
EBK BIOLOGY
10th Edition
ISBN: 8220100474729
Author: Martin
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 2TYU
VISUALIZE The structures depicted are (a) enantiomers (b) different views of the same molecule (c) geometric (cis–trans) isomers (d) both geometric isomers and enantiomers (e) structural isomers
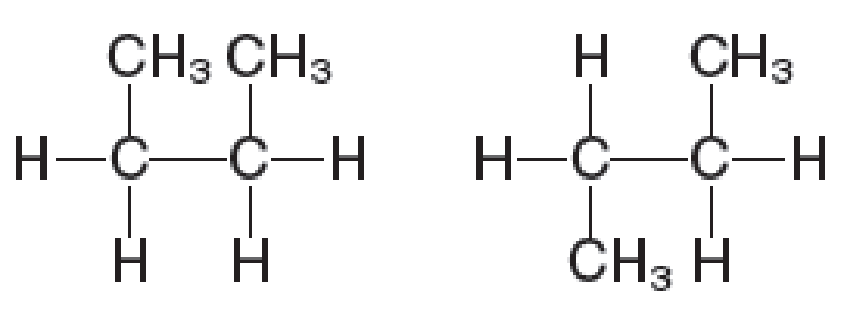
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
B-pt and M-pt of various organic compounds depends on intermolecular forces of attraction which depend on following
(a) Inter molecular/intramolecular H-bond.
(b) Dipole-Dipole interaction (Carboxyl and ether).
(c) Molecular size
(d) Surface area. (branching).
A monosaccharide designated as an aldehyde sugar contains(a) a terminal carboxyl group (b) an internal carboxyl group(c) a terminal carbonyl group (d) an internal carbonyl group(e) a terminal carboxyl group and an internal carbonyl group
Which of the structures below DOES NOT correspond to a "correct" (i.e. probable or possible) structure for a monosaccharide? (remember that unlabeled vertices are carbon atoms)
a) A
b) B
c) C
d) D
Chapter 3 Solutions
EBK BIOLOGY
Ch. 3.1 - Describe the properties of carbon that make it the...Ch. 3.1 - Define the term isomer and distinguish among the...Ch. 3.1 - Identify the major functional groups present in...Ch. 3.1 - Explain the relationship between polymers and...Ch. 3.1 - What are some of the ways that the features of...Ch. 3.1 - Prob. 2CCh. 3.1 - Prob. 3CCh. 3.1 - Prob. 4CCh. 3.1 - Prob. 5CCh. 3.2 - Distinguish among monosaccharides, disaccharides,...
Ch. 3.2 - Prob. 1CCh. 3.3 - Distinguish among fats, phospholipids, and...Ch. 3.3 - Prob. 1CCh. 3.3 - Explain why the structure of phospholipids enables...Ch. 3.4 - Give an overall description of the structure and...Ch. 3.4 - Prob. 8LOCh. 3.4 - Distinguish among the four levels of organization...Ch. 3.4 - Prob. 1CCh. 3.4 - Prob. 2CCh. 3.5 - Describe the components of a nucleotide. Name some...Ch. 3.5 - VISUALIZE Sketch a pyrimidine nucleotide subunit...Ch. 3.6 - Compare the functions and chemical compositions of...Ch. 3.6 - How can you distinguish a pentose sugar from a...Ch. 3 - Prob. 1TYUCh. 3 - VISUALIZE The structures depicted are (a)...Ch. 3 - Prob. 3TYUCh. 3 - The synthetic process by which monomers are...Ch. 3 - A monosaccharide designated as an aldehyde sugar...Ch. 3 - Structural polysaccharides typically (a) have...Ch. 3 - Saturated fatty acids are so named because they...Ch. 3 - Fatty acids in phospholipids and triacylglycerols...Ch. 3 - Which of the following levels of protein structure...Ch. 3 - Which of the following associations between R...Ch. 3 - Each phosphodiester linkage in DNA or RNA includes...Ch. 3 - PREDICT Do any of the amino acid side groups shown...Ch. 3 - PREDICT Like oxygen, sulfur forms two covalent...Ch. 3 - Hydrogen bonds and van der Waals interactions are...Ch. 3 - EVOLUTION LINK In what ways are all species alike...Ch. 3 - EVOLUTION LINK The total number of possible amino...Ch. 3 - EVOLUTION LINK Each amino acid could potentially...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- The synthetic process by which monomers are covalently linked is (a) hydrolysis (b) isomerization (c) condensation (d) glycosidic linkage (e) ester linkagearrow_forwardUsing the appropriate chemical structures describe the monomers and polymers for each of the following macromolecules; a) fats b) nucleic acidarrow_forwardUsing the appropriate chemical structures describe the monomers and polymers for each of the following macromolecules; a) proteins b) carbohydratesarrow_forward
- Φ and ψ in the Ramachandran plot (below) are: a) Rotational angles around the bond between the α-carbon and N-H (Φ) and C=O (ψ). b) Amino acid solubility in octanol (Φ) and water (ψ). c) Hydrogen bond angles in α-helices (Φ) and β-sheets (ψ). d) Amino acid solubility in water (Φ) and octanol (ψ).arrow_forwardAnswer (a.) If compound X is aspartic acid and compound Y is lysinearrow_forwardDraw all three staggered conformations for the following compound, viewed along the C₂-C3 bond. Determine which conformation is the most stable, taking into account gauche interactions and hydrogen-bonding interactions. (J. Phys. Chem. A 2001, 105, 6991-6997) Provide a reason for your choice by identifying all of the interactions that led to your decision. H H&COH C₂ C3 HỌ CH CH3 Which of the following is the most stable staggered conformer? Me H OH Me OH Me- Me Me H Conformer A OH OH OH HO H H H Conformer B Conformer C Me Conformer B because the anti OH groups avoid hydrogen bonding, which is a destabilizing effect. Conformer A because there are only two gauche interactions, and hydrogen bonding between the two OH groups is a stabilizing effect. Conformer C because there is no steric strain for Me-OH gauche interactions and hydrogen bonding between the two OH groups is a stabilizing effect. Conformer B because the large OH groups are anti to each other.arrow_forward
- (a) A homogeneous mixture which contains water as a solvent is called (b) Ni(CIO4)2-6H2O is hydrated whereas Ni(CIO.)e is (c) NaCl contains an bond whereas O2(g) contains a bond (d) A homogeneous mixture has a and composition (e) Temperature is an because it does not depend on the amount of substance (f) The maximum number of electrons that an orbital can have is (9) The energy of the lowest level in the H atom is (h) Arrange the following subshells in the H atom in order of increasing energy: 3s 4d 2р 4f 3d 2s 3p () Wavelength and frequency of radiation have an relationshiparrow_forwardNonearrow_forwardThe structures below are two different _______________ . a) Conformational Isomers b) Stereoisomers c) Constitutional Isomers d) Views of the same moleculearrow_forward
- Draw the following molecular structures: (a) disaccharide that is prominent in starch (Haworth or Chair Haworth structure) (b) disaccharide that is prominent in cellulose (Haworth or Chair Haworth structure) (c) disaccharide that is prominent in chitin (Haworth or Chair Haworth structure) (d) the monolignol (monomer) that is most prominent in lignin extracted from grassesarrow_forwardPlease identify the interactions shown in a) b) c) d) Please explain how they contribute to this molecule.arrow_forwardCONH, HQ NH (CH)3C a.) Put asterisk on chiral centers b.) label each chiral center as R or S. c.) Draw the enantiomer d.) Draw one diastereomerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license