
Concept explainers
(a)
Interpretation:
The intramolecular esterification reaction of following hydroxycarboxylic acid should be determined:
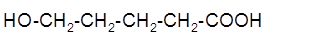
Concept Introduction:
Alcohols are the organic compounds with general chemical formula of R-OH whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
(b)
Interpretation:
The intramolecular esterification reaction of following hydroxycarboxylic acid should be determined:
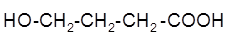
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Alcohols are the organic compounds with general chemical formula of R-OH whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- E17E.2(a) The following mechanism has been proposed for the decomposition of ozone in the atmosphere: 03 → 0₂+0 k₁ O₁₂+0 → 03 K →> 2 k₁ Show that if the third step is rate limiting, then the rate law for the decomposition of O3 is second-order in O3 and of order −1 in O̟.arrow_forward10.arrow_forwardDon't used Ai solution and don't used hand raitingarrow_forward
- 2arrow_forwardWhich of the following starting materials and reagents would be best to produce a racemic mixture of 3-methyl-3-hexanol? heptanone and 1. CH3MgBr 2. H3O+ hexanal and 1. CH3MgBr, 2. H3O+ 3-hexanone and 1. CH3MgBr, 2. H3O+ butanal and 1. CH3CH2MgBr, 2. H3O+arrow_forwardCan someone draw a reaction mechanism of this reaction please I was told that the boc l alanine is deprotonated first and acts as the nucleophile attacking the EDCL and can you please show all the intermediates and side products and the water at the endarrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning



