
Concept explainers
a)
Interpretation:
The product obtained when D-galactose reacts with nitric acid is to be stated.
Concept Introduction:
D-galactose is a monosaccharide molecule and it is sweet in taste as glucose. It is C-
Epimers are pair of stereoisoisomers which differ in configuration at one stereogenic center. In D-galactose molecule one terminal
Nitric acid has the chemical formula
a)
Answer to Problem 31P
The product obtained when D-galactose reacts with nitric acid is given below,
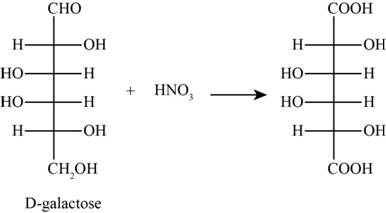
Figure 1
Explanation of Solution
The product obtained when D-galactose reacts with nitric acid is as follows,

Figure 1
b)
Interpretation:
The product obtained when D-galactose reacts with
Concept Introduction:
D-galactose is a monosaccharide molecule and it is sweet in taste as glucose. It is C-
Epimers are pair of stereoisoisomers which differ in configuration at one stereogenic center. In D-galactose molecule one terminal aldehydic and one terminal alcoholic group is present. The given ions
b)
Answer to Problem 31P
The product obtained when D-galactose reacts with
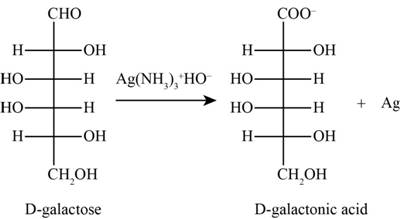
Figure 2
Explanation of Solution
The product obtained when D-galactose reacts with

Figure 2
The given ions
c)
Interpretation:
The product obtained when D-galactose reacts with
Concept Introduction:
D-galactose is a monosaccharide molecule and it is sweet in taste as glucose. It is C-
Epimers are pair of stereoisoisomers which differ in configuration at one stereogenic center. In D-galactose molecule one terminal aldehydic and one terminal alcoholic group is present.
c)
Answer to Problem 31P
The product obtained when D-galactose reacts with
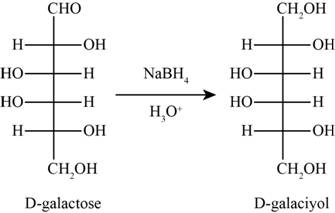
Figure 3
Explanation of Solution
The product obtained when D-galactose reacts with

Figure 3
The product obtained when D-galactose reacts with
d)
Interpretation:
The product obtained when D-galactose reacts with excess
Concept Introduction:
D-galactose is a monosaccharide molecule and it is sweet in taste as glucose. It is C-
d)
Answer to Problem 31P
The product obtained when D-galactose reacts with excess
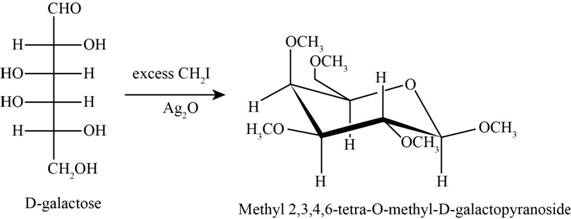
Figure 4
Explanation of Solution
The product obtained when D-galactose reacts with excess

Figure 4
e)
Interpretation:
The product obtained when D-galactose reacts with
Concept Introduction:
D-galactose is a monosaccharide molecule and it is sweet in taste as glucose. It is C-
e)
Answer to Problem 31P
The product obtained when D-galactose reacts with
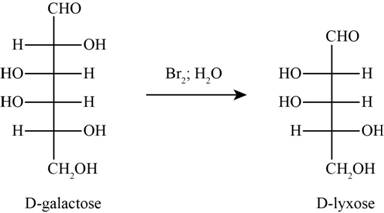
Figure 5
Explanation of Solution
The product obtained when D-galactose reacts with

Figure 5
f)
Interpretation:
The product obtained when D-galactose reacts with ethanol
Concept Introduction:
D-galactose is a monosaccharide molecule and it is sweet in taste as glucose. It is C-
f)
Answer to Problem 31P
The product obtained when D-galactose reacts with ethanol
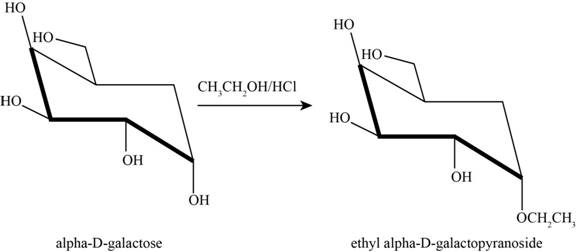
Figure 6
Explanation of Solution
The product obtained when D-galactose reacts with ethanol

Figure 6
g)
Interpretation:
The products obtained when D-galactose reacts with the given reactants are to be stated.
Concept Introduction:
D-galactose is a monosaccharide molecule and it is sweet in taste as glucose. It is C-
g)
Answer to Problem 31P
The products obtained when D-galactose reacts with the given reactants are shown in figure 7.
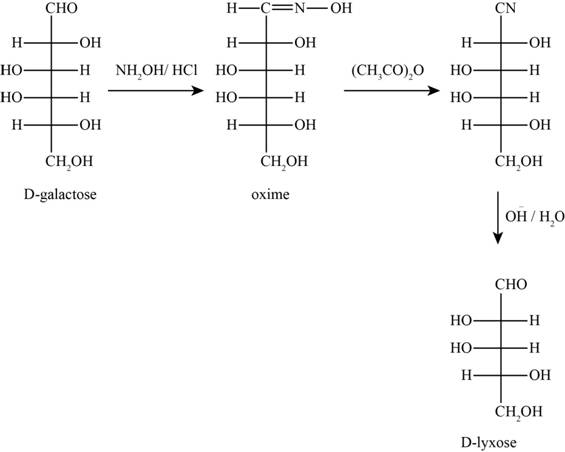
Figure 7
Explanation of Solution
The given reactants are hydroxylamine/trace acid, acetic anhydride/heat and
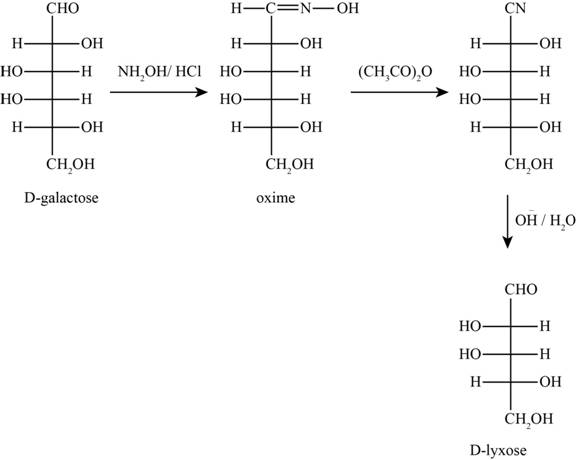
Figure 7
In the above reaction D-galactose first reacts with hydroxylamine in the presence of an acid, a compound of oxime is formed. Then it reacts with acetic anhydride with heat gives nitrile compound. Now, nitrile compound undergoes a reaction with base a compound D-Lyxose is formed.
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Draw the monomers required to synthesize this condensation polymer.arrow_forwardDraw the monomers required to synthesize this condensation polymer.arrow_forward8:44 PM Sun Apr 13 Earn Freecash.com O Measurement and Matter =1 Setting up a unit conversion 110 Eddie says... ✰ www-awu.aleks.com A student sets up the following equation to convert a measurement. (The ? stands for a number the student is going to calculate.) Fill in the missing part of this equation. Note: your answer should be in the form of one or more fractions multiplied together. (- 4 J kJ -7.0 × 10 ☐ = ? mmol.°C mol °C x10 μ Explanation Check □·□ torox.io Grey Hill LLC. All Rightsarrow_forward
- Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer formed in this reaction. Assume there are hydrogen atoms there are hydrogen atoms on the two ends of the trimer. Ignore inorganic byproducts please.arrow_forwardi need help with the folarrow_forwardPLEASE HELP NOW! URGENT!arrow_forward
- a. Determine whether each of the Followery Molecules is in the R- On the y- Configuration 1-01"/ 1-6-4 Br 4 I el Br b. Draw The Fisher projection For all the Meso compounds that can exist FOR The Following molenlearrow_forward1- Refer to the monosaccharides below to answer each of the following question(s): CH₂OH CHO CH₂OH CH₂OH 0 H- OH 0 0 HO- H H- -OH HO H HO H H OH HO- H CH₂OH H. OH HO H HO- H CH₂OH CH₂OH CH3 a. Sorbose b. Rhamnose c. Erythrulose d. Xylulose Classify each sugar by type; for example, glucose is an aldohexose. a. Xylulose is .. b. Erythrulose is . c. Sorbose is .. d. Rhamnose is .. 2- Consider the reaction below to answer the following question(s). CHO H OH CH₂OH CH₂OH HO- H HO HO + H. -OH HO OH HO. H OH OH H -OH H OH CH₂OH Q Z a. Refer to Exhibit 25-11. Place a triangle around the anomeric carbon in compound Q. Compound Z is: b. 1. the D-anomer. 2. the a-anomer. 3. the ẞ-anomer. 4. the L-anomer. c. Which anomer is the LEAST stable? d. Q and Z are cyclic examples of: a. acetals b. hemiacetals c. alditols d. hemialditolsarrow_forwardi need help identifying the four carbon oxygen bonds in the following:arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,



