75 0.50 0.25 0.00 0.00 0.25 0.50 2.00 (B) 1.75 1.50 Final Volume, L 1.25 1.00 0.75 0.50- 0.25 0.00 0.75 1.00 0.00 0.25 Volume O₂ added, L 2 0.50 0.75 1.00 Volume O₂ added, L 2 2.00 2.00 (C) (D) 1.75 1.75 1.50 1.50 Final Volume, L 1.25 1.00 0.75 0.50 Final Volume, L 1.25 1.00 0.75 0.50 0.25 0.25 0.00 0.00 0.00 0.25 0.50 0.75 1.00 0.00 0.25 Volume O₂ added, L 0.50 0.75 1.00 Volume O₂ added, L 2arrow_forward
- Leucine is an essential amino acid with the systematic name 2-amino-3-methylpentanoic acid. It has pai 2.36 and pKa2 = 9.60. H2N-C(R)H-COOH and R is -CH2-CH(CH3)2 A. Draw the condensed structure for leucine, and label all chirality centers with an asterisk. B. How many possible stereoisomers of leucine are there? C. Draw a Fischer projection of L-leucine and label the chirality center(s) as R or S. D. What is the p/ of leucine? E. Draw the structure of the predominant form of leucine at 10.00. F. Draw the structure of the predominant form of leucine at pH = 1.50. G. Leucine is described as an essential amino acid. What does this mean? H. Show the alkyl halide you would use to prepare leucine by the amidomalonate method. =arrow_forwarda) Write out 6 completely different reactions of acetophenone (reagent, product). b) Write out 3 preparations of 1-methylcyclohexanol, using a different starting material for each one. You may use preps where you just change the functional group, and/or preps where you construct the carbon chain. c) Write out 3 preparations of 2-ethoxybenzoic acid, a different starting material for each one. You may use preps where you just change the functional group, and/or preps where you construct the carbon chain.arrow_forward12. CH3 OH OH H&C CH3 H₂C N OH H₂C CH3 H&C CH3 H₂C' CH3 H.C CH3OH H.C CH2CH3OH CH3CEN Which one of these 17 compounds is represented by this IR and this 'H NMR spectrum? IR Spectrum 3000 4000 3000 NMR Spectrum 2000 £500 RAVENUMBER 2000 1500 9 8 6 5 10 HP-00-290 ppm m 1000 500 1000 4 °arrow_forward
- Draw the structure of (E,6R) 6-methoxy-4-hepten-2-one. Give the IUPAC name of this compound, including stereochemistry. Draw the most stable chair conformation of (cis) 1,3-isobutylcyclohexane. H HC=CCH₂ CH2CH3 EN(CH3)2 -CN(CH3)2arrow_forward10. Write out the mechanism (intermediate/transition state) for this reaction; indicate stereochemistry in product. H3C CH₂OH CH3 SN1 Harrow_forwardWrite "most" under the member of each trio which is most stable. Write "least under the member of each trio which is least stable. b) Draw a Fischer projection of a pair of enantiomers with three chiral carbons. Which of these two would you expect to be more soluble in water? Why? 1-butanol 1-heptanol Which of these two would you expect to have the higher boiling point? Why? hexyl methyl ether 1-heptanolarrow_forward
- Write "most" under the most acidic compound. Write "least" under the least acidic compound. OH NO₂ OCH3 Br 9. Compound X, C50H84F2, reacts with excess H2/Pd to give a C50H88F2 compound. How many rings are in X? How many double bonds are in X? Show your work.arrow_forward4. State whether these two are: a) the same molecule b) c) d) different compounds that are not isomers constitutional isomers diastereomers e) enantiomers CH3 CH₁₂ H OH HO H H OH HO H CH, CH₂ 5. a) How many stereocenters does this compound have? b) How many stereoisomers are possible for this compound? CH₂ OH CHCHarrow_forwardCalculating the pH at equivalence of a titration A chemist titrates 210.0 mL of a 0.1003 M hydrobromic acid (HBr) solution with 0.7550M KOH solution at 25 °C. Calculate the pH at equivalence. Round your answer to 2 decimal places. Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of KOH solution added. pH = ] ☑ o0o 18 Ararrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
