
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.11, Problem 22P
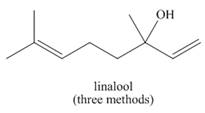
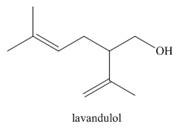
Linalool (the Chapter 9 opening molecule) and lavandulol are two of the major components of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandulol be formed by reduction of a carbonyl compound? (c) Why can’t linalool be prepared by a similar pathway?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
None
Carbohydrates- Draw out the Hawthorne structure for a sugar from the list given in class. Make sure to write out all atoms except for carbons within the ring. Make sure that groups off the carbons in the ring are in the correct orientation above or below the plane. Make sure that bonds are in the correct orientation. Include the full name of the sugar.
You can draw out your curve within the text box or upload a drawing below.
How many milliliters of 97.5(±0.5) wt% H2SO4 with a density of 1.84(±0.01) g/mL will you need to prepare 2.000 L of 0.110 M H2SO4?
If the uncertainty in delivering H2SO4 is ±0.01 mL, calculate the absolute uncertainty in the molarity (0.110 M). Assume there is negligible uncertainty in the formula mass of H2SO4 and in the final volume (2.000 L) and assume random error.
Chapter 17 Solutions
Organic Chemistry (6th Edition)
Ch. 17.1 - Prob. 1PCh. 17.2 - Which carbonyl groups in the anticancer drug taxol...Ch. 17.2 - Prob. 3PCh. 17.4 - Problem 20.4 What alcohol is formed when each...Ch. 17.4 - Problem 20.5 What aldehyde or ketone is needed to...Ch. 17.5 - Problem 20.8 Draw the products formed (including...Ch. 17.7 - Problem 20.10 Draw a stepwise mechanism for the...Ch. 17.7 - Prob. 10PCh. 17.7 - Problem 20.12 Draw the products formed from ...Ch. 17.7 - Prob. 12P
Ch. 17.7 - Prob. 13PCh. 17.8 - Prob. 14PCh. 17.8 - Problem-20.16 Review the oxidation reactions using...Ch. 17.9 - Problem-20.17 Write the step(s) needed to convert ...Ch. 17.9 - Prob. 18PCh. 17.10 - Problem 20.21 Draw the product of each reaction.
...Ch. 17.10 - Problem 20.22 Draw the products (including...Ch. 17.11 - Problem 20.23 What Grignard reagent and carbonyl...Ch. 17.11 - Problem 20.24 Linalool (the Chapter 9 opening...Ch. 17.11 - Problem 20.25 What Grignard reagent and carbonyl...Ch. 17.12 - Prob. 24PCh. 17.13 - Problem 20.28 What ester and Grignard reagent are...Ch. 17.13 - Prob. 27PCh. 17.13 - Problem 20.30 What reagent is needed to convert ...Ch. 17.13 - Prob. 29PCh. 17.14 - What carboxylic acid formed from each alkyl halide...Ch. 17 - 20.37 Devise a synthesis of each alcohol from...Ch. 17 - 20.38 Draw the products formed when pentanal is...Ch. 17 - 20.39 Draw the product formed when is treated...Ch. 17 - The stereochemistry of the products of reduction...Ch. 17 - Prob. 40PCh. 17 - 20.42 Draw the products or each reduction...Ch. 17 - 20.44 Draw all stereoisomers formed in each...Ch. 17 - 20.54 Draw a stepwise mechanism for the following...Ch. 17 - Prob. 57PCh. 17 - Prob. 58PCh. 17 - 20.57 What ester and Grignard reagent are needed...Ch. 17 - 20.58 What organolithium reagent and carbonyl...Ch. 17 - 20.59 What epoxide and organometallic reagent are...Ch. 17 - Prob. 62PCh. 17 - 20.69 An unknown compound A (molecular formula )...Ch. 17 - 20.70 Treatment of compound C (molecular formula )...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used hand raiting and don't used Ai solutionarrow_forward* How many milliliters of 97.5(±0.5) wt% H2SO4 with a density of 1.84(±0.01) g/mL will you need to prepare 2.000 L of 0.110 M H2SO4? * If the uncertainty in delivering H2SO4 is ±0.01 mL, calculate the absolute uncertainty in the molarity (0.110 M). Assume there is negligible uncertainty in the formula mass of NaOH and in the final volume (2.000 L) and assume random error.arrow_forwardYou are tasked with creating a calibration curve for the absorbance of cobalt solutions of various concentrations. You must prepare 5 standards with concentrations between 1.00 mg/L and 10.0 mg/L Co2+. You have a stock solution with a concentration of 40 mg/L Co2+ and all the standard lab glassware including transfer pipets and flasks. Explain how you would make your 5 standard solutions of various concentrations, including what glassware you would use to measure and prepare each solution.arrow_forward
- Predict the product and write the mechanism. CH3-CH=CH-CH2-CH3 + NBS- hv CCl4arrow_forwardHow exactly is carbon disulfide used in industry? Specifically, where does it come in during rubber or textile production and what is the chemical processes?arrow_forwardA researcher has developed a new analytical method to determine the percent by mass iron in solids. To test the new method, the researcher purchases a standard reference material sample that is 2.85% iron by mass. Analysis of the iron standard with the new method returns values of 2.75%, 2.89%, 2.77%, 2.81%, and 2.87%. Does the new method produce a result that is significantly different from the standard value at the 95% confidence level?arrow_forward
- Create a drawing of an aceral with at least 2 isopropoxy groups, and a total of 11 carbon atomsarrow_forward4. Predict the major product(s) for each of the following reactions. HBr (1 equiv.) peroxide, A a. b. NBS, peroxide, Aarrow_forwardIn addition to the separation techniques used in this lab (magnetism, evaporation, and filtering), there are other commonly used separation techniques. Some of these techniques are:Distillation – this process is used to separate components that have significantly different boiling points. The solution is heated and the lower boiling point substance is vaporized first. The vapor can be collected and condensed and the component recovered as a pure liquid. If the temperature of the mixture is then raised, the next higher boiling component will come off and be collected. Eventually only non-volatile components will be left in the original solution.Centrifugation – a centrifuge will separate mixtures based on their mass. The mixture is placed in a centrifuge tube which is then spun at a high speed. Heavier components will settle at the bottom of the tube while lighter components will be at the top. This is the technique used to separate red blood cells from blood plasma.Sieving – this is…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License