
Bundle: Introduction to General, Organic and Biochemistry, 11th + OWLv2, 4 terms (24 months) Printed Access Card
11th Edition
ISBN: 9781305705159
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.13P
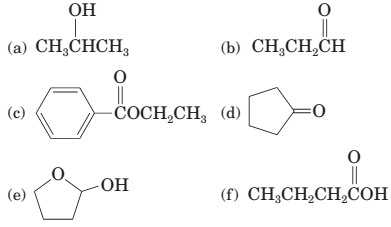
17-13 Which compounds contain carbonyl groups?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write structural formulas for the major products by
doing addition reactions
1. You must add H2 as Pt is catalyst it does not take part in reactions
only speed up the process
H₂
CH2=CH-CH3
Pt
2. Add HCI break it into H and Cl
CH3
HCI
3. Add Br2 only CC14 is catalyst
CH3-CH=CH2
B12
CCl4
4. Add water to this and draw major product, H2SO4 is catalyst you have add
water H20 in both the reaction below
H₂SO4
CH3-CH=CH2
CH3
H2SO4/H₂O
CH3-C=CH2
reflux
?
Plan the synthesis of the following compound using the starting
material provided and any other reagents needed as long as
carbon based reagents have 3 carbons or less. Either the
retrosynthesis or the forward synthesis (mechanisms are not
required but will be graded if provided) will be accepted if all
necessary reagents and intermediates are shown (solvents and
temperature requirements are not needed unless specifically
involved in the reaction, i.e. DMSO in the Swern oxidation or
heat in the KMnO4 oxidation).
H
H
Hint These are benzene substitution reactions.
ALCI3 and UV light are catalyst no part in reactions and triangle A means
heating.
A. Add ethyl for Et in benzene ring alkylation reaction EtCl =
CH3CH2CL
1) EtC1 / AlCl3 / A
?
B: Add Br to benzene ring ( substitution)
2) Br₂ / uv light
?
C Add (CH3)2 CHCH2 in benzene ring ( substitution)
(CH3)2CHCH,C1 / AICI,
?
Chapter 17 Solutions
Bundle: Introduction to General, Organic and Biochemistry, 11th + OWLv2, 4 terms (24 months) Printed Access Card
Ch. 17.2 - Problem 17-1 Wrtie the IUPAC name for each...Ch. 17.2 - Prob. 17.2PCh. 17.2 - Prob. 17.3PCh. 17.4 - Prob. 17.4PCh. 17.4 - Prob. 17.5PCh. 17.4 - Problem 17-6 Show the reaction of benzaldehyde...Ch. 17.4 - Problem 17-7 Identify all hemiacetals and acetals...Ch. 17.5 - Prob. 17.8PCh. 17 - 17-9 Answer true or false. (a) The one aldehyde...Ch. 17 - Prob. 17.10P
Ch. 17 - 17-11 What is the difference in structure between...Ch. 17 - 17-12 Is it possible for the carbon atom of a...Ch. 17 - 17-13 Which compounds contain carbonyl groups?Ch. 17 - 17-14 Following are structural formulas for two...Ch. 17 - 17-15 Draw structural formulas for the four...Ch. 17 - Prob. 17.16PCh. 17 - Prob. 17.17PCh. 17 - 17-18 Draw structural formulas for these ketones....Ch. 17 - 17-19 Write the JUPAC names for these compounds.Ch. 17 - Prob. 17.20PCh. 17 - 17-2 1 Explain why each name is incorrect. Write...Ch. 17 - Prob. 17.22PCh. 17 - Prob. 17.23PCh. 17 - 17-24 In each pair of compounds, select the one...Ch. 17 - Prob. 17.25PCh. 17 - 17-26 Account for the fact that acetone has a...Ch. 17 - 17-27 Pentane, 1-butanol, and butanal all have...Ch. 17 - 17-28 Show how acetaldehyde can form hydrogen...Ch. 17 - 17-29 Why can’t two molecules of acetone form a...Ch. 17 - 17-30 Answer true or false. (a) The reduction of...Ch. 17 - 17-3 1 Draw a structural formula for the principal...Ch. 17 - Prob. 17.32PCh. 17 - 17-33 What simple chemical test could you use to...Ch. 17 - 17-34 Explain why liquid aldehydes are often...Ch. 17 - 17-35 Suppose that you take a bottle of...Ch. 17 - 17-36 Explain why the reduction of an aldehyde...Ch. 17 - Prob. 17.37PCh. 17 - Prob. 17.38PCh. 17 - Prob. 17.39PCh. 17 - Prob. 17.40PCh. 17 - Prob. 17.41PCh. 17 - Prob. 17.42PCh. 17 - Prob. 17.43PCh. 17 - Prob. 17.44PCh. 17 - Prob. 17.45PCh. 17 - Prob. 17.46PCh. 17 - 17-47 What is the characteristic structural...Ch. 17 - Prob. 17.48PCh. 17 - Prob. 17.49PCh. 17 - Prob. 17.50PCh. 17 - Prob. 17.51PCh. 17 - Prob. 17.52PCh. 17 - Prob. 17.53PCh. 17 - 17-54 Following is the structure of...Ch. 17 - Prob. 17.55PCh. 17 - Prob. 17.56PCh. 17 - Prob. 17.57PCh. 17 - Prob. 17.58PCh. 17 - Prob. 17.59PCh. 17 - 17-60 1-Propanol can be prepared by the reduction...Ch. 17 - Prob. 17.61PCh. 17 - 17-62 Show how to bring about these conversions....Ch. 17 - Prob. 17.63PCh. 17 - Prob. 17.64PCh. 17 - Prob. 17.65PCh. 17 - Prob. 17.66PCh. 17 - 17-67 Draw structural formulas for these...Ch. 17 - Prob. 17.68PCh. 17 - 17-69 Propanal (bp 49°C) and 1-propanol (bp 97°C)...Ch. 17 - 17-70 What simple chemical test could you use to...Ch. 17 - Prob. 17.71PCh. 17 - 17-72 The following molecule is an enediol; each...Ch. 17 - 17-73 Alcohols can be prepared by the...Ch. 17 - 17-74 Glucose, C6H12O6, contains an aldehyde group...Ch. 17 - Prob. 17.75PCh. 17 - Prob. 17.76PCh. 17 - Prob. 17.77PCh. 17 - 17-78 Complete the following equation for these...Ch. 17 - 17-79 Write an equation for each conversion. (a)...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the mechanism to make the alcohol 2-hexanol. Draw the Mechanism to make the alcohol 1-hexanol.arrow_forwardDraw the mechanism for the formation of diol by starting with 1-pentanal in... basic conditions then acidic conditions then draw the mechanism for the formation of a carboxylic acid from your product.arrow_forwardIdentify each chiral carbon as either R or S. Identify the overall carbohydrates as L or Darrow_forward
- Ethers can be formed via acid-catalyzed acetal formation. Draw the mechanism for the molecule below and ethanol.arrow_forwardHOCH, H HO CH-OH OH H OH 11 CH₂OH F II OH H H 0 + H OHarrow_forwardDraw the mechanism for the formation of diol by starting with one pen and all in... basic conditions then acidic conditions then draw the mechanism for the formation of a carboxylic acid from your product.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY