
EBK CHEMISTRY FOR CHANGING TIMES
14th Edition
ISBN: 8220100663482
Author: MCCREARY
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 18, Problem 2CHQ
Interpretation Introduction
Interpretation:
Whether the experiment mimic that of the stomach and what are the differences that might exist between the conditions of the experiment and those of the stomach are to be written.
Concept introduction:
- Antacids are composed of active ingredients (which are a cure or remedy to an adverse symptom) along with other inactive ingredients (which control the timing and method of delivery of the drug).
- Active ingredients mainly compose of hydroxides and carbonates of magnesium, aluminum and calcium.
- Vinegar solution is actually acetic acid, a weak acid. In the experiment, addition of the tablets into vinegar solution shows the neutralization reaction that takes place in the acidic environment of the stomach. A neutralization reaction can be written as
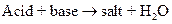
- Heartburns occur when the gastric contents from the stomach flows back into the esophagus.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Curved arrows are used to illustrate the flow of electrons.
Using the provided structures, draw the curved arrows that
epict the mechanistic steps for the proton transfer between
a hydronium ion and a pi bond.
Draw any missing organic structures in the empty boxes.
Be sure to account for all lone-pairs and charges as well as
bond-breaking and bond-making steps.
2 56°F
Mostly cloudy
F1
Drawing Arrows
>
Q Search
F2
F3
F4
▷11
H.
H
: CI:
H
+
Undo
Reset
Done
DELL
Calculate the chemical shifts in 13C and 1H NMR for 4-chloropropiophenone ? Write structure and label hydrogens and carbons. Draw out the benzene ring structure when doing it
1) Calculate the longest and shortest wavelengths in the Lyman and Paschen series.
2) Calculate the ionization energy of He* and L2+ ions in their ground states.
3) Calculate the kinetic energy of the electron emitted upon irradiation of a H-atom in ground state by a 50-nm radiation.
Chapter 18 Solutions
EBK CHEMISTRY FOR CHANGING TIMES
Ch. 18 - Prob. 1RQCh. 18 - Prob. 2RQCh. 18 - Prob. 3RQCh. 18 - Prob. 4RQCh. 18 - Prob. 5RQCh. 18 - Prob. 6RQCh. 18 - What are prostaglandins? How do prostaglandins...Ch. 18 - Prob. 8RQCh. 18 - Prob. 9RQCh. 18 - Describe a synergistic effect involving drugs.
Ch. 18 - Prob. 11RQCh. 18 - Prob. 12RQCh. 18 - Prob. 13RQCh. 18 - Prob. 14RQCh. 18 - Prob. 15RQCh. 18 - Prob. 16RQCh. 18 - Prob. 17PCh. 18 - Classify the following drugs as natural,...Ch. 18 - Prob. 19PCh. 18 - Prob. 20PCh. 18 - Prob. 21PCh. 18 - Prob. 22PCh. 18 - Prob. 23PCh. 18 - Prob. 24PCh. 18 - Prob. 25PCh. 18 - Prob. 26PCh. 18 - Which of the following was originally marketed as...Ch. 18 - Prob. 28PCh. 18 - Which of the following is not considered to be an...Ch. 18 - How does heroin differ from morphine in its...Ch. 18 - Prob. 31PCh. 18 - How are endorphins related to (a) the anesthetic...Ch. 18 - Prob. 33PCh. 18 - Prob. 34PCh. 18 - Prob. 35PCh. 18 - Prob. 36PCh. 18 - Prob. 37PCh. 18 - Prob. 38PCh. 18 - Prob. 39PCh. 18 - Prob. 40PCh. 18 - Prob. 41PCh. 18 - Prob. 42PCh. 18 - Prob. 43PCh. 18 - Prob. 44PCh. 18 - Prob. 45PCh. 18 - Prob. 46PCh. 18 - Give three differences between hormones and...Ch. 18 - Prob. 48PCh. 18 - Prob. 49PCh. 18 - Prob. 50PCh. 18 - Prob. 51PCh. 18 - Prob. 52PCh. 18 - Prob. 53PCh. 18 - Prob. 54PCh. 18 - Prob. 55PCh. 18 - Prob. 56PCh. 18 - Prob. 57PCh. 18 - Which of the following is often treated with...Ch. 18 - Prob. 59PCh. 18 - Prob. 60PCh. 18 - Prob. 61PCh. 18 - Prob. 62PCh. 18 - Prob. 63PCh. 18 - Prob. 64PCh. 18 - Prob. 65PCh. 18 - Prob. 66PCh. 18 - Prob. 67PCh. 18 - What is an agonist? What is an antagonist?Ch. 18 - Prob. 69PCh. 18 - Prob. 70PCh. 18 - Prob. 71PCh. 18 - What is the difference between drug abuse and drug...Ch. 18 - Review the information on structure and function...Ch. 18 - Prob. 74APCh. 18 - Prob. 75APCh. 18 - Prob. 76APCh. 18 - Prob. 77APCh. 18 - Prob. 78APCh. 18 - Prob. 79APCh. 18 - Prob. 80APCh. 18 - Prob. 81APCh. 18 - Prob. 82APCh. 18 - Prob. 83APCh. 18 - Prob. 84APCh. 18 - Prob. 85APCh. 18 - Prob. 86APCh. 18 - Prob. 87APCh. 18 - Prob. 88APCh. 18 - Prob. 89APCh. 18 - Prob. 90APCh. 18 - Prob. 91APCh. 18 - Prob. 92APCh. 18 - Prob. 93APCh. 18 - Prob. 94APCh. 18 - Prob. 95APCh. 18 - Which of the following compounds are classified as...Ch. 18 - Prob. 97APCh. 18 - Prob. 98APCh. 18 - Prob. 99APCh. 18 - Prob. 100APCh. 18 - Prob. 101APCh. 18 - Prob. 102APCh. 18 - Prob. 103APCh. 18 - Prob. 104APCh. 18 - Prob. 18.1CTECh. 18 - Prob. 18.2CTECh. 18 - Prob. 18.3CTECh. 18 - Prob. 18.4CTECh. 18 - Prob. 18.5CTECh. 18 - Prob. 18.6CTECh. 18 - Prob. 18.7CTECh. 18 - Prob. 18.8CTECh. 18 - Prob. 1CGPCh. 18 - Prob. 2CGPCh. 18 - Prob. 3CGPCh. 18 - Prob. 4CGPCh. 18 - Prob. 5CGPCh. 18 - Prob. 6CGPCh. 18 - Prob. 7CGPCh. 18 - Prob. 8CGPCh. 18 - Prob. 9CGPCh. 18 - Prob. 10CGPCh. 18 - Prob. 11CGPCh. 18 - Prob. 12CGPCh. 18 - Prob. 13CGPCh. 18 - Prob. 14CGPCh. 18 - Prob. 1CHQCh. 18 - Prob. 2CHQCh. 18 - Prob. 3CHQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Calculate the ionization energy of He+ and Li²+ ions in their ground states. Thannnxxxxx sirrr Ahehehehehejh27278283-4;*; shebehebbw $+$;$-;$-28283773838 hahhehdvaarrow_forwardPlleeaasseee solllveeee question 3 andd thankss sirr, don't solve it by AI plleeaasseee don't use AIarrow_forwardCalculate the chemical shifts in 13C and 1H NMR for 4-chloropropiophenone ? Write structure and label hydrogens and carbonsarrow_forward
- 4. Read paragraph 4.15 from your textbook, use your calculated lattice energy values for CuO, CuCO3 and Cu(OH)2 an explain thermal decomposition reaction of malachite: Cu2CO3(OH)2 →2CuO + H2O + CO2 (3 points)arrow_forwardPlease sirrr soollveee these parts pleaseeee and thank youuuuuarrow_forwardIII O Organic Chemistry Using wedges and dashes in skeletal structures Draw a skeletal ("line") structure for each of the molecules below. Be sure your structures show the important difference between the molecules. key O O O O O CHON Cl jiii iiiiiiii You can drag the slider to rotate the molecules. Explanation Check Click and drag to start drawing a structure. Q Search X G ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Use F 3 W C 3/5arrow_forward
- 3. Use Kapustinskii's equation and data from Table 4.10 in your textbook to calculate lattice energies of Cu(OH)2 and CuCO3 (4 points)arrow_forward2. Copper (II) oxide crystalizes in monoclinic unit cell (included below; blue spheres 2+ represent Cu²+, red - O²-). Use Kapustinski's equation (4.5) to calculate lattice energy for CuO. You will need some data from Resource section of your textbook (p.901). (4 points) CuOarrow_forwardWhat is the IUPAC name of the following compound? OH (2S, 4R)-4-chloropentan-2-ol O (2R, 4R)-4-chloropentan-2-ol O (2R, 4S)-4-chloropentan-2-ol O(2S, 4S)-4-chloropentan-2-olarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY