
Diagram how soap cleans our hands are to be explained.
Answer to Problem 24R
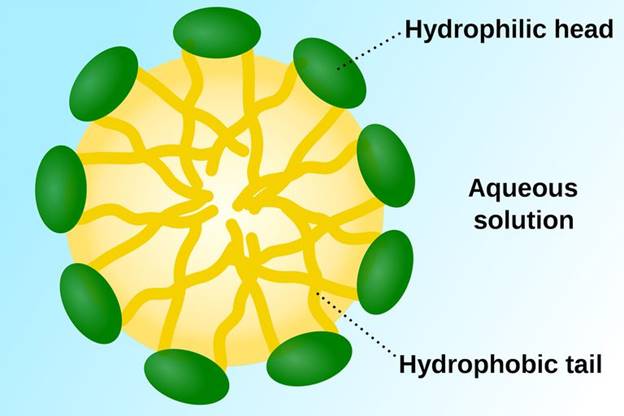
Explanation of Solution
Soap is an excellent cleanser. Soap has an ability to remove oil, which naturally does not mix with skin. Organic part of soap is polar. Its water loving part (hydrophilic) connects with water molecules with ion dipole interaction and hydrogen bonding. Water fearing (hydrophobic) part of soap is nonpolar hydrocarbon chain, connects with each other with dispersion force. Forming structures called micelles. In this carboxylate groups which are negatively charged sphere is present. The micelles ofsoap repel each other and remain dispersed in water.
Conclusion:
Soaps are excellent cleansers, ability to act as an emulsifying agent.
Chapter 21 Solutions
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Additional Science Textbook Solutions
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
College Physics: A Strategic Approach (3rd Edition)
Cosmic Perspective Fundamentals
Microbiology: An Introduction
Microbiology: An Introduction
Organic Chemistry (8th Edition)
- I need correct answer not chatgptarrow_forwardWhat is the resistance (in (2) of a 27.5 m long piece of 17 gauge copper wire having a 1.150 mm diameter? 0.445 ΧΩarrow_forwardFind the ratio of the diameter of silver to iron wire, if they have the same resistance per unit length (as they might in household wiring). d. Ag dFe = 2.47 ×arrow_forward
- Find the ratio of the diameter of silver to iron wire, if they have the same resistance per unit length (as they might in household wiring). d Ag = 2.51 dFe ×arrow_forwardShow that the units 1 v2/Q = 1 W, as implied by the equation P = V²/R. Starting with the equation P = V²/R, we can get an expression for a watt in terms of voltage and resistance. The units for voltage, V, are equivalent to [? v2 v2 A, are equivalent to J/C ✓ X . Therefore, 1 = 1 = 1 A V1 J/s Ω V-A X = 1 W. . The units for resistance, Q, are equivalent to ? The units for current,arrow_forwardPlease solve and answer the question correctly please. Thank you!!arrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





