
Concept explainers
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
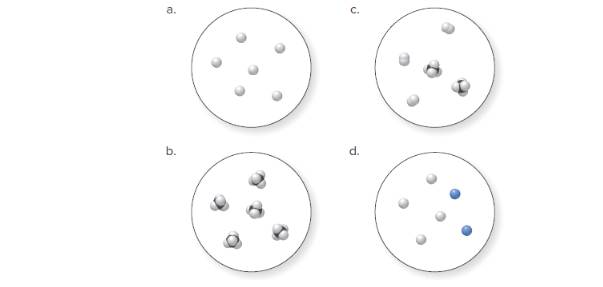
(a)
Interpretation:
Whether the given molecular art is a pure compound, mixture, or a pure element needs to be classified.

Concept Introduction:
An element is known as the pure substance that cannot be broken down further using chemical methods. The methods are such as electrolysis, cooling, heating, and reactions with other chemical substances.
A compound is known as the pure substance which is made up of more than two different atoms that are bonded chemically to one another. Using chemical methods, a compound can be destroyed. It can be broken down into simpler compounds or into its elements.
A mixture is the combination of more than two dissimilar elemental substances or compounds. The mixture is not a pure substance, but it is the combination of different atoms of elements. Mixtures are of two kinds, Heterogenous and Homogeneous.
Answer to Problem 19P
Molecular art 'a' − Pure element.
Explanation of Solution
As per the definitions of element, compound and mixture:
The pure element is the substance that doesn't separate into simple substances through the chemical procedures.
A pure compound is a substance formed by combinations of two or more elements.
A mixture is made up of the combination of one or more components with several compositions.
Now, based on these definitions, molecular art (a) signifies pure elements.
(b)
Interpretation:
Whether the below molecular art is pure compound, mixture, or a pure element needs to be determined.
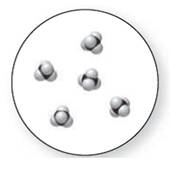
Concept Introduction:
An element is known as the pure substance that cannot be broken down further using chemical methods. The methods are such as electrolysis, cooling, heating, and reactions with other chemical substances.
A compound is known as the pure substance which is made up of more than two different atoms that are bonded chemically to one another. Using chemical methods, a compound can be destroyed. It can be broken down into simpler compounds or into its elements.
A mixture is the combination of more than two dissimilar elemental substances or compounds. The mixture is not a pure substance, but it is the combination of different atoms of elements. Mixtures are of two kinds, Heterogenous and Homogeneous.
Answer to Problem 19P
Molecular art 'b' − Pure compounds
Explanation of Solution
As per the definitions of element, compound and mixture:
The pure element is the substance that doesn't separate into simple substances through the chemical procedures.
A pure compound is a substance formed by combinations of two or more elements.
A mixture is made up of the combination of one or more components with several compositions.
Now based on these definitions, molecular art (b) signifies pure compounds.
(c)
Interpretation:
Whether the below compound is a pure compound, mixture, or a pure element needs to be determined.
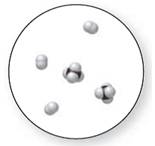
Concept Introduction:
An element is known as the pure substance that cannot be broken down further using chemical methods. The methods are such as electrolysis, cooling, heating, and reactions with other chemical substances.
A compound is known as the pure substance which is made up of more than two different atoms that are bonded chemically to one another. Using chemical methods, a compound can be destroyed. It can be broken down into simpler compounds or into its elements.
A mixture is the combination of more than two dissimilar elemental substances or compounds. The mixture is not a pure substance, but it is the combination of different atoms of elements. Mixtures are of two kinds, Heterogenous and Homogeneous.
Answer to Problem 19P
Molecular art 'c' − Mixture
Explanation of Solution
As per the definitions of element, compound and mixture:
The pure element is the substance that doesn't separate into simple substances through the chemical procedures.
A pure compound is a substance formed by combinations of two or more elements.
A mixture is made up of the combination of one or more components with several compositions.
Now based on these definitions, molecular art (c) signifies mixture.
(c)
Interpretation:
Whether the below compound is a pure compound, mixture, or a pure element needs to be determined.
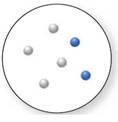
Concept Introduction:
An element is known as the pure substance that cannot be broken down further using chemical methods. The methods are such as electrolysis, cooling, heating, and reactions with other chemical substances.
A compound is known as the pure substance which is made up of more than two different atoms that are bonded chemically to one another. Using chemical methods, a compound can be destroyed. It can be broken down into simpler compounds or into its elements.
A mixture is the combination of more than two dissimilar elemental substances or compounds. The mixture is not a pure substance, but it is the combination of different atoms of elements. Mixtures are of two kinds, Heterogenous and Homogeneous.
Answer to Problem 19P
Mixture
Explanation of Solution
As per the definitions of element, compound and mixture:
The pure element is the substance that doesn't separate into simple substances through the chemical procedures.
A pure compound is a substance formed by combinations of two or more elements.
A mixture is made up of the combination of one or more components with several compositions.
Now, based on these definitions, molecular art (d) signifies mixture.
Want to see more full solutions like this?
Chapter 1 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
Additional Science Textbook Solutions
Anatomy & Physiology (6th Edition)
Human Biology: Concepts and Current Issues (8th Edition)
Campbell Essential Biology with Physiology (5th Edition)
Organic Chemistry
Campbell Biology (11th Edition)
- AN IR spectrum, a 13 CMR spectrum, and a 1 HMR spectrum were obtained for an unknown structure with a molecular formula of C9H10. Draw the structure of this compound.arrow_forwardAN IR spectrum, a 13 CMR spectrum, and a 1 HMR spectrum were obtained for an unknown structure with a molecular formula of C9H10. Draw the structure of this compound.arrow_forward(a) What is the hybridization of the carbon in the methyl cation (CH3*) and in the methyl anion (CH3¯)? (b) What is the approximate H-C-H bond angle in the methyl cation and in the methyl anion?arrow_forward
- Q8: Draw the resonance structures for the following molecule. Show the curved arrows (how you derive each resonance structure). Circle the major resonance contributor.arrow_forwardQ4: Draw the Lewis structures for the cyanate ion (OCN) and the fulminate ion (CNO). Draw all possible resonance structures for each. Determine which form for each is the major resonance contributor.arrow_forwardIn the following molecule, indicate the hybridization and shape of the indicated atoms. CH3 N CH3 HÖ: H3C CI: ::arrow_forward
- Q3: Draw the Lewis structures for nitromethane (CH3NO2) and methyl nitrite (CH3ONO). Draw at least two resonance forms for each. Determine which form for each is the major resonance contributor.arrow_forwardQ1: Draw a valid Lewis structures for the following molecules. Include appropriate charges and lone pair electrons. If there is more than one Lewis structure available, draw the best structure. NH3 Sulfate Boron tetrahydride. C3H8 (linear isomer) OCN NO3 CH3CN SO2Cl2 CH3OH2*arrow_forwardQ2: Draw all applicable resonance forms for the acetate ion CH3COO. Clearly show all lone pairs, charges, and arrow formalism.arrow_forward
- Please correct answer and don't used hand raitingarrow_forward9. The following reaction, which proceeds via the SN1/E1 mechanisms, gives three alkene products (A, B, C) as well as an ether (D). (a) Show how each product arises mechanistically. (b) For the alkenes, determine the major product and justify your answer. (c) What clues in the reaction as shown suggest that this reaction does not go by the SN2/E2 mechanism route? (CH3)2CH-CH-CH3 CH3OH 1 Bl CH3OH ⑧· (CH3)2 CH-CH=CH2 heat H ⑥③ (CH3)2 C = C = CH3 © СнЗ-С-Снаснз сна (CH 3 ) 2 C H G H CH 3 оснзarrow_forwardPlease Don't used hand raitingarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





