
Concept explainers
(a)
Interpretation:
The amine needs to be labeled as 10, 20 or 30.
Concept introduction:
Answer to Problem 73P
A tertiary (3o) amine.
Explanation of Solution
Amines are derivatives which are derived from ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group. They can be called as alkylamines and arylamines.
Whether an amine is primary (1o), secondary (2o) or tertiary (3o) depends on the number of hydrogen atoms replaced by an alkyl or aryl group in ammonia. The amine is a primary amine if one hydrogen atom is replaced, it is a secondary amine if 2 hydrogen atoms are replaced and if three hydrogen atoms are replaced, it is known as a tertiary amine.
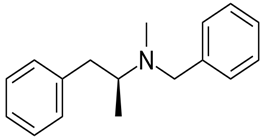
Here, all three hydrogen atoms are replaced and therefore this amine called a tertiary (3o) amine.
(b)
Interpretation:
The chiral center of the benzphetamine molecule needs to be labeled.
Concept Introduction:
The molecules with a chiral center can form a superimposable mirror image and is known as enantiomers. There should not be any plane of symmetry in a molecule to be chiral. A plane that bisects a molecule into two equal halves is known as a plane of symmetry. If there is a plane of symmetry in a molecule and it is identical to any of its mirror image, it is considered as achiral. The chiral center is carbon attached to 4 different groups attached to it.
Answer to Problem 73P
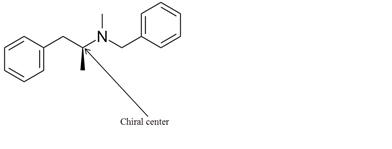
Explanation of Solution
The chiral center in the given molecule is shown as follows:
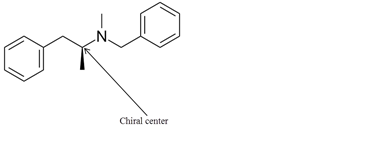
The labeled carbon atom is attached to 4 different groups.
The chiral center has four different groups attached to carbon and no plane of symmetry. The molecule with a chiral center shows optical isomerism. The chiral center is a stereocenter holding the atoms in space, the mirror image so formed are not superimposable.
(c)
Interpretation:
The enantiomers of benzphetamine molecule needs to be determined.
Concept introduction:
The chiral molecules which are mirror images of each other are known as enantiomers. They are non-superimposable mirror images, or they cannot be placed on top of each other.
Answer to Problem 73P
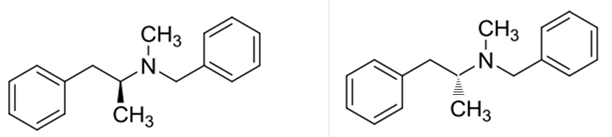
Explanation of Solution
Benzphetamine has two enantiomers due to its one chiral center. It rotates the plane-polarized light. Here, S-benzphetamine rotates the light anticlockwise and R- benzphetamine rotates it clockwise.
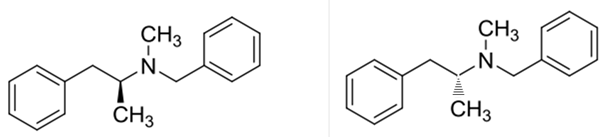
Benzphetaminecan can be called as a racemic mixture of two enantiomers, which are R(+) and S(-).
(d)
Interpretation:
The constitutional isomer that contains a primary amine needs to be drawn.
Concept introduction:
Constitutional isomers have the same molecular formula but different structural connectivity. The number of each atom in both the molecules needs to be counted and the arrangement is observed to check whether two molecules are a constitutional isomer of each other or not.
Answer to Problem 73P
Explanation of Solution
Whether an amine is primary (1o), secondary (2o) or tertiary (3o) depends on the number of hydrogen atoms replaced by an alkyl or aryl group in ammonia. The amine is a primary amine if one hydrogen atom is replaced, it is a secondary amine if 2 hydrogen atoms are replaced and if three hydrogen atoms are replaced it is known as a tertiary amine.
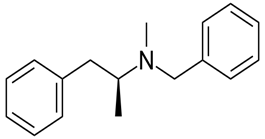
The given compound is as follows:
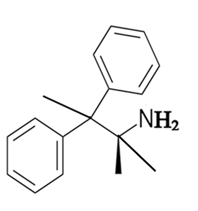
The constitutional isomer with a primary amine is as follows:
Here, only one hydrogen atom is replaced therefore this amine called a primary amine (1o).
(e)
Interpretation:
The constitutional isomer that contains a tertiary amine needs to be determined.
Concept introduction:
Constitutional isomers have the same molecular formula but different structural connectivity. The number of each atom in both the molecules needs to be counted and the arrangement is observed to check whether two molecules are a constitutional isomer of each other or not.
Answer to Problem 73P

Explanation of Solution
Whether an amine is primary (1o), secondary (2o) or tertiary (3o) depends on the number of hydrogen atoms replaced by an alkyl or aryl group in ammonia. The amine is a primary amine if one hydrogen atom is replaced, it is a secondary amine if 2 hydrogen atoms are replaced and if three hydrogen atoms are replaced it is known as a tertiary amine.
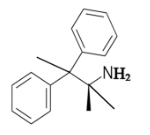
The given compound is as follows:
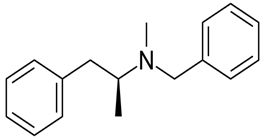
The constitutional isomer with a tertiary amine is represented as follows:
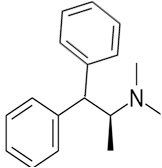
Here all 3 hydrogen atomsare replaced and therefore this amine called as a tertiary amine (3o).
(f)
Interpretation:
The structure of benzphetaminehychloride molecule needs to be drawn.
Concept introduction:
Benzphetamine hydrochloride can be defined as the hydrochloride salt version of benzphetamine.
Answer to Problem 73P
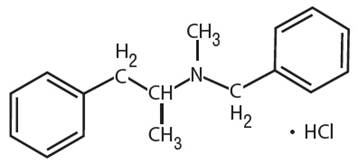
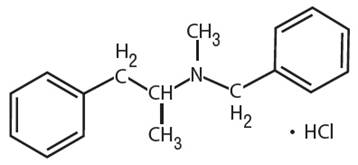
Explanation of Solution
Benzphetamine hydrochloride can be defined as the hydrochloride salt version of benzphetamine. Its molecular formula is C17H22ClN. It has the following structure.
(g)
Interpretation:
The products formed if benzphetamine is treated with acetic acid needs to be determined.
Concept introduction:
Amines are derived from ammonia and formed by replacement of one or more hydrogen atoms by alkyl or aryl groups. They can be called as alkylamines and arylamines. Benzphetamine is a tertiary amine.
Answer to Problem 73P
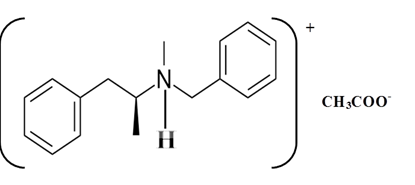
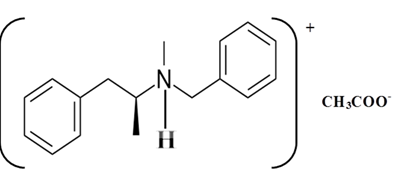
Explanation of Solution
Whether an amine is primary (1o), secondary (2o) or tertiary (3o) depends on the number of hydrogen atoms replaced by an alkyl or aryl group in ammonia. The amine is a primary amine if one hydrogen atom is replaced, it is a secondary amine if 2 hydrogen atoms are replaced and if three hydrogen atoms are replaced it is known as a tertiary amine.
Benzphetamine is a tertiary ammine. It has basic properties and it accepts protons from acids. Acetic acid donates a proton to this amine and formed below the salt.
Want to see more full solutions like this?
Chapter 18 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
- Identify the compound with the longest carbon - nitrogen bond. O CH3CH2CH=NH O CH3CH2NH2 CH3CH2C=N CH3CH=NCH 3 The length of all the carbon-nitrogen bonds are the samearrow_forwardIdentify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forwardH H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forward
- Rank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forwardRank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forwardchoose the description that best describes the geometry for the following charged species ch3-arrow_forward
- Why isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forwardWhat is the approximate bond angle around the nitrogen atom? HNH H Harrow_forwardOH 1. NaOCH2CH3 Q 2. CH3CH2Br (1 equiv) H3O+ Select to Draw 1. NaOCH2 CH3 2. CH3Br (1 equiv) heat Select to Edit Select to Drawarrow_forward
- Complete and balance the following half-reaction in acidic solution. Be sure to include the proper phases for all species within the reaction. S₂O₃²⁻(aq) → S₄O₆²⁻(aq)arrow_forwardQ Select to Edit NH3 (CH3)2CHCI (1 equiv) AICI 3 Select to Draw cat. H2SO4 SO3 (1 equiv) HO SOCl2 pyridine Select to Edit >arrow_forwardComplete and balance the following half-reaction in basic solution. Be sure to include the proper phases for all species within the reaction. Zn(s) → Zn(OH)₄²⁻(aq)arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



