
Concept explainers
Fill in the lettered reagents needed for each reaction.
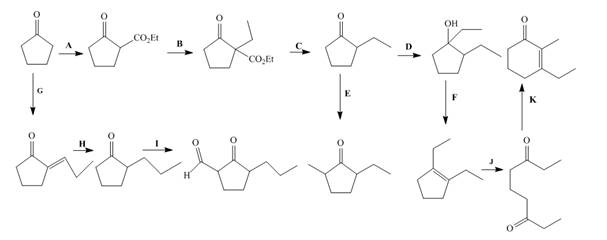
Interpretation: The lettered reagents that are needed for the given reactions are to be shown.
Concept Introduction: In crossed claisen condensation reaction, the base abstracts the acidic proton from
Answer to Problem 24.48P
The lettered reagents that are needed for the given reactions are shown below.
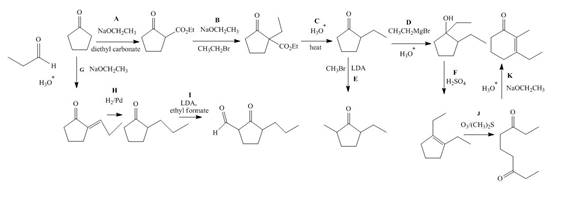
Explanation of Solution
The lettered reagent A is shown as,
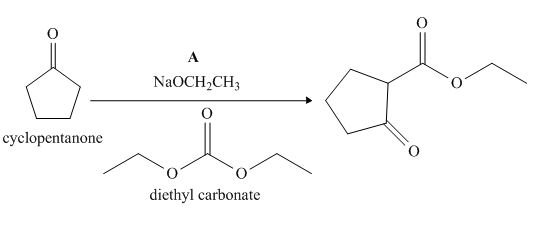
Figure 1
The given compound, cyclopentanone is treated with the base, sodium ethoxide that results in the formation of an enolate ion. Then, the enolate ion reacts with the compound, diethyl carbonate to form the desired compound,
The lettered reagent B is shown as,
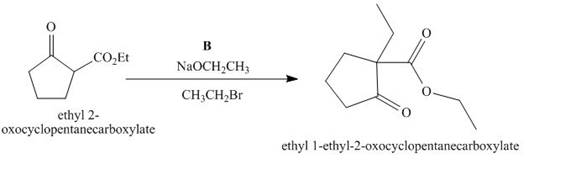
Figure 2
The compound,
The lettered reagent C is shown as,
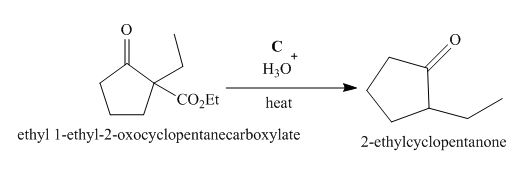
Figure 3
The compound,
The lettered reagent D is shown as,
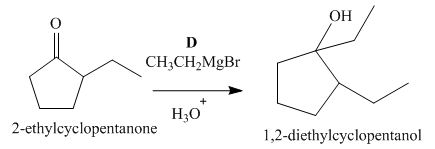
Figure 4
The compound,
The lettered reagent E is shown as,
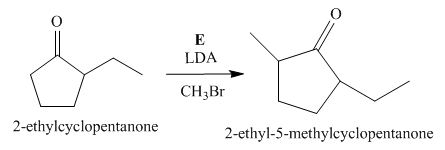
Figure 5
The compound,
The lettered reagent F is shown as,
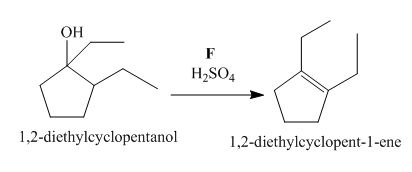
Figure 6
The compound,
The lettered reagent G is shown as,
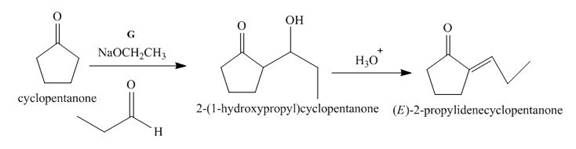
Figure 7
The given compound, cyclopentanone is treated with the base, sodium ethoxide that results in the formation of an enolate ion. Then, the enolate ion reacts with the compound, propanal to form the compound,
The lettered reagent H is shown as,
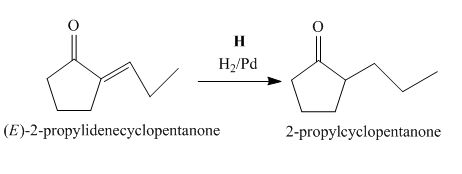
Figure 8
The compound,
The lettered reagent I is shown as,
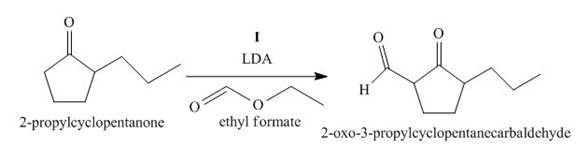
Figure 9
The compound,
The lettered reagent J is shown as,
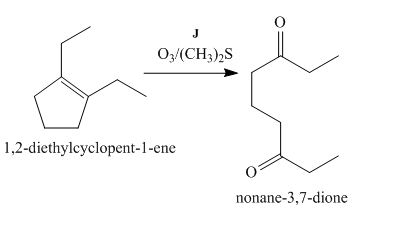
Figure 10
The compound,
The lettered reagent K is shown as,
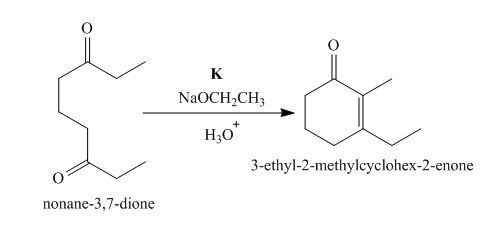
Figure 11
The intramolecular aldol reaction takes place in the compound,
The complete filled reagents that are needed for the given reactions are shown as,
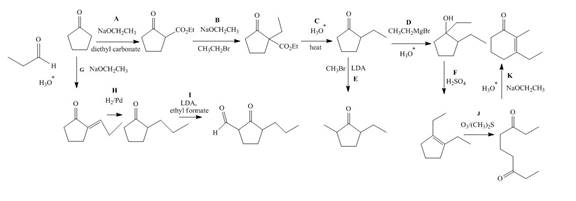
Figure 12
The lettered reagents that are needed for the given reactions are shown in Figure 12.
Want to see more full solutions like this?
Chapter 24 Solutions
ORGANIC CHEMISTRY
- What is the name of the following compound? SiMe3arrow_forwardK Draw the starting structure that would lead to the major product shown under the provided conditions. Drawing 1. NaNH2 2. PhCH2Br 4 57°F Sunny Q Searcharrow_forward7 Draw the starting alkyl bromide that would produce this alkyne under these conditions. F Drawing 1. NaNH2, A 2. H3O+ £ 4 Temps to rise Tomorrow Q Search H2arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

