
ORGANIC CHEMISTRY
5th Edition
ISBN: 9781259977596
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 24.69P
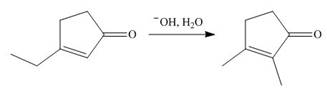
Devise a stepwise mechanism for the following reaction. (Hint: The mechanism begins with the conjugate addition of
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Describe a sequence of photophysical processes that can be followed by radiation adsorbed by a molecule in the ground state to give rise to phosphorescent emission.
State two similarities between fluorescence and phosphorescence.
State three photophysical processes that can be related to the effects of incident radiation on a molecule in its ground state. Consider that radiation can give rise to fluorescent emission, but not phosphorescent emission.
Chapter 24 Solutions
ORGANIC CHEMISTRY
Ch. 24 - Prob. 24.1PCh. 24 - Prob. 24.2PCh. 24 - Problem 24.3
What unsaturated carbonyl compound is...Ch. 24 - Prob. 24.4PCh. 24 - Prob. 24.5PCh. 24 - Prob. 24.6PCh. 24 - Problem 24.7
Draw the products formed in each...Ch. 24 - Problem 24.8
Draw the products formed in the...Ch. 24 - Prob. 24.9PCh. 24 - Prob. 24.10P
Ch. 24 - Prob. 24.11PCh. 24 - Prob. 24.12PCh. 24 - Prob. 24.13PCh. 24 - Prob. 24.14PCh. 24 - Prob. 24.15PCh. 24 - Problem 24.16
What ester is formed when each...Ch. 24 - Prob. 24.17PCh. 24 - Prob. 24.18PCh. 24 -
Draw the products of each reaction.
a. b.
Ch. 24 - Problem 24.20
Two steps in a synthesis of the...Ch. 24 - Prob. 24.21PCh. 24 - Problem 24.22
Which of the following compounds can...Ch. 24 - Prob. 24.23PCh. 24 - Problem 24.24
What starting materials are needed...Ch. 24 - Problem 24.25
Draw the products when each pair of...Ch. 24 - Prob. 24.26PCh. 24 - Problem 24.27
What starting materials are needed...Ch. 24 - Prob. 24.28PCh. 24 - 24.29 What steps are needed to convert A to B?
Ch. 24 - Prob. 24.30PCh. 24 - 24.31 Draw the product formed in each directed...Ch. 24 - Prob. 24.32PCh. 24 - 24.33 What starting materials are needed to...Ch. 24 - Prob. 24.34PCh. 24 - Prob. 24.35PCh. 24 - 24.36 Identify the structures of C and D in the...Ch. 24 - Prob. 24.37PCh. 24 - Prob. 24.38PCh. 24 - 24.39 Draw the product formed from a Claisen...Ch. 24 - Prob. 24.40PCh. 24 - 24.41 Even though B contains three ester groups, a...Ch. 24 - Prob. 24.42PCh. 24 - Prob. 24.43PCh. 24 - 24.44 Vetivone is isolated from vetiver, a...Ch. 24 - Draw the product of each Robinson annulation from...Ch. 24 - Prob. 24.46PCh. 24 - 24.47 Draw the organic products formed in each...Ch. 24 - 24.48 Fill in the lettered reagents needed for...Ch. 24 - Prob. 24.49PCh. 24 - Prob. 24.50PCh. 24 - Prob. 24.51PCh. 24 - 24.52 Draw a stepwise mechanism for the following...Ch. 24 - Prob. 24.53PCh. 24 - Prob. 24.54PCh. 24 - Prob. 24.55PCh. 24 - Prob. 24.56PCh. 24 - Prob. 24.57PCh. 24 - Prob. 24.58PCh. 24 - Prob. 24.59PCh. 24 - 24.60 Devise a synthesis of each compound from the...Ch. 24 - 24.61 Devise a synthesis of each compound from...Ch. 24 - 24.62 Devise a synthesis of each compound from ,...Ch. 24 - Prob. 24.63PCh. 24 - Prob. 24.64PCh. 24 - 24.65 Answer the following questions about...Ch. 24 - Prob. 24.66PCh. 24 - Prob. 24.67PCh. 24 - Prob. 24.68PCh. 24 - 24.69 Devise a stepwise mechanism for the...Ch. 24 - 24.70 Draw a stepwise mechanism for the following...Ch. 24 - Prob. 24.71PCh. 24 - Prob. 24.72PCh. 24 - Prob. 24.73P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License