
Concept explainers
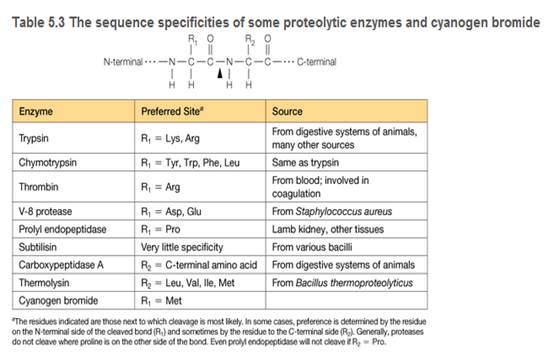
You are a summer intern in a clinical hematology lab. The lab director gives you a sample of a patient's blood proteins and asks you to characterize the thrombin in the sample. She also tells you that thrombin is a serine protease important in blood clotting (see Table 5.3), and this patient is a newborn with uncontrolled bleeding.
a. To characterize the thrombin in the sample, you must remove two proteins that interfere with the thrombin activity assay: cytochrome c and lactoglobin. You find some CM-cellulose (see Figure 5A.5) and a phosphate buffer (pH 6.4) on the shelf in your lab. You decide to load the protein sample onto a column of CM-cellulose equilibrated in the pH = 6.4 buffer. Predict the order of elution for the three proteins shown in the accompanying table. At pH = 6.4, which protein(s) do you predict will remain bound to the column?
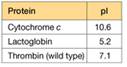
b. List two different ways you could change the buffer to elute the bound protein(s) and achieve proper separation of the proteins.
c. You are surprised to observe that the patient's thrombin flows through the CM-cellulose column at pH = 6.4 and does not bind. Confident in your technique, you suspect the patient's thrombin is different from wild-type thrombin. Using a different buffer system, you manage to purify some of the patient's thrombin, and you submit the purified sample for amino acid sequencing. The sequence analysis shows that the patient's thrombin contains a mutation in the enzyme active site. A lysine residue in the wild type has been mutated to an asparagine in the patient's thrombin. Does this mutation explain the anomalous CM- cellulose binding behavior you observed?
d. How many
e. Based on their side-chain structures, compare and contrast the potential of Lys and Asn to form noncovalent interactions. In other words, can each form H bonds and/or salt bridges and/or van der Waals contacts?
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Biochemistry: Concepts and Connections
- write the ionization equilibrium for cysteine and calculate the piarrow_forwardplease answerarrow_forwardf. The genetic code is given below, along with a short strand of template DNA. Write the protein segment that would form from this DNA. 5'-A-T-G-G-C-T-A-G-G-T-A-A-C-C-T-G-C-A-T-T-A-G-3' Table 4.5 The genetic code First Position Second Position (5' end) U C A G Third Position (3' end) Phe Ser Tyr Cys U Phe Ser Tyr Cys Leu Ser Stop Stop Leu Ser Stop Trp UCAG Leu Pro His Arg His Arg C Leu Pro Gln Arg Pro Leu Gin Arg Pro Leu Ser Asn Thr lle Ser Asn Thr lle Arg A Thr Lys UCAG UCAC G lle Arg Thr Lys Met Gly Asp Ala Val Gly Asp Ala Val Gly G Glu Ala UCAC Val Gly Glu Ala Val Note: This table identifies the amino acid encoded by each triplet. For example, the codon 5'-AUG-3' on mRNA specifies methionine, whereas CAU specifies histidine. UAA, UAG, and UGA are termination signals. AUG is part of the initiation signal, in addition to coding for internal methionine residues. Table 4.5 Biochemistry, Seventh Edition 2012 W. H. Freeman and Company B eviation: does it play abbreviation:arrow_forward
- Answer all of the questions please draw structures for major productarrow_forwardfor glycolysis and the citric acid cycle below, show where ATP, NADH and FADH are used or formed. Show on the diagram the points where at least three other metabolic pathways intersect with these two.arrow_forwardanswer the questions please all of them should be answeredarrow_forward
- Burk plot is shown below. Calculate Km and max for this enzyme. show workarrow_forwardInsert Format Tools Extensions Help Normal text ▾ Arial C 2 10 3 + BIUA Student Guide (continued) Record data and conclusions about the mystery food sample either below or in a lab notebook. Step 2: Protein Test (Biuret Solution) Gelatin Water [Mystery Food (Positive Control) (Negative Control) Sample pink purple no change no change They mystery food sample does not contain protein because the color of the test tube wasn't pink or purple Color Conclusion They mystery food sample does not contain protein because the color of the test tube wasn't pink or purple Step 3: Lipid Test (Sudan Red Solution) Vegetable Oil Water (Positive Control) (Negative Control) Mystery Food Sample floating red no change floating red the mystery food dosnt contain lipids because the test tube has floating red 75 % 87 8 9 7 ChromeOS C Device will pow 26.battery lea powerarrow_forwardThe rate data from an enzyme catalyzed reaction with and without an inhibitor present is found in the image. Question: what is the KM and Vm and the nature of inhibitionarrow_forward
- 1. Estimate the concentration of an enzyme within a living cell. Assume that: (a): fresh tissue is 80% water and all of it is intracellular (b): the total soluble protein represents 15% of the weight (c): all the soluble proteins are enzymes (d): the average molecular weight of the proteins is 150,000 (E): about 100 different enzymes are present please help I am lostarrow_forwardPlease helparrow_forwardThe following data were recorded for the enzyme catalyzed conversion of S -> P. Question: Estimate the Vmax and Km. What would be the rate at 2.5 and 5.0 x 10-5 M [S] ?arrow_forward
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College





