
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
4th Edition
ISBN: 9781260562620
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 23P
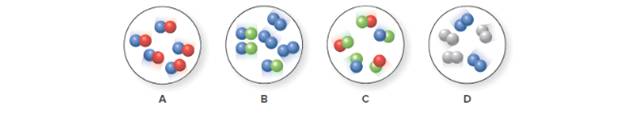
(a) Which representation(s) illustrate a mixture of two elements? (b) Which representation(s) illustrate a mixture of a compound and an element?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used hand raiting and don't used Ai solution
(9 Pts) In one of the two Rare Earth element rows of the periodic table, identify an exception tothe general ionization energy (IE) trend. For the two elements involved, answer the followingquestions. Be sure to cite sources for all physical data that you use.a. (2 pts) Identify the two elements and write their electronic configurations.b. (2 pts) Based on their configurations, propose a reason for the IE trend exception.c. (5 pts) Calculate effective nuclear charges for the last electron in each element and theAllred-Rochow electronegativity values for the two elements. Can any of these valuesexplain the IE trend exception? Explain how (not) – include a description of how IErelates to electronegativity.
Don't used hand raiting and don't used Ai solution
Chapter 1 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
Ch. 1.1 - Prob. 1.1PCh. 1.2 - Characterize each process as a physical change or...Ch. 1.2 - Does the molecular art represent a chemical change...Ch. 1.3 - Classify each example of molecular art as a pure...Ch. 1.3 - (a) Which representation(s) of molecular art...Ch. 1.3 - Classify each item as a pure substance or a...Ch. 1.3 - Classify each item as an element or a compound:...Ch. 1.4 - What term is used for each of the following units:...Ch. 1.4 - If a nanometer is one-billionth of a meter...Ch. 1.4 - Prob. 1.8P
Ch. 1.4 - Using the prefixes in Table 1.2, determine which...Ch. 1.5 - How many significant figures does each number...Ch. 1.5 - Indicate whether each zero in the following...Ch. 1.5 - Round off each number in Sample Problem 1.4 to two...Ch. 1.5 - Carry out each caluction and give the answer using...Ch. 1.5 - Carry out each calculation and give the answer...Ch. 1.6 - Write each number in scientific notation. 93,200...Ch. 1.6 - Lab results for a routine check-up showed an...Ch. 1.6 - Convert each number to its standard form. 6.5103...Ch. 1.7 - Write two conversion factors for each pair of...Ch. 1.7 - Carry out each of the following conversions. 25 L...Ch. 1.7 - On a flight from Bangkok, Thailand, to Taipei,...Ch. 1.7 - What is the volume of liquid contained in the...Ch. 1.7 - Carry out each of the following conversions. 6,250...Ch. 1.8 - A patient is prescribed 0.100 mg of a drug that is...Ch. 1.8 - Prob. 1.13PPCh. 1.8 - Prob. 1.14PCh. 1.9 - Prob. 1.14PPCh. 1.9 - Prob. 1.15PCh. 1.10 - How does the mass of liquid A in cylinder [1]...Ch. 1.10 - Calculate the mass in grams of 10.0 mL of diethyl...Ch. 1.10 - If a 120-lb woman uses five 2.0lb lead weights in...Ch. 1.10 - If a household recycles 10.5 kg of plastic bottles...Ch. 1.10 - If the density of a liquid is 0.80 g/mL, what is...Ch. 1 - Classify each example of molecular art as a pure...Ch. 1 - (a) Which representation(s) in Problem 1.19...Ch. 1 - Label each component in the molecular art as an...Ch. 1 - Label each component in the molecular art as an...Ch. 1 - (a) Which representation(s) illustrate a mixture...Ch. 1 - Label each of the following conversions as a...Ch. 1 - Classify each process as a chemical or physical...Ch. 1 - Classify each process as a chemical or physical...Ch. 1 - When a chunk of dry ice (solid carbon dioxide) is...Ch. 1 - The inexpensive preparation of nitrogen-containing...Ch. 1 - What is the temperature on the given Fahrenheit...Ch. 1 - a. What is the length of the given crayon in...Ch. 1 - Label each quantity as an exact or inexact number....Ch. 1 - Label each quantity as an exact or inexact number....Ch. 1 - Which quantity in each pair is larger? 5 mL or 5...Ch. 1 - Which quantity in each pair is larger? 10 km or 10...Ch. 1 - How many significant figures does each number...Ch. 1 - Prob. 36PCh. 1 - Prob. 37PCh. 1 - Prob. 38PCh. 1 - Prob. 39PCh. 1 - Prob. 40PCh. 1 - Prob. 41PCh. 1 - Prob. 42PCh. 1 - Prob. 43PCh. 1 - Prob. 44PCh. 1 - Prob. 45PCh. 1 - Rank the numbers in each group from smallest to...Ch. 1 - Prob. 47PCh. 1 - Prob. 48PCh. 1 - Prob. 49PCh. 1 - What is the volume of liquid contained in the...Ch. 1 - Prob. 51PCh. 1 - (a) If there are 15 mL in one tablespoon, how many...Ch. 1 - The concentration of mercury, a toxi pollutant, in...Ch. 1 - Prob. 54PCh. 1 - Prob. 55PCh. 1 - Prob. 56PCh. 1 - Prob. 57PCh. 1 - Prob. 58PCh. 1 - Prob. 59PCh. 1 - Prob. 60PCh. 1 - Which temperature in each pair is higher? a....Ch. 1 - Prob. 62PCh. 1 - Prob. 63PCh. 1 - Draw a graduated cylinder similar to Problem 1.63...Ch. 1 - The given beaker contains 100 mL of water. Draw an...Ch. 1 - Prob. 66PCh. 1 - Prob. 67PCh. 1 - Prob. 68PCh. 1 - If milk has a density of 1.03 g/mL, what is the...Ch. 1 - Prob. 70PCh. 1 - Prob. 71PCh. 1 - Prob. 72PCh. 1 - A lab test showed an individual’s chloesterol leve...Ch. 1 - Hemoglobin is a protein that transports oxygen...Ch. 1 - Prob. 75PCh. 1 - A soccer weighed 70.7 kg before a match, drank 1.8...Ch. 1 - Liposuction is a cosmetic procedure used to remove...Ch. 1 - A single 1-oz serving of tortilla chips contains...Ch. 1 - Prob. 79PCh. 1 - Prob. 80PCh. 1 - Prob. 81PCh. 1 - Prob. 82PCh. 1 - Prob. 83PCh. 1 - Prob. 84PCh. 1 - Often the specific amount of a drug to be...Ch. 1 - Prob. 86CPCh. 1 - Prob. 87CPCh. 1 - Prob. 88CPCh. 1 - Prob. 89CPCh. 1 - Prob. 90CPCh. 1 - Prob. 91CPCh. 1 - If the proper dosage of a medication is 10g/kg of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Give the IUPAC name for each compound.
Organic Chemistry
11. In the early 1800s, French naturalist Jean Baptiste Lamarck suggested that the best explanation for the rel...
Campbell Biology: Concepts & Connections (9th Edition)
An obese 55-year-old woman consults her physician about minor chest pains during exercise. Explain the physicia...
Biology: Life on Earth with Physiology (11th Edition)
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- please solve this, and help me know which boxes to check. Thank you so much in advance.arrow_forwardElectronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Describe how electronegativity is illustrated on the periodic table including trends between groups and periods and significance of atom size.arrow_forwardDefine the term “transition.” How does this definition apply to the transition metals?arrow_forward
- Describe how the properties of the different types of elements (metals, nonmetals, metalloids) differ.arrow_forwardUse a textbook or other valid source to research the physical and chemical properties of each element listed in Data Table 1 using the following as a guideline: Ductile (able to be deformed without losing toughness) and malleable (able to be hammered or pressed permanently out of shape without breaking or cracking) or not ductile or malleable Good, semi, or poor conductors of electricity and heat High or low melting and boiling points Occur or do not occur uncombined/freely in nature High, intermediate, or low reactivity Loses or gains electrons during reactions or is not reactivearrow_forwardProvide the Physical and Chemical Properties of Elements of the following elements listedarrow_forward
- Questions 4 and 5arrow_forwardFor a titration of 40.00 mL of 0.0500 M oxalic acid H2C2O4 with 0.1000 M KOH, calculate the pH at each of the following volume of KOH used in the titration: 1) before the titration begin;2) 15 mL; 3) 20 mL; 4) 25 mL; 5) 40 mL; 6) 50 mL. Ka1 = 5.90×10^-2, Ka2 = 6.50×10^-5 for oxalic acid.arrow_forwardPredict the major organic product(s), if any, of the following reactions. Assume all reagents are in excess unless otherwise indicated.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Types of Matter: Elements, Compounds and Mixtures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=dggHWvFJ8Xs;License: Standard YouTube License, CC-BY