
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
4th Edition
ISBN: 9781265982959
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 54P
Draw the products of each acid-base reaction.
a.
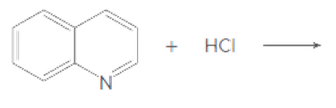
b.
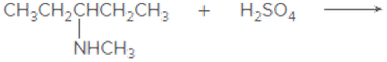
c.
d.
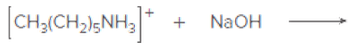
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
please solve this, and help me know which boxes to check. Thank you so much in advance.
Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Describe how electronegativity is illustrated on the periodic table including trends between groups and periods and significance of atom size.
Define the term “transition.” How does this definition apply to the transition metals?
Chapter 18 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
Ch. 18.1 - Prob. 18.1PPCh. 18.1 - Prob. 18.1PCh. 18.1 - Prob. 18.2PCh. 18.1 - Prob. 18.3PCh. 18.2 - Prob. 18.2PPCh. 18.2 - Prob. 18.4PCh. 18.2 - Prob. 18.5PCh. 18.3 - Prob. 18.3PPCh. 18.4 - Decaffeinated coffee is produced by extracting the...Ch. 18.4 - Prob. 18.7P
Ch. 18.5 - Prob. 18.8PCh. 18.5 - Naloxone is a drug used to treat overdoses of...Ch. 18.6 - Prob. 18.4PPCh. 18.6 - Prob. 18.10PCh. 18.6 - Prob. 18.11PCh. 18.6 - Name each ammonium salt. a. ( CH3 NH3)+Cl b. [( CH...Ch. 18.6 - Prob. 18.6PPCh. 18.6 - Prob. 18.12PCh. 18.7 - Prob. 18.13PCh. 18.7 - Prob. 18.14PCh. 18.8 - Prob. 18.15PCh. 18.8 - Prob. 18.16PCh. 18.8 - Prob. 18.17PCh. 18.8 - Prob. 18.18PCh. 18.9 - Prob. 18.19PCh. 18.9 - Prob. 18.20PCh. 18.9 - Prob. 18.21PCh. 18.10 - Prob. 18.22PCh. 18 - Prob. 23PCh. 18 - Prob. 24PCh. 18 - Prob. 25PCh. 18 - Prob. 26PCh. 18 - Prob. 27PCh. 18 - Prob. 28PCh. 18 - Prob. 29PCh. 18 - Prob. 30PCh. 18 - Prob. 31PCh. 18 - Prob. 32PCh. 18 - Give an acceptable name for each amine. a. b.Ch. 18 - Give an acceptable name for each amine. a. b.Ch. 18 - Give an acceptable name for each amine. a. b. c....Ch. 18 - Give an acceptable name for each amine. a. CH3(...Ch. 18 - Prob. 37PCh. 18 - Prob. 38PCh. 18 - Prob. 39PCh. 18 - Prob. 40PCh. 18 - Prob. 41PCh. 18 - Prob. 42PCh. 18 - Prob. 43PCh. 18 - Prob. 44PCh. 18 - Which compound in each pair has the higher boiling...Ch. 18 - Which compound in each pair has the higher boiling...Ch. 18 - Draw the hydrogen-bonding interactions that occur...Ch. 18 - Prob. 48PCh. 18 - Prob. 49PCh. 18 - Which compound has the higher water solubility:...Ch. 18 - Prob. 51PCh. 18 - Prob. 52PCh. 18 - Draw the products of each acid-base reaction. a....Ch. 18 - Draw the products of each acid-base reaction. a....Ch. 18 - Prob. 55PCh. 18 - Prob. 56PCh. 18 - What type of nitrogen heterocycle occurs in both...Ch. 18 - Only one of the N atoms in nicotine has a trigonal...Ch. 18 - Prob. 59PCh. 18 - Prob. 60PCh. 18 - Why are aqueous solutions of an alkaloid slightly...Ch. 18 - Prob. 62PCh. 18 - Prob. 63PCh. 18 - Explain why patients with Parkinson’s disease...Ch. 18 - Prob. 65PCh. 18 - Prob. 66PCh. 18 - Prob. 67PCh. 18 - Prob. 68PCh. 18 - Locate the atoms of 2-phenylethylamine in the...Ch. 18 - Locate the atoms of 2-phenylethylamine in the...Ch. 18 - Give an example of an antihistamine. Explain how...Ch. 18 - Give an example of an anti-ulcer drug, and explain...Ch. 18 - Prob. 73PCh. 18 - Prob. 74PCh. 18 - Prob. 75PCh. 18 - Prob. 76PCh. 18 - Prob. 77PCh. 18 - Prob. 78PCh. 18 - Prob. 79PCh. 18 - Why do some antihistamines cause drowsiness while...Ch. 18 - Prob. 81PCh. 18 - Prob. 82PCh. 18 - Compare the structures of morphine and heroin....Ch. 18 - Prob. 84CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Describe how the properties of the different types of elements (metals, nonmetals, metalloids) differ.arrow_forwardUse a textbook or other valid source to research the physical and chemical properties of each element listed in Data Table 1 using the following as a guideline: Ductile (able to be deformed without losing toughness) and malleable (able to be hammered or pressed permanently out of shape without breaking or cracking) or not ductile or malleable Good, semi, or poor conductors of electricity and heat High or low melting and boiling points Occur or do not occur uncombined/freely in nature High, intermediate, or low reactivity Loses or gains electrons during reactions or is not reactivearrow_forwardProvide the Physical and Chemical Properties of Elements of the following elements listedarrow_forward
- Questions 4 and 5arrow_forwardFor a titration of 40.00 mL of 0.0500 M oxalic acid H2C2O4 with 0.1000 M KOH, calculate the pH at each of the following volume of KOH used in the titration: 1) before the titration begin;2) 15 mL; 3) 20 mL; 4) 25 mL; 5) 40 mL; 6) 50 mL. Ka1 = 5.90×10^-2, Ka2 = 6.50×10^-5 for oxalic acid.arrow_forwardPredict the major organic product(s), if any, of the following reactions. Assume all reagents are in excess unless otherwise indicated.arrow_forward
- Predict the major organic product(s), if any, of the following reactions. Assume all reagents are in excess unless otherwise indicated.arrow_forwardHow many signals would you expect to find in the 1 H NMR spectrum of each given compound? Part 1 of 2 2 Part 2 of 2 HO 5 ☑ Х IIIIII***** §arrow_forwardA carbonyl compound has a molecular ion with a m/z of 86. The mass spectra of this compound also has a base peak with a m/z of 57. Draw the correct structure of this molecule. Drawingarrow_forward
- Can you draw this using Lewis dot structures and full structures in the same way they are so that I can better visualize them and then determine resonance?arrow_forwardSynthesize the following compound from cyclohexanol, ethanol, and any other needed reagentsarrow_forwardFor a titration of 20.00 mL of 0.0500 M H2SO4 with 0.100 M KOH, calculate the pH at each of the following volume of KOH used in the titration: 1) before the titration begin; 2) 10.00 mL; 3) 20.00 mL; 4) 30.00 mL. Ka2 = 1.20×10-2 for H2SO4.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY