
(a)
Interpretation:
The product formed when butyric acid reacts with ethanol in the presence of
Concept introduction:
Esterification occurs when a
Answer to Problem 20.27AP
The product formed when butyric acid reacts with ethanol in the presence of
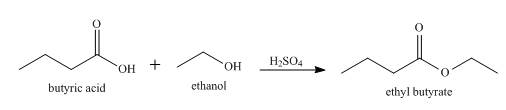
Explanation of Solution
When butyric acid reacts with ethanol in the presence of

Figure 1
When butyric acid reacts with ethanol in presence of
(b)
Interpretation:
The product formed when butyric acid reacts with aqueous
Concept introduction:
An acid-base reaction occurs through a transfer of proton from an acid to a base. The base gets protonated and the acid gets deprotonated. The reaction results in the formation of a conjugate acid and a conjugate base as the products. The type of products formed affects the direction of reaction at equilibrium.
Answer to Problem 20.27AP
The product formed when butyric acid reacts with aqueous
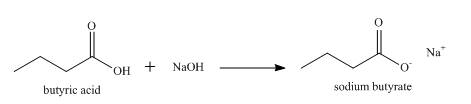
Explanation of Solution
When a carboxylic acid reacts with a base, the hydrogen atom is abstracted by the base and a salt of carboxylic acid is formed.
So, when butyric acid reacts with aqueous

Figure 2
The product formed when butyric acid reacts with aqueous
(c)
Interpretation:
The product formed when butyric acid reacts with
Concept introduction:
The substances which on addition removes oxygen atom or hydrogen atom from the other substance, that is, reduces the other substances are known as reducing agents. Reducing agents themselves get oxidized. Carboxylic acid is reduced when reacts with reducing agent,
Answer to Problem 20.27AP
The product formed when butyric acid reacts with
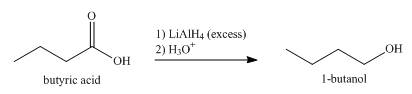
Explanation of Solution
Carboxylic acid is reduced when reacts with reducing agent,
When butyric acid reacts with excess of

Figure 3
When butyric acid reacts with excess of
(d)
Interpretation:
The product formed when butyric acid is heated is to be stated.
Concept introduction:
Carboxylic acids such as
Answer to Problem 20.27AP
When butyric acid is heated, no product is formed.
Explanation of Solution
The structure of butyric acid does not have any electron-withdrawing group like
Butyric acid on heating gives no reaction.
(e)
Interpretation:
The product formed when butyric acid reacts with
Concept introduction:
Carboxylic acids react with
Answer to Problem 20.27AP
The product formed when butyric acid reacts with
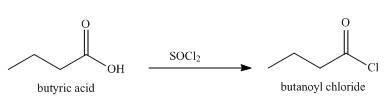
Explanation of Solution
When butyric acid reacts with

Figure 4
The product formed when butyric acid reacts with
(f)
Interpretation:
The product formed when butyric acid reacts with diazomethane in ether is to be stated.
Concept introduction:
Esterification occurs when carboxylic acid reacts with diazomethane in ether. Esters end with a suffix,
Answer to Problem 20.27AP
The product formed when butyric acid reacts with diazomethane in ether is shown below.
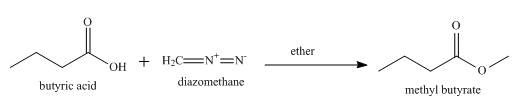
Explanation of Solution
Esterification occurs when butyric acid reacts with diazomethane in ether and methyl butyrate is formed as a product along with the release of nitrogen gas. The complete reaction is shown below.

Figure 5
The product formed when butyric acid reacts with diazomethane in ether is methyl butyrate.
(g)
Interpretation:
The product formed when
Concept introduction:
Acetal is formed when an alcohol in excess reacts with an aldehyde or ketone in acidic medium. The
Answer to Problem 20.27AP
The product formed when
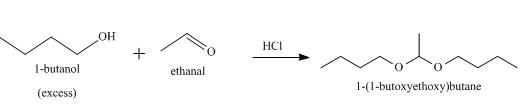
Explanation of Solution
When an alcohol reacts with an aldehyde in presence of an acid catalyst, a hemiacetal is formed as an intermediate which further reacts to produce acetal with the release of water. The acidic catalyst is used because the alcohol is a weak nucleophile. So on addition of an acid, the oxygen of aldehyde gets protonated and a carbocation is formed. The alcohol easily attacks the carbocation to form the germinal diether derivative of aldehyde, that is, an acetal. When

Figure 6
When
(h)
Interpretation:
The product formed when butanoyl chloride reacts with benzene in
Concept introduction:
Friedel crafts acylation reaction is a reaction in which an
Answer to Problem 20.27AP
The product formed when butanoyl chloride reacts with benzene in
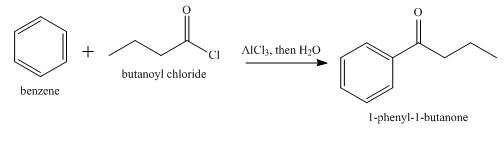
Explanation of Solution
Benzene undergoes Friedel Crafts acylation reaction on reaction with butanoyl chloride in presence of

Figure 7
The product formed when butanoyl chloride reacts with benzene in
(i)
Interpretation:
The product formed when
Concept introduction:
Wolff Kishner Reduction is a reaction in which aldehydes and ketones are converted into
Answer to Problem 20.27AP
The product formed when
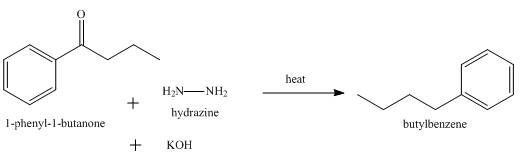
Explanation of Solution
Wolff Kishner reduction takes place when

Figure 8
The product formed when
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry, Ebook And Single-course Homework Access
- Consider this step in a radical reaction: Y What type of step is this? Check all that apply. Draw the products of the step on the right-hand side of the drawing area below. If more than one set of products is possible, draw any set. Also, draw the mechanism arrows on the left-hand side of the drawing area to show how this happens. ionization propagation initialization passivation none of the abovearrow_forward22.16 The following groups are ortho-para directors. (a) -C=CH₂ H (d) -Br (b) -NH2 (c) -OCHS Draw a contributing structure for the resonance-stabilized cation formed during elec- trophilic aromatic substitution that shows the role of each group in stabilizing the intermediate by further delocalizing its positive charge. 22.17 Predict the major product or products from treatment of each compound with Cl₁/FeCl₂- OH (b) NO2 CHO 22.18 How do you account for the fact that phenyl acetate is less reactive toward electro- philic aromatic substitution than anisole? Phenyl acetate Anisole CH (d)arrow_forwardShow how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid.arrow_forward
- Help me solve this problem. Thank you in advance.arrow_forward22.7 Predict the monoalkylated products of the following reactions with benzene. (a) AlCl3 Ya (b) AlCl3 (c) H3PO4 (d) 22.8 Think-Pair-Share AICI3 The reaction below is a common electrophilic aromatic substitution. SO3 H₂SO4 SO₂H (a) Draw the reaction mechanism for this reaction using HSO,+ as the electrophile. (b) Sketch the reaction coordinate diagram, where the product is lower in energy than the starting reactant. (c) Which step in the reaction mechanism is highest in energy? Explain. (d) Which of the following reaction conditions could be used in an electrophilic aro- matic substitution with benzene to provide substituted phenyl derivatives? (i) AICI3 HNO3 H₂SO4 K2Cr2O7 (iii) H₂SO4 (iv) H₂PO₁arrow_forwardIs an acid-base reaction the only type of reaction that would cause leavening products to rise?arrow_forward
- Help me understand this! Thank you in advance.arrow_forward22.22 For each compound, indicate which group on the ring is more strongly activating and then draw a structural formula of the major product formed by nitration of the compound. Br CHO (a) CH3 (b) (c) CHO CH3 SO₂H (d) ☑ OCHS NO₂ (e) (f) CO₂H NHCOCH3 NHCOCH, (h) CHS 22.23 The following molecules each contain two aromatic rings. (b) 000-100- H3C (a) (c) Which ring in each undergoes electrophilic aromatic substitution more readily? Draw the major product formed on nitration.arrow_forwardV Consider this step in a radical reaction: Br: ? What type of step is this? Check all that apply. Draw the products of the step on the right-hand side of the drawing area below. If more than one set of products is possible, draw any set. Also, draw the mechanism arrows on the left-hand side of the drawing area to show how this happens. ⚫ionization termination initialization neutralization none of the abc Explanation Check 80 Ο F3 F1 F2 2 F4 01 % do5 $ 94 #3 X 5 C MacBook Air 25 F5 F6 66 ©2025 ˇ F7 29 & 7 8arrow_forward
- Show how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid.arrow_forwardno aiarrow_forwardPolymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer formed in this reaction. Assume there are hydrogen atoms there are hydrogen atoms on the two ends of the trimer. Ignore inorganic byproducts.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
