
Organic Chemistry - With Access (Custom)
9th Edition
ISBN: 9781337031745
Author: McMurry
Publisher: Cengage
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19.13, Problem 22P
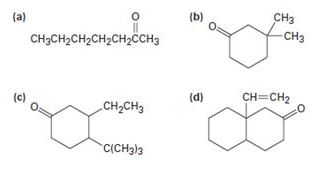
How might conjugate addition reactions of lithium diorganocopper reagents be used to synthesize the following compounds?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Given 10.0 g of NaOH, what volume of a 0.100 M solution of H2SO4 would be required to exactly react all the NaOH?
3.50 g of Li are combined with 3.50 g of N2. What is the maximum mass of Li3N that can be produced?
6 Li + N2 ---> 2 Li3N
3.50 g of Li are combined with 3.50 g of N2. What is the maximum mass of Li3N that can be produced?
6 Li + N2 ---> 2 Li3N
Chapter 19 Solutions
Organic Chemistry - With Access (Custom)
Ch. 19.1 - Prob. 1PCh. 19.1 - Draw structures corresponding to the following...Ch. 19.2 - Prob. 3PCh. 19.2 - How would you carry out the following reactions?...Ch. 19.4 - Treatment of an aldehyde or ketone with cyanide...Ch. 19.4 - p-Nitrobenzaldehyde is more reactive toward...Ch. 19.5 - Prob. 7PCh. 19.5 - The oxygen in water is primarily (99.8) 16O, but...Ch. 19.6 - Prob. 9PCh. 19.8 - Show the products you would obtain by...
Ch. 19.8 - Prob. 11PCh. 19.8 - Prob. 12PCh. 19.9 - Prob. 13PCh. 19.10 - Prob. 14PCh. 19.10 - Prob. 15PCh. 19.11 - What carbonyl compound and what phosphorus ylide...Ch. 19.11 - -Carotene, a yellow food-coloring agent and...Ch. 19.12 - Prob. 18PCh. 19.12 - Prob. 19PCh. 19.13 - Prob. 20PCh. 19.13 - Treatment of 2-cyclohexenone with HCN/KCN yields a...Ch. 19.13 - How might conjugate addition reactions of lithium...Ch. 19.14 - How might you use IR spectroscopy to determine...Ch. 19.14 - Prob. 24PCh. 19.14 - Prob. 25PCh. 19.14 - Prob. 26PCh. 19.SE - Each of the following substances can be prepared...Ch. 19.SE - Prob. 28VCCh. 19.SE - Prob. 29VCCh. 19.SE - Prob. 30MPCh. 19.SE - Prob. 31MPCh. 19.SE - Prob. 32MPCh. 19.SE - Prob. 33MPCh. 19.SE - Prob. 34MPCh. 19.SE - Prob. 35MPCh. 19.SE - It is not uncommon for organic chemists to prepare...Ch. 19.SE - Prob. 37MPCh. 19.SE - Prob. 38MPCh. 19.SE - Prob. 39MPCh. 19.SE - Prob. 40MPCh. 19.SE - Aldehydes and ketones react with thiols to yield...Ch. 19.SE - Prob. 42MPCh. 19.SE - When cyclohexanone is heated in the presence of a...Ch. 19.SE - Prob. 44MPCh. 19.SE - The Meerwein-Ponndorf-Verley reaction involves...Ch. 19.SE - Propose a mechanism to account for the formation...Ch. 19.SE - Prob. 47MPCh. 19.SE - Prob. 48MPCh. 19.SE - Treatment of an , -unsaturated ketone with basic...Ch. 19.SE - Prob. 50MPCh. 19.SE - Prob. 51MPCh. 19.SE - Prob. 52MPCh. 19.SE - Prob. 53MPCh. 19.SE - Prob. 54APCh. 19.SE - Draw and name the seven aldehydes and ketones with...Ch. 19.SE - Give IUPAC names for the following compounds:Ch. 19.SE - Draw structures of compounds that fit the...Ch. 19.SE - Predict the products of the reaction of (1)...Ch. 19.SE - Show how you might use a Wittig reaction to...Ch. 19.SE - How would you use a Grignard reaction on an...Ch. 19.SE - Prob. 61APCh. 19.SE - Prob. 62APCh. 19.SE - How would you synthesize the following substances...Ch. 19.SE - Carvone is the major constituent of spearmint oil....Ch. 19.SE - How would you synthesize the following compounds...Ch. 19.SE - At what position would you expect to observe IR...Ch. 19.SE - Acidcatalyzed dehydration of...Ch. 19.SE - Choose the structure that best fits the IR...Ch. 19.SE - Propose structures for molecules that meet the...Ch. 19.SE - Prob. 70APCh. 19.SE - Prob. 71APCh. 19.SE - When 4hydroxybutanal is treated with methanol in...Ch. 19.SE - Prob. 73APCh. 19.SE - Prob. 74APCh. 19.SE - Prob. 75APCh. 19.SE - Prob. 76APCh. 19.SE - Prob. 77APCh. 19.SE - Tamoxifen is a drug used in the treatment of...Ch. 19.SE - Compound A, MW 86, shows an IR absorption at 1730...Ch. 19.SE - Compound B is isomeric with A (Problem 19-79) and...Ch. 19.SE - The 1HNMR spectrum shown is that of a compound...Ch. 19.SE - Prob. 82APCh. 19.SE - Propose structures for ketones or aldehydes that...Ch. 19.SE - Prob. 84APCh. 19.SE - Prob. 85APCh. 19.SE - The proton and carbon NMR spectra for each of...Ch. 19.SE - The proton NMR spectrum for a compound with...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Concentration Trial1 Concentration of iodide solution (mA) 255.8 Concentration of thiosulfate solution (mM) 47.0 Concentration of hydrogen peroxide solution (mM) 110.1 Temperature of iodide solution ('C) 25.0 Volume of iodide solution (1) used (mL) 10.0 Volume of thiosulfate solution (5:03) used (mL) Volume of DI water used (mL) Volume of hydrogen peroxide solution (H₂O₂) used (mL) 1.0 2.5 7.5 Time (s) 16.9 Dark blue Observations Initial concentration of iodide in reaction (mA) Initial concentration of thiosulfate in reaction (mA) Initial concentration of hydrogen peroxide in reaction (mA) Initial Rate (mA's)arrow_forwardDraw the condensed or line-angle structure for an alkene with the formula C5H10. Note: Avoid selecting cis-/trans- isomers in this exercise. Draw two additional condensed or line-angle structures for alkenes with the formula C5H10. Record the name of the isomers in Data Table 1. Repeat steps for 2 cyclic isomers of C5H10arrow_forwardExplain why the following names of the structures are incorrect. CH2CH3 CH3-C=CH-CH2-CH3 a. 2-ethyl-2-pentene CH3 | CH3-CH-CH2-CH=CH2 b. 2-methyl-4-pentenearrow_forward
- Draw the line-angle formula of cis-2,3-dichloro-2-pentene. Then, draw the line-angle formula of trans-2,3-dichloro-2-pentene below. Draw the dash-wedge formula of cis-1,3-dimethylcyclohexane. Then, draw the dash-wedge formula of trans-1,3-dimethylcyclohexane below.arrow_forwardRecord the amounts measured and calculate the percent yield for Part 2 in the table below. Dicyclopentadiene measured in volume Cyclopentadiene measured in grams 0 Measured Calculated Mol Yield Mass (g) or Volume (mL) Mass (g) or Volume (ml) 0.6 2.955 Part 2 Measurements and Results Record the amounts measured and calculate the percent yield for Part 2 in the table below. 0.588 0.0044 2.868 0.0434 N/A Table view List view Measured Calculated Mol $ Yield Melting Point (C) Mass (g) or Volume (ml) Mass (g) or Volume (ml.) Cyclopentadiene 0.1 0.08 0.001189 measured in volume Maleic Anhydride 0.196 N/A cis-norbornene-5,6-endo- dicarboxylic anhydride 0.041 0.0002467 N/A N/A N/A 0.002 N/A N/A 128arrow_forwardDraw the condensed structural formula and line-angle formula for each: 2,3-dimethylheptane 3-bromo-2-pentanol 3-isopropyl-2-hexene 4-chlorobutanoic acidarrow_forward
- Record the IUPAC names for each of the structures shown below. a) b) c) OH d) OH e)arrow_forwardA solution of 14 g of a nonvolatile, nonelectrolyte compound in 0.10 kg of benzene boils at 81.7°C. If the BP of pure benzene is 80.2°C and the K, of benzene is 2.53°C/m, calculate the molar mass of the unknown compound. AT₁ = Km (14)arrow_forwardPlease help me answer the following questions. My answers weren't good enough. Need to know whyy the following chemicals were not used in this experiment related to the melting points and kf values. For lab notebook not a graded assignments.arrow_forward
- Draw the arrow pushing reaction mechanism. DO NOT ANSWER IF YOU WONT DRAW IT. Do not use chat gpt.arrow_forwardComplete the following esterification reaction by drawing the structural formula of the product formed. HOH HO i catalyst catalyst OH HO (product has rum flavor) (product has orange flavor)arrow_forwardThe statements in the tables below are about two different chemical equilibria. The symbols have their usual meaning, for example AG stands for the standard Gibbs free energy of reaction and K stands for the equilibrium constant. In each table, there may be one statement that is faise because it contradicts the other three statements. If you find a false statement, check the box next to t Otherwise, check the "no false statements" box under the table. statement false? AG"1 no false statements: statement false? AG-0 0 InK-0 0 K-1 0 AH-TAS no false statements 2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License