
Concept explainers
(1)
Interpretation:
The most acidic hydrogens in the given compounds are to be indicated.
Concept introduction:
The substances that produce hydrogen ions (
According to the definition of Bronsted and Lowry, acids are the substances that donate protons whereas bases are the substances that accept protons.
(1)
Answer to Problem 22.60SP
The most acidic hydrogens in the given compounds are shown in Figure 1.
Explanation of Solution
In a molecule, different types of protons are present. However, when acting as acids, only the most acidic proton participates in the acid-base reaction. The protons of
Acidic hydrogen is defined as that hydrogen which is attached to more electronegative atoms like oxygen and fluorine. The compound (f) does not contain any acidic hydrogen atom in the given compounds.
The most acidic hydrogens in the given compounds are shown below.
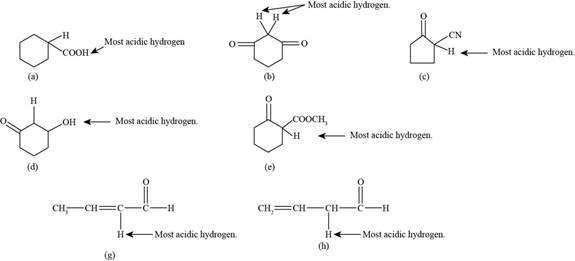
Figure 1
(2)
Interpretation:
The structures of important resonance contributors of the anions of the given compounds are to be drawn.
Concept introduction:
The delocalization of electrons due to presence of lone pair and double bond is called resonating structure. The structure is more stable if the resonating structures are more.
(2)
Answer to Problem 22.60SP
The structures of important resonance contributors of the anions of the given compounds are shown below.
Explanation of Solution
The formation of carbanion occurs when acidic hydrogen is removed from a compound. The important resonance contributors of the anions that results from the removal of most acidic hydrogen are shown below.
(a)
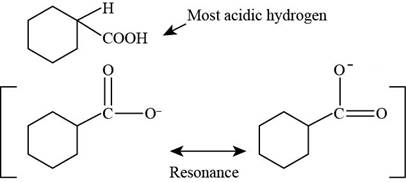
Figure 2
(b)
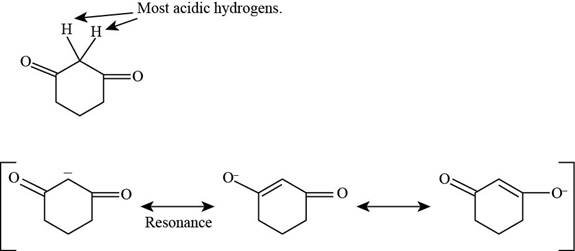
Figure 3
(c)
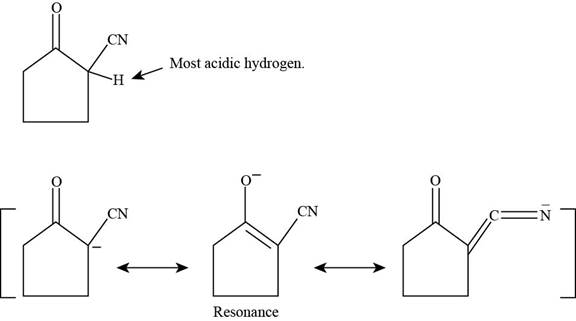
Figure 4
(d)
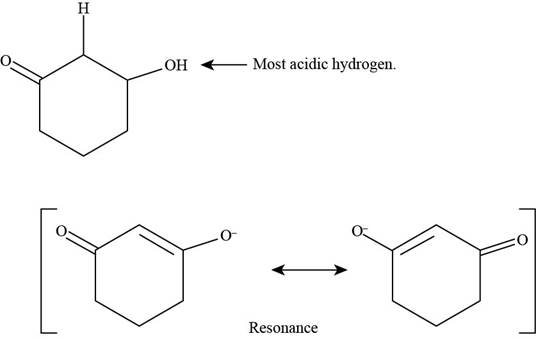
Figure 5
(e)
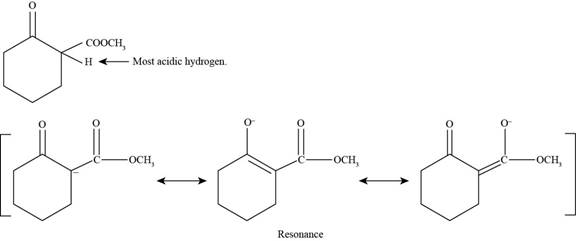
Figure 6
(f)
The given compound is shown below.
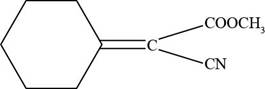
Figure 7
The given compound does not contain any acidic hydrogen atom. Therefore, it does not show resonance structures.
(g)
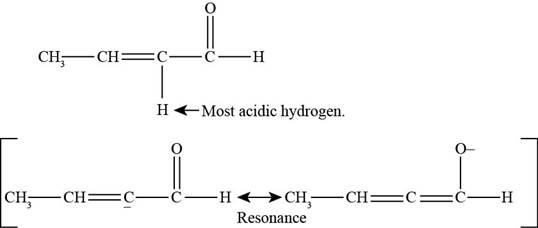
Figure 8
(h)
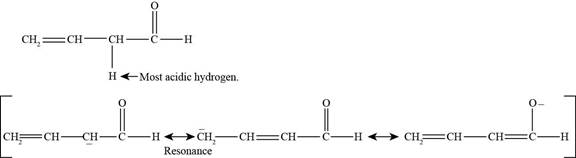
Figure 9
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry (9th Edition)
- What is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forwardIn the two chair conformations of glucose, the most stable is the one with all the OH groups in the equatorial position. Is this correct?arrow_forward
- please help me with my homeworkarrow_forwardhelparrow_forwardThe temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forward
- QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
