
Chemistry: Atoms First
2nd Edition
ISBN: 9780073511184
Author: Julia Burdge, Jason Overby Professor
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 6.43QP
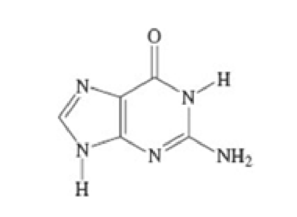
Draw a resonance structure of the guanine molecule shown here, which is part of the DNA structure.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculate the maximum volume of carbon dioxide gas
Predict the major product of the following reaction and then draw a curved arrow mechanism for its formation.
Part: 0/2
Part 1 of 2
H₂SO
heat
: OH
90
Draw the structure of the major product.
Click and drag to start drawing a
structure.
3
Draw a curved arrow mechanism for the reaction, adding steps as necessary. Be sure to include all electrons that are necessary to the mechanism and all
nonzero formal charges.
C
Ö-H
H
+
-S-OH
.0.
Add/Remove step
X
टे
Click and drag to start
drawing a structure.
Chapter 6 Solutions
Chemistry: Atoms First
Ch. 6.2 - Classify the following bonds as nonpolar, polar,...Ch. 6.2 - Classify the following bonds as nonpolar, polar,...Ch. 6.2 - Prob. 1PPBCh. 6.2 - Electrostatic potential maps are shown for HCl and...Ch. 6.2 - Prob. 6.2WECh. 6.2 - Prob. 2PPACh. 6.2 - Prob. 2PPBCh. 6.2 - Prob. 2PPCCh. 6.2 - Prob. 6.3WECh. 6.2 - Prob. 3PPA
Ch. 6.2 - Prob. 3PPBCh. 6.2 - Prob. 3PPCCh. 6.2 - Prob. 6.2.1SRCh. 6.2 - Prob. 6.2.2SRCh. 6.2 - Prob. 6.2.3SRCh. 6.2 - Prob. 6.2.4SRCh. 6.3 - Draw the Lewis structure for carbon disulfide...Ch. 6.3 - Prob. 4PPACh. 6.3 - Prob. 4PPBCh. 6.3 - Prob. 4PPCCh. 6.3 - Prob. 6.3.1SRCh. 6.3 - Prob. 6.3.2SRCh. 6.4 - The widespread use of fertilizers has resulted in...Ch. 6.4 - Prob. 5PPACh. 6.4 - Prob. 5PPBCh. 6.4 - Prob. 5PPCCh. 6.4 - Formaldehyde (CH2O), which can be used 10 preserve...Ch. 6.4 - Prob. 6PPACh. 6.4 - Prob. 6PPBCh. 6.4 - Prob. 6PPCCh. 6.4 - Prob. 6.4.1SRCh. 6.4 - Prob. 6.4.2SRCh. 6.5 - Prob. 6.7WECh. 6.5 - Prob. 7PPACh. 6.5 - Prob. 7PPBCh. 6.5 - Prob. 7PPCCh. 6.5 - Prob. 6.5.1SRCh. 6.5 - Prob. 6.5.2SRCh. 6.6 - Prob. 6.8WECh. 6.6 - Prob. 8PPACh. 6.6 - Prob. 8PPBCh. 6.6 - Prob. 8PPCCh. 6.6 - Prob. 6.9WECh. 6.6 - Prob. 9PPACh. 6.6 - Prob. 9PPBCh. 6.6 - Elements in the same group exhibit similar...Ch. 6.6 - Prob. 6.10WECh. 6.6 - Draw three resonance structures for the hydrogen...Ch. 6.6 - Draw two resonance structures for each speciesone...Ch. 6.6 - Prob. 10PPCCh. 6.6 - Prob. 6.6.1SRCh. 6.6 - Prob. 6.6.2SRCh. 6.6 - Prob. 6.6.3SRCh. 6.6 - Prob. 6.6.4SRCh. 6 - Prob. 6.1QPCh. 6 - Prob. 6.2QPCh. 6 - Prob. 6.3QPCh. 6 - Prob. 6.4QPCh. 6 - Prob. 6.5QPCh. 6 - Prob. 6.6QPCh. 6 - Prob. 6.7QPCh. 6 - Prob. 6.8QPCh. 6 - Prob. 6.9QPCh. 6 - Define electronegativity and explain the...Ch. 6 - Prob. 6.11QPCh. 6 - Prob. 6.12QPCh. 6 - Prob. 6.13QPCh. 6 - Prob. 6.14QPCh. 6 - Prob. 6.15QPCh. 6 - Prob. 6.16QPCh. 6 - Arrange the following bonds in order of increasing...Ch. 6 - Prob. 6.18QPCh. 6 - Prob. 6.19QPCh. 6 - Prob. 6.20QPCh. 6 - Prob. 6.21QPCh. 6 - Prob. 6.22QPCh. 6 - Prob. 6.23QPCh. 6 - Prob. 6.24QPCh. 6 - Prob. 6.25QPCh. 6 - Prob. 6.26QPCh. 6 - Prob. 6.27QPCh. 6 - Prob. 6.28QPCh. 6 - Prob. 6.29QPCh. 6 - Prob. 6.30QPCh. 6 - Prob. 6.31QPCh. 6 - Prob. 6.32QPCh. 6 - Prob. 6.33QPCh. 6 - Prob. 6.34QPCh. 6 - Draw all of the resonance structures for the...Ch. 6 - Prob. 6.36QPCh. 6 - Prob. 6.37QPCh. 6 - Draw three resonance structures for the molecule...Ch. 6 - Draw three reasonable resonance structures for the...Ch. 6 - Indicate which of the following are resonance...Ch. 6 - Prob. 6.41QPCh. 6 - Prob. 6.42QPCh. 6 - Draw a resonance structure of the guanine molecule...Ch. 6 - Prob. 6.44QPCh. 6 - Give three examples of compounds that do not...Ch. 6 - Prob. 6.46QPCh. 6 - Prob. 6.47QPCh. 6 - Prob. 6.48QPCh. 6 - Prob. 6.49QPCh. 6 - Prob. 6.50QPCh. 6 - Prob. 6.51QPCh. 6 - Prob. 6.52QPCh. 6 - Prob. 6.53QPCh. 6 - Draw Lewis structures for the radical species ClF2...Ch. 6 - Prob. 6.55QPCh. 6 - Prob. 6.56QPCh. 6 - Prob. 6.57QPCh. 6 - Prob. 6.58QPCh. 6 - Prob. 6.59QPCh. 6 - Prob. 6.60QPCh. 6 - Give an example of an ion or molecule containing...Ch. 6 - Prob. 6.62QPCh. 6 - Prob. 6.63QPCh. 6 - Prob. 6.64QPCh. 6 - Are the following statements true or false? (a)...Ch. 6 - Prob. 6.66QPCh. 6 - Prob. 6.67QPCh. 6 - Most organic acids can be represented as RCOOH,...Ch. 6 - Prob. 6.69QPCh. 6 - Prob. 6.70QPCh. 6 - Prob. 6.71QPCh. 6 - The following species have been detected in...Ch. 6 - Prob. 6.73QPCh. 6 - Prob. 6.74QPCh. 6 - The triiodide ion (I3) in which the I atoms are...Ch. 6 - Prob. 6.76QPCh. 6 - Prob. 6.77QPCh. 6 - The chlorine nitrate (ClONO2) molecule is believed...Ch. 6 - Prob. 6.79QPCh. 6 - For each of the following organic molecules draw a...Ch. 6 - Prob. 6.81QPCh. 6 - Draw Lewis structures for the following organic...Ch. 6 - Draw Lewis structures for the following four...Ch. 6 - Prob. 6.84QPCh. 6 - Prob. 6.85QPCh. 6 - Draw three resonance structures for (a) the...Ch. 6 - Prob. 6.87QPCh. 6 - Prob. 6.88QPCh. 6 - Prob. 6.89QPCh. 6 - Draw a Lewis structure for nitrogen pentoxide...Ch. 6 - Prob. 6.91QPCh. 6 - Nitrogen dioxide (NO2) is a stable compound....Ch. 6 - Prob. 6.93QPCh. 6 - Vinyl chloride (C2H3Cl) differs from ethylene...Ch. 6 - Prob. 6.95QPCh. 6 - Prob. 6.96QPCh. 6 - In 1999 an unusual cation containing only nitrogen...Ch. 6 - Prob. 6.98QPCh. 6 - Prob. 6.99QPCh. 6 - Electrostatic potential maps for three compounds...Ch. 6 - Which of the following atoms must always obey the...Ch. 6 - Prob. 6.2KSPCh. 6 - Prob. 6.3KSPCh. 6 - How many lone pairs are on the central atom in the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw a curved arrow mechanism for its formation. You may need to re-draw structures to show certain bonds. Ensure that HSO is used as the base to deprotonate the ẞ carbon when necessary. C HO : OH HO: OH =s = + 1 Add/Remove step X Click and drag to start drawing a structure.arrow_forwardWhich of the following could 1,2-ethanediol be directly synthesized from? OH HO О 0 0. O ?arrow_forwardDesign a synthesis of 1,2-diethoxyethane from an alkene. Select the single best answer for each part. Part: 0/3 Part 1 of 3 Which of the following could 1,2-diethoxyethane be directly synthesized from? O HO 0 HO.... OH HO HO × 5 > ?arrow_forward
- Using reaction free energy to predict equilibrium composition Consider the following equilibrium: N2 (g) + 3H2 (g) = 2NH3 (g) AG⁰ = -34. KJ Now suppose a reaction vessel is filled with 8.06 atm of nitrogen (N2) and 2.58 atm of ammonia (NH3) at 106. °C. Answer the following questions about this system: ? rise Under these conditions, will the pressure of N2 tend to rise or fall? ☐ x10 fall Is it possible to reverse this tendency by adding H₂? In other words, if you said the pressure of N2 will tend to rise, can that be changed to a tendency to fall by adding H₂? Similarly, if you said the pressure of N2 will tend to fall, can that be changed to a tendency to rise by adding H₂? If you said the tendency can be reversed in the second question, calculate the minimum pressure of H₂ needed to reverse it. Round your answer to 2 significant digits. yes no ☐ atm ☑ 5 00. 18 Ararrow_forwardi need help with the followingarrow_forwardUsing reaction free energy to predict equilibrium composition Consider the following equilibrium: 2NO(g) +Cl₂ (g) = 2NOC1 (g) AGº = -41. kJ Now suppose a reaction vessel is filled with 8.90 atm of chlorine (C12) and 5.71 atm of nitrosyl chloride (NOC1) at 1075. °C. Answer the following questions about this system: rise Under these conditions, will the pressure of NOCI tend to rise or fall? x10 fall Is it possible to reverse this tendency by adding NO? In other words, if you said the pressure of NOCI will tend to rise, can that be changed to a tendency to fall by adding NO? Similarly, if you said the pressure of NOCI will tend to fall, can that be changed to a tendency to rise by adding NO? yes no If you said the tendency can be reversed in the second question, calculate the minimum pressure of NO needed to reverse it. Round your answer to 2 significant digits. atm ☑ 18 Ararrow_forward
- Identifying the major species in weak acid or weak base equilibria The preparations of two aqueous solutions are described in the table below. For each solution, write the chemical formulas of the major species present at equilibrium. You can leave out water itself. Write the chemical formulas of the species that will act as acids in the 'acids' row, the formulas of the species that will act as bases in the 'bases' row, and the formulas of the species that will act as neither acids nor bases in the 'other' row. You will find it useful to keep in mind that HCN is a weak acid. acids: 0.29 mol of NaOH is added to 1.0 L of a 1.2M HCN solution. bases: ☑ other: 0.09 mol of HCl is added to acids: 1.0 L of a solution that is bases: 0.3M in both HCN and KCN. other: 0,0,... ? 00. 18 Ar 日arrow_forwardIdentifying the major species in weak acid or weak base equilibria The preparations of two aqueous solutions are described in the table below. For each solution, write the chemical formulas of the major species present at equilibrium. You can leave out water itself. Write the chemical formulas of the species that will act as acids in the 'acids' row, the formulas of the species that will act as bases in the 'bases' row, and the formulas of the species that will act as neither acids nor bases in the 'other' row. You will find it useful to keep in mind that HF is a weak acid. acids: 0.2 mol of KOH is added to 1.0 L of a 0.5 M HF solution. bases: Х other: ☐ acids: 0.10 mol of HI is added to 1.0 L of a solution that is 1.4M in both HF and NaF. bases: other: ☐ 0,0,... ด ? 18 Ararrow_forwardIdentifying the major species in weak acid or weak base equilibria The preparations of two aqueous solutions are described in the table below. For each solution, write the chemical formulas of the major species present at equilibrium. You can leave out water itself. Write the chemical formulas of the species that will act as acids in the 'acids' row, the formulas of the species that will act as bases in the 'bases' row, and the formulas of the species that will act as neither acids nor bases in the 'other' row. You will find it useful to keep in mind that NH3 is a weak base. acids: ☐ 1.8 mol of HCl is added to 1.0 L of a 1.0M NH3 bases: ☐ solution. other: ☐ 0.18 mol of HNO3 is added to 1.0 L of a solution that is 1.4M in both NH3 and NH₁Br. acids: bases: ☐ other: ☐ 0,0,... ? 000 18 Ar B 1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY