
Concept explainers
18-4 Answer true or false.
(a) The
(b) The VSEPR model predicts bond angles of 180° about the carbonyl carbon of a carboxyl group.
(c) The VSEPR model predicts bond angles of 109.5° about the oxygen of the OH group of a carboxyl group.
(d) The carbonyl carbon of a carboxyl group can be a stereocenter, depending on its location within a molecule.
(e) Carboxylic acids can be prepared by chromic acid oxidation of primary alcohols and of
(f) The product of chromic acid oxidation of hexanoic acid is 1-hexanol.
(a)
Interpretation:
Whether the statement.
The functional groups of carboxylic acids are a carbonyl group bonded to a hydroxyl group is true or false.
Concept Introduction:
Carboxylic acid group contain one carbonyl group and one hydroxyl group that is −COOH.
Explanation of Solution
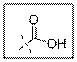
As per the given structure the carboxyl group contains a carbonyl group which is bonded to a hydroxyl group. So the statement is true.
Carboxylic acid functional group is known as COOH. When this group is elaborately written it shows two functional groups. Those groups are carbonyl group and another is hydroxyl group.
So the carboxyl group contains a carbonyl group which is bonded to a hydroxyl group.
(b)
Interpretation:
Whether the statement.
The VSEPR model predicts bond angles of
Concept Introduction:
Carboxylic acid group contain one carbonyl group and one hydroxyl group. VSEPR theory stands for valence shell electron pair repulsion theory. This theory predict the geometry of molecules on the basis of the number of electron pairs present surrounding the central atoms.
Explanation of Solution
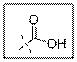
As per the given structure the carboxyl group contains a carbonyl group which is bonded to a hydroxyl group. The angle is
Carboxylic acid functional group is known as COOH. When this group is elaborately written it shows two functional groups. Those groups are carbonyl group and another is hydroxyl group. The hybridization of the carbonyl carbon is sp2 and thus expected bond angle is
(c)
Interpretation:
Whether the statement.
The VSEPR model predicts bond angles of
Concept Introduction:
Carboxylic acid group contain one carbonyl group and one hydroxyl group and the oxygen atom of hydroxyl group is sp3 hybridized. VSEPR theory stands for valence shell electron pair repulsion theory. This theory predict the geometry of molecules on the basis of the number of electron pairs present surrounding the central atoms.
Explanation of Solution
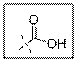
As per the given structure the carboxyl group contains a carbonyl group which is bonded to a hydroxyl group. The oxygen atom of hydroxyl group is sp3 hybridized. So expected bond angle is angle is
But due to two lone pair electrons on oxygen atom bond angle will be lesser than
Carboxylic acid functional group is known as COOH. When this group is elaborately written it shows two functional groups. Those groups are carbonyl group and another is hydroxyl group. The hybridization of the oxygen atom of hydroxyl group is sp3 and thus expected bond angle is
(d)
Interpretation:
Whether the statement.
The carbonyl carbon of a carboxyl group can be a stereocenter, depending on its location within a molecule is true or false.
Concept Introduction:
Carbonyl carbon has double bonded oxygen atom and also linked with hydroxyl group. Stereocentre is defined as the centre in which all the groups attached to the atom generally carbon atom, should be different.
Explanation of Solution
As carbonyl group has double bonded oxygen atom and thus it can’t be a stereocenter. So the statement is false.
Carboxyl group is a carbonyl group bonded to a hydroxyl group. Carbonyl carbon being double bonded to oxygen it is not a stereocenter.
(e)
Interpretation:
Whether the statement.
Carboxylic acid can be prepared by chromic acid oxidation of primary alcohols and of aldehydes is true or false.
Concept Introduction:
Chromic acid is very strong oxidizing agent. Oxidizing agent is defined as the oxidant which gains electrons and reduced in the chemical reaction.
Explanation of Solution
Chromic acid being very strong oxidizing agent it is possible to oxidize primary alcohols and aldehydes to carboxylic acid. So the statement is true.
When primary alcohols are oxidized with chromic acid then the alcohol is first converted to aldehyde which in turn converted to carboxylic acid.
(f)
Interpretation:
Whether the statement.
The product of chromic acid oxidation of hexanoic acid is 1-hexanol is true or false.
Concept Introduction:
When 1-hexanol is oxidized it is converted to hexanoic acid. Oxidation is the process in which either loss of electrons or increase in the oxidation number takes plcae.
Explanation of Solution
1-Hexanol can be converted to hexanoic acid after treatment with chromic acid but the product of chromic acid oxidation of hexanoic acid is not 1-hexanol.
So the statement is false.
1-Hexanol can be converted to hexanoic acid with chromic acid. But 1-hexanol can’t be obtained after oxidizing hexanoic acid with chromic acid. For getting 1-hexanol from hexanoic acid reducing agent is required instead of chromic acid.
Want to see more full solutions like this?
Chapter 18 Solutions
Student Solutions Manual for Bettelheim/Brown/Campbell/Farrell/Torres' Introduction to General, Organic and Biochemistry, 11th
- Which carbocation is more stable?arrow_forwardAre the products of the given reaction correct? Why or why not?arrow_forwardThe question below asks why the products shown are NOT the correct products. I asked this already, and the person explained why those are the correct products, as opposed to what we would think should be the correct products. That's the opposite of what the question was asking. Why are they not the correct products? A reaction mechanism for how we arrive at the correct products is requested ("using key intermediates"). In other words, why is HCl added to the terminal alkene rather than the internal alkene?arrow_forward
- My question is whether HI adds to both double bonds, and if it doesn't, why not?arrow_forwardStrain Energy for Alkanes Interaction / Compound kJ/mol kcal/mol H: H eclipsing 4.0 1.0 H: CH3 eclipsing 5.8 1.4 CH3 CH3 eclipsing 11.0 2.6 gauche butane 3.8 0.9 cyclopropane 115 27.5 cyclobutane 110 26.3 cyclopentane 26.0 6.2 cycloheptane 26.2 6.3 cyclooctane 40.5 9.7 (Calculate your answer to the nearest 0.1 energy unit, and be sure to specify units, kJ/mol or kcal/mol. The answer is case sensitive.) H. H Previous Nextarrow_forwardA certain half-reaction has a standard reduction potential Ered +1.26 V. An engineer proposes using this half-reaction at the anode of a galvanic cell that must provide at least 1.10 V of electrical power. The cell will operate under standard conditions. Note for advanced students: assume the engineer requires this half-reaction to happen at the anode of the cell. Is there a minimum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, check the "yes" box and calculate the minimum. Round your answer to 2 decimal places. If there is no lower limit, check the "no" box.. Is there a maximum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, check the "yes" box and calculate the maximum. Round your answer to 2 decimal places. If there is no upper limit, check the "no" box. yes, there is a minimum. 1 red Πν no minimum Oyes, there is a maximum. 0 E red Dv By using the information in the ALEKS…arrow_forward
- (11pts total) Consider the arrows pointing at three different carbon-carbon bonds in the molecule depicted below. Bond B Bond A Bond C a. (2pts) Which bond between A-C is weakest? Which is strongest? Place answers in appropriate boxes. Weakest Bond Strongest Bond b. (4pts) Consider the relative stability of all cleavage products that form when bonds A, B, AND C are homolytically cleaved/broken. Hint: cleavage products of bonds A, B, and C are all carbon radicals. i. Which ONE cleavage product is the most stable? A condensed or bond line representation is fine. ii. Which ONE cleavage product is the least stable? A condensed or bond line representation is fine. c. (5pts) Use principles discussed in lecture, supported by relevant structures, to succinctly explain the why your part b (i) radical is more stable than your part b(ii) radical. Written explanation can be no more than one-two succinct sentence(s)!arrow_forward. 3°C with TH 12. (10pts total) Provide the major product for each reaction depicted below. If no reaction occurs write NR. Assume heat dissipation is carefully controlled in the fluorine reaction. 3H 24 total (30) 24 21 2h • 6H total ● 8H total 34 래 Br2 hv major product will be most Substituted 12 hv Br NR I too weak of a participate in P-1 F₂ hv Statistically most favored product will be major = most subst = thermo favored hydrogen atom abstractor to LL Farrow_forwardFive chemistry project topic that does not involve practicalarrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co




