
Concept explainers
17-9 Answer true or false.
(a) The one aldehyde and the one ketone with a molecular formula of C3H6O are constitutional isomers.
(b)
(c) The VSEPR model predicts bond angles of 1200 about the carbonyl carbon of aldehydes and ketones.
(d) The carbonyl carbon of a ketone is a stereocenter.
(a)
Interpretation:
Answer true or false for the following statement.
The one aldehyde and one ketone with a molecular formula of
Concept Introduction:
Constitutional isomers have the same molecular formula but different structural formulas or connectivity with atoms is different. In aldehydes, the carbonyl group is bonded to a hydrogen atom and ketones carbonyl group is bonded to two carbon atoms.
Answer to Problem 17.9P
The one aldehyde and one ketone with a molecular formula of
Explanation of Solution
The given molecular formula:
For this molecular formula we can draw both aldehyde and ketone. They are as follows.
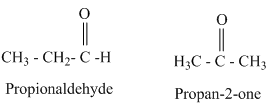
From the above structures aldehydes and ketones having same molecular formula are constitutional isomers.
Therefore, the given statement is true.
(b)
Interpretation:
Answer true or false for the following statement.
Aldehydes and ketones both contain a carbonyl group.
Concept Introduction:
In aldehydes, the carbonyl group is bonded to a hydrogen atom and ketones carbonyl group is bonded to two carbon atoms.
Answer to Problem 17.9P
Aldehydes and ketones both contain a carbonyl group is true statement.
Explanation of Solution
In aldehydes, the carbonyl group is bonded to a hydrogen atom and in ketone carbonyl group is bonded to two carbon atoms. Hence, both an aldehyde and a ketone have a carbonyl group.

Where, R is an alkyl group.
Therefore, the given statement is true.
(c)
Interpretation:
Answer true or false for the following statement.
The VSEPR model predicts bond angles of 1200 about the carbonyl carbon of aldehydes and ketones.
Concept Introduction:
In aldehydes, the carbonyl group is bonded to a hydrogen atom and ketones carbonyl group is bonded to two carbon atoms. VSEPR model predicts the geometry of the molecule with the help of number of pair of electrons present around the central atoms and bond angle of the molecule.
Answer to Problem 17.9P
The VSEPR model predicts bond angles of 1200 about the carbonyl carbon if aldehydes and ketones is the true statement.
Explanation of Solution
In aldehydes, the carbonyl group is bonded to a hydrogen atom and ketones carbonyl group is bonded to two carbon atoms. The carbonyl group of aldehyde and ketone are represented are as follows.
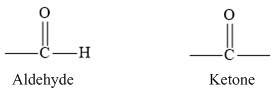
According to VSEPR theory, three pair of electron groups forms a Trigonal planar geometry with bond angle of 1200.
Therefore, the given statement is true.
(d)
Interpretation:
Answer true or false for the following statement.
The carbonyl carbon of ketone is a stereocentre.
Concept Introduction:
A stereocentre is a tetrahedral carbon atom in which four different groups are attached to it. When an atom generally carbon is linked with four different groups, then that centre is known as chiral centre.
Answer to Problem 17.9P
The carbonyl carbon of ketone is a stereocenter is the false statement.
Explanation of Solution
In aldehydes, the carbonyl group is bonded to a hydrogen atom and in ketone carbonyl group is bonded to two carbon atoms. The carbonyl group of aldehyde and ketone are represented are as follows.

In ketones, the carbonyl carbon is boded to carbonyl oxygen by a double bond group and bonded to two other carbon atoms.
Therefore, the given statement is false.
Want to see more full solutions like this?
Chapter 17 Solutions
Bundle: Introduction to General, Organic and Biochemistry, 11th + OWLv2, 4 terms (24 months) Printed Access Card
- Molecular ion peak: the peak corresponding to the intact morecure (with a positive charge) 4. What would the base peak and Molecular ion peaks when isobutane is subjected to Mass spectrometry? Draw the structures and write the molecular weights of the fragments. 5. Circle most stable cation a) tert-butyl cation b) Isopropyl cation c) Ethyl cation. d)Methyl cationarrow_forwardHow many arrangements are there of 15 indistinguishable lattice gas particles distributed on: a.V = 15 sites b.V = 16 sites c.V = 20 sitesarrow_forwardFor which element is the 3d subshell higher in energy than that 4s subshell? Group of answer choices Zr Ca V Niarrow_forward
- ii) Molecular ion peak :the peak corresponding to the intact molecule (with a positive charge) What would the base peak and Molecular ion peaks when isobutane is subjected to Mass spectrometry? Draw the structures and write the molecular weights of the fragments. Circle most stable cation a) tert-butyl cation b) Isopropyl cation c) Ethyl cation. d) Methyl cation 6. What does a loss of 15 represent in Mass spectrum? a fragment of the molecule with a mass of 15 atomic mass units has been lost during the ionization Process 7. Write the isotopes and their % abundance of isotopes of i) Clarrow_forwardChoose a number and match the atomic number to your element on the periodic table. For your element, write each of these features on a side of your figure. 1. Element Name and symbol 2. Family and group 3. What is it used for? 4. Sketch the Valence electron orbital 5. What ions formed. What is it's block on the periodic table. 6. Common compounds 7. Atomic number 8. Mass number 9. Number of neutrons- (show calculations) 10. Sketch the spectral display of the element 11.Properties 12. Electron configuration 13. Submit a video of a 3-meter toss in slow-moarrow_forward[In this question, there are multiple answers to type in a "fill-in-the-blank" fashion - in each case, type in a whole number.] Consider using Slater's Rules to calculate the shielding factor (S) for the last electron in silicon (Si). There will be electrons with a 0.35 S-multiplier, electrons with a 0.85 S-multiplier, and electrons with a 1.00 S-multiplier.arrow_forward
- Provide the unknown for the given data.arrow_forwardDraw the Lewis structures of two methanol (CH3OH) molecules and depict hydrogenbonding between them with dashed lines. Show all lone pairs. Provide a thorough analysis to apply concept idea into other problems.arrow_forwardSteps and explanation please.arrow_forward
- How could you distinguish between each pair of compounds below using IR? For each pair citeone bond and it’s frequency that you could use to distinguish between them. Please provide thorough analysis to apply into further problems.arrow_forwardSteps and explanation please.arrow_forwardSteps and explanation on how to solve.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning




