
EP ESSENTIAL ORG.CHEM.-MOD.MASTERING
3rd Edition
ISBN: 9780133858501
Author: Bruice
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.6, Problem 14P
What amino acid would be formed using the N-phthalimidomalonic ester synthesis when the following
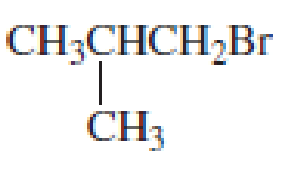
- a. CH3SCH2CH2Br
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
1. Amides are comparably unreactive to nucleophilic acyl substitution, yet the B-lactam antibiotics
shown) are reactive with a hydroxyl group side chain of an amino acid. Why?
R
HO
Please give the reactions of the following of its esterification reaction:
1. benzoic acid
2. acetic acid + benzyl alcohol
Which of the following product(s) would not be a side product if aniline is brominated without the protection
of its amino group?
A 3-bromoaniline
D. 3,5-dibromoaniline
B. 24,6-tribromoaniline
O A and D
O C. 2.4-dibromoaniline
O Band C
Chapter 17 Solutions
EP ESSENTIAL ORG.CHEM.-MOD.MASTERING
Ch. 17.1 - a. Explain why, when the imidazole ring of...Ch. 17.2 - Prob. 2PCh. 17.3 - Prob. 3PCh. 17.3 - Prob. 4PCh. 17.3 - Prob. 6PCh. 17.4 - Calculate the pI of each of the following amino...Ch. 17.4 - a. Which amino acid has the lowest pI value? b....Ch. 17.5 - What aldehyde is formed when valine is treated...Ch. 17.5 - Prob. 10PCh. 17.5 - Prob. 11P
Ch. 17.5 - Prob. 12PCh. 17.6 - Prob. 13PCh. 17.6 - What amino acid would be formed using the...Ch. 17.6 - What amino acid would be formed when the aldehyde...Ch. 17.7 - Pig liver esterase is an enzyme that catalyzes the...Ch. 17.8 - Prob. 17PCh. 17.8 - Prob. 18PCh. 17.8 - Prob. 19PCh. 17.8 - Prob. 20PCh. 17.10 - Prob. 21PCh. 17.10 - Prob. 22PCh. 17.10 - Why does cyanogen bromide not cleave on the C-side...Ch. 17.10 - Prob. 24PCh. 17.10 - Prob. 26PCh. 17.12 - Prob. 27PCh. 17.13 - a. Which would have the greatest percentage of...Ch. 17 - Draw the predominant form of the following amino...Ch. 17 - What is the pI of serine?Ch. 17 - Prob. 31PCh. 17 - Prob. 32PCh. 17 - Which would have a higher percentage of negative...Ch. 17 - Draw the form of aspartate that predominates at...Ch. 17 - Prob. 35PCh. 17 - A professor was preparing a manuscript for...Ch. 17 - a. Why is the pKa of the glutamate side chain...Ch. 17 - Prob. 38PCh. 17 - Determine the amino acid sequence of a polypeptide...Ch. 17 - Prob. 40PCh. 17 - Prob. 41PCh. 17 - Three peptides were obtained from a trypsin...Ch. 17 - Prob. 43PCh. 17 - After the polypeptide shown here was treated with...Ch. 17 - The disulfide bridges of a polypeptide were...Ch. 17 - -Amino acids can be prepared by treating an...Ch. 17 - Reaction of a polypeptide with carboxypeptidase A...Ch. 17 - Prob. 48PCh. 17 - Prob. 49PCh. 17 - Show how valine can be prepared by a. a Strecker...Ch. 17 - Prob. 51PCh. 17 - Why is proline never found in an -helix?Ch. 17 - Determine the amino acid sequence of a polypeptide...Ch. 17 - Prob. 55PCh. 17 - A chemist wanted to test his hypothesis that the...Ch. 17 - A normal polypeptide and a mutant of the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the reaction type. a. hydrolysis (in acid) b. hydration c. dehydration d. acid base e. esterification f. amide synthesis g. hydrolysis (in base)arrow_forwardWhat is the difference between the aminolysis of an ester and that of an acyl halide?arrow_forward2. In the synthesis of peptides, carboxylic acids are condensed with amines in the present of a reagent such as dicyclohexylcarbodiimide (DCC) [Section 25.6]. a. Propose a mechanism for the following, using curved arrow notation. H₂N. glycine OH H₂N 요. glycine OH DCC H₂N. N H gly-gly LOHarrow_forward
- Mineral oil was used in Fischer Esterification to separate the ester product from the aqueous reactants, preventing the reaction from reversing upon reaction with water. Mineral oil was used for experiments at home. How would this be carried out in professional laboratories? Can something else aside from mineral oil be used?arrow_forwardSome penicillins cannot be administered orally because their B-lactam is rapidly hydrolyzed by the acidic environment of the stomach. Draw the product that forms in the following hydrolysis reaction. нн N. R. но エーZarrow_forwardWhich of the following substituent is an ortho/para-directing activator and an ortho/para-directing deactivator? a amino group b halogen group c nitro group d carboxyl grouparrow_forward
- And write the Molar Mass.arrow_forwardpls explain what other compounds or reagents would be needed to form it given you have HCl and 4-methyl-2-hexene what other compounds or reagents are needed to form N-ethyl-4-methylhex-2-amine?arrow_forward6. Complete the following aqueous reactions: a. N-methylethanamide + NaOH b. N,N-diethylpropanamide + HBr → c. What are these reactions called?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY