
Concept explainers
(i)
Interpretation: The Bronsted acid or base present in the below reaction should be identified.
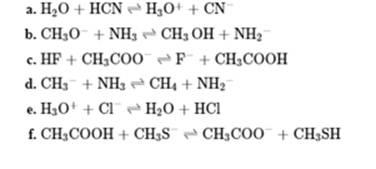
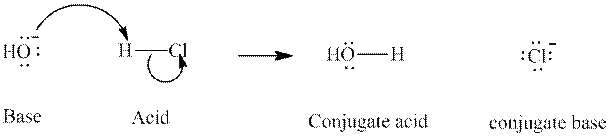
Concept introduction: In accordance with Bronsted definition an acid act as proton donor and a base can act as proton acceptor. Thus in a typical acid-base reaction, the fundamental principle is a lone pair of base reaches out for an acidic proton. Similarly, curved arrows are used for departing conjugate base. After deprotonation, the species left with negative charge is refers as conjugate base of acid while the other with positive charge is termed conjugate acid of given base. For example,
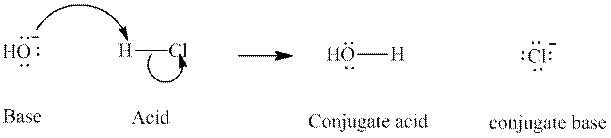
The strength of various conjugate acid-base pairs varies inversely to one another; the strong acid has a weak conjugate base and the strong base has weak conjugate acid and vice-versa.
(ii)
Interpretation: Whether the equilibrium is favored to left or right should be identified in below reactions.
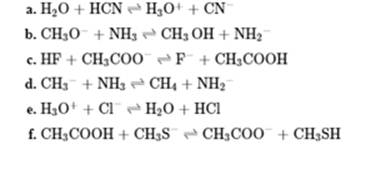
Concept introduction: In accordance with Bronsted definition an acid act as proton donor and a base can act as proton acceptor. Thus in a typical acid-base reaction, the fundamental principle is a lone pair of base reaches out for an acidic proton. Similarly, curved arrows are used for departing conjugate base. After deprotonation, the species left with negative charge is refers as conjugate base of acid while the other with positive charge is termed conjugate acid of given base. For example,
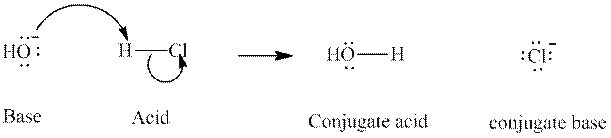
The strength of various conjugate acid-base pairs varies inversely to one another; the strong acid has a weak conjugate base and the strong base has weak conjugate acid and vice-versa.
Smaller the magnitude of
(iii)
Interpretation: The value of
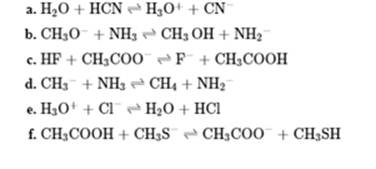
Concept introduction: In accordance with Bronsted definition an acid act as proton donor and a base can act as proton acceptor. Thus in a typical acid-base reaction, the fundamental principle is a lone pair of base reaches out for an acidic proton. Similarly, curved arrows are used for departing conjugate base. After deprotonation, the species left with negative charge is refers as conjugate base of acid while the other with positive charge is termed conjugate acid of given base. For example,
The strength of various conjugate acid-base pairs varies inversely to one another; the strong acid has a weak conjugate base and strong base has weak conjugate acid and vice-versa.
Smaller the magnitude of
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic Chemistry: Structure and Function
- 3. Use Kapustinskii's equation and data from Table 4.10 in your textbook to calculate lattice energies of Cu(OH)2 and CuCO3 (4 points)arrow_forward2. Copper (II) oxide crystalizes in monoclinic unit cell (included below; blue spheres 2+ represent Cu²+, red - O²-). Use Kapustinski's equation (4.5) to calculate lattice energy for CuO. You will need some data from Resource section of your textbook (p.901). (4 points) CuOarrow_forwardWhat is the IUPAC name of the following compound? OH (2S, 4R)-4-chloropentan-2-ol O (2R, 4R)-4-chloropentan-2-ol O (2R, 4S)-4-chloropentan-2-ol O(2S, 4S)-4-chloropentan-2-olarrow_forward
- Use the reaction coordinate diagram to answer the below questions. Type your answers into the answer box for each question. (Watch your spelling) Energy A B C D Reaction coordinate E A) Is the reaction step going from D to F endothermic or exothermic? A F G B) Does point D represent a reactant, product, intermediate or transition state? A/ C) Which step (step 1 or step 2) is the rate determining step? Aarrow_forward1. Using radii from Resource section 1 (p.901) and Born-Lande equation, calculate the lattice energy for PbS, which crystallizes in the NaCl structure. Then, use the Born-Haber cycle to obtain the value of lattice energy for PbS. You will need the following data following data: AH Pb(g) = 196 kJ/mol; AHƒ PbS = −98 kJ/mol; electron affinities for S(g)→S¯(g) is -201 kJ/mol; S¯(g) (g) is 640kJ/mol. Ionization energies for Pb are listed in Resource section 2, p.903. Remember that enthalpies of formation are calculated beginning with the elements in their standard states (S8 for sulfur). The formation of S2, AHF: S2 (g) = 535 kJ/mol. Compare the two values, and explain the difference. (8 points)arrow_forwardIn the answer box, type the number of maximum stereoisomers possible for the following compound. A H H COH OH = H C Br H.C OH CHarrow_forward
- 7. Magnesium is found in nature in the form of carbonates and sulfates. One of the major natural sources of zinc is zinc blende (ZnS). Use relevant concepts of acid-base theory to explain this combination of cations and anions in these minerals. (2 points)arrow_forward6. AlF3 is insoluble in liquid HF but dissolves if NaF is present. When BF3 is added to the solution, AlF3 precipitates. Write out chemical processes and explain them using the principles of Lewis acid-base theory. (6 points)arrow_forward5. Zinc oxide is amphoteric. Write out chemical reactions for dissolution of ZnO in HCl(aq) and in NaOH(aq). (3 points)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





