
Organic Chemistry, Loose-leaf Version
8th Edition
ISBN: 9781305865549
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 18, Problem 18.21P
Interpretation Introduction
Interpretation:
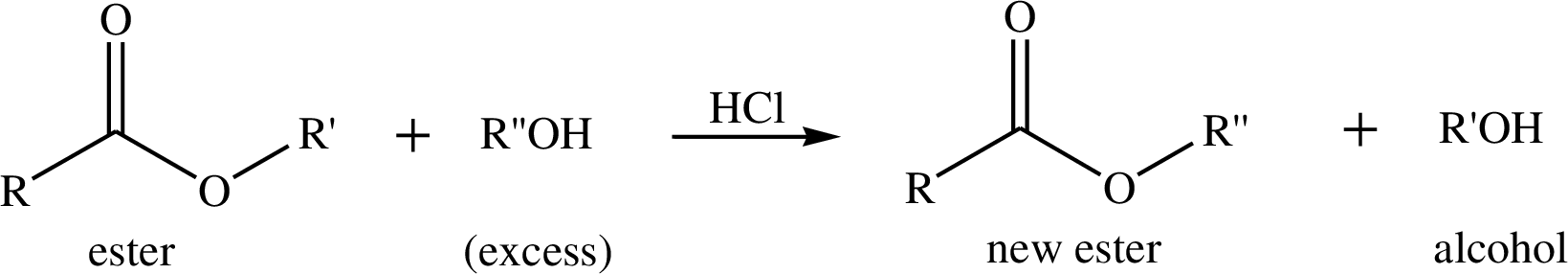
Way in which the given observation bears on the formation of a terahedral carbonyl addition intermediate during acid-catalysed easter hydrolyis, the corresponding transformation has to be shown.
Concept Introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
What is the product of the reaction?
F3C.
CF3
OMe
NaOH / H₂O
What is the product of the reaction?
F3C.
CF3
OMe
NaOH / H₂O
What would you expect to be the major product obtained from the following reaction? Please explain what is happening here. Provide a detailed explanation and a drawing showing how the reaction occurs. The correct answer to this question is V.
Chapter 18 Solutions
Organic Chemistry, Loose-leaf Version
Ch. 18.1 - Prob. 18.1PCh. 18.2 - Prob. 18.2PCh. 18.4 - Prob. 18.3PCh. 18.4 - Prob. 18.4PCh. 18.4 - Synthesis of nitriles by nucleophilic displacement...Ch. 18.5 - Complete the following transesterification...Ch. 18.6 - Complete and balance equations for the following...Ch. 18.8 - Prob. AQCh. 18.8 - Several compounds have been found to inhibit...Ch. 18.8 - Prob. CQ
Ch. 18.8 - The following sequence of steps is used to create...Ch. 18.9 - Prob. 18.8PCh. 18.9 - Prob. 18.9PCh. 18.10 - Prob. 18.10PCh. 18.10 - Show how to convert (R)-2-phenylpropanoic acid to...Ch. 18 - Prob. 18.12PCh. 18 - Write the IUPAC name for each compound. (a)...Ch. 18 - Prob. 18.14PCh. 18 - Prob. 18.15PCh. 18 - Propose a structural formula for compound A,...Ch. 18 - Propose a structural formula for compound B,...Ch. 18 - Propose a structural formula for each compound...Ch. 18 - Draw a structural formula for the principal...Ch. 18 - Prob. 18.20PCh. 18 - Prob. 18.21PCh. 18 - Prob. 18.22PCh. 18 - Prob. 18.23PCh. 18 - Prob. 18.24PCh. 18 - Show the product expected when the following...Ch. 18 - The reagent diisobutylaluminum hydride (DIBALH)...Ch. 18 - Prob. 18.27PCh. 18 - Prob. 18.28PCh. 18 - Prob. 18.29PCh. 18 - Nicotinic acid, more commonly named niacin, is one...Ch. 18 - Prob. 18.31PCh. 18 - Prob. 18.32PCh. 18 - Prob. 18.33PCh. 18 - Prob. 18.34PCh. 18 - Prob. 18.35PCh. 18 - Isoniazid, a drug used to treat tuberculosis, is...Ch. 18 - Prob. 18.37PCh. 18 - A step in a synthesis of PGE1 (prostaglandin E1,...Ch. 18 - Prob. 18.39PCh. 18 - Prob. 18.40PCh. 18 - Show how to synthesize 5-nonanone from...Ch. 18 - Prob. 18.42PCh. 18 - The following sequence of steps converts...Ch. 18 - Prob. 18.44PCh. 18 - Prob. 18.45PCh. 18 - Prob. 18.46PCh. 18 - Prob. 18.47PCh. 18 - Following is a retrosynthetic analysis for the...Ch. 18 - Prob. 18.50PCh. 18 - Given this retrosynthetic analysis, propose a...Ch. 18 - Prob. 18.52PCh. 18 - Prob. 18.53PCh. 18 - Prob. 18.54PCh. 18 - In Problem 7.28, we saw this step in Johnsons...Ch. 18 - Prob. 18.56PCh. 18 - Prob. 18.57PCh. 18 - Prob. 18.58PCh. 18 - Prob. 18.59PCh. 18 - Prob. 18.60PCh. 18 - Prob. 18.61PCh. 18 - Prob. 18.62PCh. 18 - Using your reaction roadmap as a guide, show how...Ch. 18 - Using your reaction roadmap as a guide, show how...Ch. 18 - Using your reaction roadmap as a guide, show how...Ch. 18 - Using your reaction roadmap as a guide, show how...Ch. 18 - Minoxidil is a molecule that causes hair growth in...Ch. 18 - Prob. 18.69PCh. 18 - Prob. 18.70PCh. 18 - Prob. 18.71P
Knowledge Booster
Similar questions
- Please answer the question for the reactions, thank youarrow_forwardWhat is the product of the following reaction? Please include a detailed explanation of what is happening in this question. Include a drawing showing how the reagent is reacting with the catalyst to produce the correct product. The correct answer is IV.arrow_forwardPlease complete the reactions, thank youarrow_forward
- Consider the synthesis. What is compound Y? Please explain what is happening in this question. Provide a detailed explanation and a drawing to show how the compound Y creates the product. The correct answer is D.arrow_forwardWhat would be the major product of the following reaction? Please include a detailed explanation of what is happening in this question. Include steps and a drawing to show this reaction proceeds and how the final product is formed. The correct answer is B. I put answer D and I don't really understand what is going on in the question.arrow_forwardWhat is the product of the following reaction? Please explain what is happening in this question. Provide a detailed explanation and a drawing showing how the reagent is reacting with the catalysts to product the correct product. The correct answer is B.arrow_forward
- What is the missing intermediate 1 and the final product 2. Please include a detailed explanation explaining the steps of malonic ester synthesis. Please include drawings of the intermediate and how it occurs and how the final product is former.arrow_forwardWhat would be the reagents and conditions above and below the arrow that will complete the proposed acetoacetic ester synthesis? If it cannot be done efficiently, then I will choose that answer. There could be 2 or 4 reagents involved. Please provide a detailed explanation and drawings showing how it would proceed with the correct reagents.arrow_forwardFor benzene, the ∆H° of vaporization is 30.72 kJ/mol and the ∆S° of vaporization is 86.97 J/mol・K. At 1.00 atm and 228.0 K, what is the ∆G° of vaporization for benzene, in kJ/mol?arrow_forward
- The reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reaction. it is spontaneous only at High T, it is spontaneous at low T it is nonspontaneous at all T it is spontanrous at all T. it is non spontaneous only at low T.arrow_forwardThe reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reactionarrow_forwardWhich of the following has the largest standard molar entropy, S° (298.15 K) He H2 NaCl KBr Hgarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
