
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 9Q
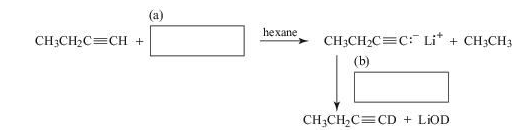
Supply the missing reagents.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
3. Assign absolute configuration (Rors) to each chirality center.
a.
H
Nitz
C.
он
b.
0
H-C. C
H
7
C.
་-4
917-417
refs
H
1つ
८
ડુ
d.
Но
f.
-2-
01
Ho
-OH
2HN
How many signals do you expect in the H NMR spectrum for this molecule?
Br
Br
Write the answer below.
Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H
atoms that would contribute to the same signal as the H already highlighted red.
Note for advanced students: In this question, any multiplet is counted as one signal.
Number of signals in the 'H NMR spectrum.
For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to
the same signal as the H atom already highlighted red.
If no other H atoms will contribute, check the box at right.
No additional Hs to color in top
molecule
For the molecule in the bottom drawing area, highlight in red any other H atoms that will
contribute to the same signal as the H atom already highlighted red.
If no other H atoms will contribute, check the box at right.
No additional Hs to color in bottom
molecule
In the drawing area below, draw the major products of this organic reaction:
1. NaOH
?
2. CH3Br
If there are no major products, because nothing much will happen to the reactant under these reaction conditions, check the box under the drawing area
instead.
No reaction.
Click and drag to start drawing a
structure.
☐ : A
ค
Chapter 3 Solutions
Organic Chemistry
Ch. 3 - Prob. 1PPCh. 3 - PRACTICE PROBLEM 3.2
Write equations showing the...Ch. 3 - PRACTICE PROBLEM 3.3 Which of the following are...Ch. 3 - Prob. 4PPCh. 3 - PRACTICE PROBLEM 3.5 Formic acid (HCO2H) has...Ch. 3 - Prob. 6PPCh. 3 - Prob. 7PPCh. 3 - Prob. 8PPCh. 3 - PRACTICE PROBLEM 3.9 Predict the outcome of the...Ch. 3 - Prob. 10PP
Ch. 3 - Prob. 11PPCh. 3 - Prob. 12PPCh. 3 - Prob. 13PPCh. 3 - Prob. 14PPCh. 3 - Prob. 15PPCh. 3 - Prob. 16PPCh. 3 - Prob. 17PPCh. 3 - What is the conjugate base of each of the...Ch. 3 - List the bases you gave as answers to Problem 3.20...Ch. 3 - 3.22 What is the conjugate acid of each of the...Ch. 3 - List the acids you gave as answers to Problem 3.22...Ch. 3 - Designate the Lewis acid and Lewis base in each of...Ch. 3 - Prob. 23PCh. 3 - Prob. 24PCh. 3 - Write an equation, using the curved-arrow...Ch. 3 - When methyl alcohol is treated with NaH, the...Ch. 3 - 3.30 What reaction will take place if ethyl...Ch. 3 - 3.31 (a) The of formic acid. What is the? (b)...Ch. 3 - Acid HA has pKa=20; acid HB has pKa=10. (a) Which...Ch. 3 - Prob. 30PCh. 3 - 3.34 (a) Arrange the following compounds in order...Ch. 3 - 3.35 Arrange the following compounds in order of...Ch. 3 - 3.36 Arrange the following in order of increasing...Ch. 3 - Prob. 34PCh. 3 - 3.38 Supply the curved arrows necessary for the...Ch. 3 - Glycine is an amino acid that can be obtained from...Ch. 3 - 3.40 Malonic acid, , is a diprotic acid. The for...Ch. 3 - 3.41 The free-energy change, , for the ionization...Ch. 3 - 3.42 At the enthalpy change, , for the ionization...Ch. 3 - The compound at right has (for obvious reasons)...Ch. 3 - 3.44.
(a) Given the above sequence of...Ch. 3 - Prob. 42PCh. 3 - Prob. 43PCh. 3 - 3.47 As noted in Table 3.1, the of acetone, , is...Ch. 3 - Formamide (HCONH2) has a pKa of approximately 25....Ch. 3 - List all the chemical species likely to be present...Ch. 3 - Prob. 2LGPCh. 3 - Prob. 3LGPCh. 3 - Prob. 4LGPCh. 3 - Prob. 1QCh. 3 - Which of the following is the strongest base? (a)...Ch. 3 - Prob. 3QCh. 3 - Prob. 4QCh. 3 - Prob. 5QCh. 3 - Which would be the weakest base? (a) CH3CO2Na (b)...Ch. 3 - What acid-base reaction (if any) would occur when...Ch. 3 - The pKa of CH3NH3+ equals 10.6; the pKa of...Ch. 3 - 3.9 Supply the missing reagents.
Ch. 3 - 3.10 Supply the missing intermediates and...
Additional Science Textbook Solutions
Find more solutions based on key concepts
14. Squid Propulsion
Squid usually move by using their tins, but they can utilize a form of “jet propulsion,” e...
College Physics: A Strategic Approach (3rd Edition)
Why can algae and cyanobacteria be considered indicators of productivity as well as of pollution?
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
The bioremediation process shown in the photograph is used to remove benzene and other hydrocarbons from soil c...
Microbiology: An Introduction
A womans father has ornithine transcarbamylase deficiency (OTD), an X-linked recessive disorder producing menta...
Genetic Analysis: An Integrated Approach (3rd Edition)
89. Classify each chemical reaction as a synthesis, decomposition, single-displacement, or double-displacement ...
Introductory Chemistry (6th Edition)
Pigeons may exhibit a checkered or plain color pattern. In a series of controlled matings, the following data w...
Concepts of Genetics (12th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the major products of the following organic reaction: NC Δ ? Some important Notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to draw bonds carefully to show important geometric relationships between substituents. Note: if your answer contains a complicated ring structure, you must use one of the molecular fragment stamps (available in the menu at right) to enter the ring structure. You can add any substituents using the pencil tool in the usual way. Click and drag to start drawing a structure. Х аarrow_forwardPredict the major products of this organic reaction. Be sure you use dash and wedge bonds to show stereochemistry where it's important. + ☑ OH 1. TsCl, py .... 文 P 2. t-BuO K Click and drag to start drawing a structure.arrow_forwardConsider this organic reaction: ( Draw the major products of the reaction in the drawing area below. If there won't be any major products, because this reaction won't happen at a significant rate, check the box under the drawing area instead. Click and drag to start drawing a structure. Х : а ค 1arrow_forward
- In the drawing area below, draw the major products of this organic reaction: If there are no major products, because nothing much will happen to the reactant under these reaction conditions, check the box under the drawing area instead. 1. NaH 2. CH3Br ? Click and drag to start drawing a structure. No reaction. : ☐ Narrow_forward+ Predict the major product of the following reaction. : ☐ + ☑ ค OH H₂SO4 Click and drag to start drawing a structure.arrow_forwardConsider this organic reaction: ... OH CI Draw the major products of the reaction in the drawing area below. If there won't be any major products, because this reaction won't happen at a significant rate, check the box under the drawing area instead. ☐ No Reaction. Click and drag to start drawing a structure. : аarrow_forward
- Consider the following reactants: Br Would elimination take place at a significant rate between these reactants? Note for advanced students: by significant, we mean that the rate of elimination would be greater than the rate of competing substitution reactions. yes O no If you said elimination would take place, draw the major products in the upper drawing area. If you said elimination would take place, also draw the complete mechanism for one of the major products in the lower drawing area. If there is more than one major product, you may draw the mechanism that leads to any of them. Major Products:arrow_forwardDraw one product of an elimination reaction between the molecules below. Note: There may be several correct answers. You only need to draw one of them. You do not need to draw any of the side products of the reaction. OH + ! : ☐ + Х Click and drag to start drawing a structure.arrow_forwardFind one pertinent analytical procedure for each of following questions relating to food safety analysis. Question 1: The presence of lead, mercury and cadmium in canned tuna Question 2: Correct use of food labellingarrow_forward
- Formulate TWO key questions that are are specifically in relation to food safety. In addition to this, convert these questions into a requirement for chemical analysis.arrow_forwardWhat are the retrosynthesis and forward synthesis of these reactions?arrow_forwardWhich of the given reactions would form meso product? H₂O, H2SO4 III m CH3 CH₂ONa CH3OH || H₂O, H2SO4 CH3 1. LiAlH4, THF 2. H₂O CH3 IVarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY