
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
9th Edition
ISBN: 9781337598255
Author: Spencer L. Seager
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17, Problem 17.9E
Which of the following molecules can have enantiomers? Identify any chiral carbon atoms.
a. 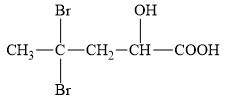
b. 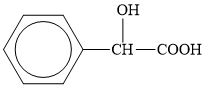
c. 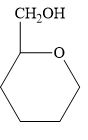
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Describe the structural differences between iso- and heteropolyacids.
What is the pH of the Tris buffer after the addition of 10 mL of 0.01M NaOH? How would I calculate this?
Why do isopolianions form polymeric species with a defined molecular weight? What does it depend on?
Chapter 17 Solutions
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
Ch. 17 - Prob. 17.1ECh. 17 - Describe whether each of the following substances...Ch. 17 - Prob. 17.3ECh. 17 - Prob. 17.4ECh. 17 - Prob. 17.5ECh. 17 - Why are carbon atoms 1 and 3 of glyceraldehyde not...Ch. 17 - Prob. 17.7ECh. 17 - Which of the following molecules can have...Ch. 17 - Which of the following molecules can have...Ch. 17 - Prob. 17.10E
Ch. 17 - Prob. 17.11ECh. 17 - Prob. 17.12ECh. 17 - Prob. 17.13ECh. 17 - Draw Fischer projections for both the D and L...Ch. 17 - Prob. 17.15ECh. 17 - Prob. 17.16ECh. 17 - Prob. 17.17ECh. 17 - Prob. 17.18ECh. 17 - Prob. 17.19ECh. 17 - Prob. 17.20ECh. 17 - Prob. 17.21ECh. 17 - Prob. 17.22ECh. 17 - Prob. 17.23ECh. 17 - Prob. 17.24ECh. 17 - Prob. 17.25ECh. 17 - Prob. 17.26ECh. 17 - Prob. 17.27ECh. 17 - Prob. 17.28ECh. 17 - Prob. 17.29ECh. 17 - Prob. 17.30ECh. 17 - Prob. 17.31ECh. 17 - Prob. 17.32ECh. 17 - Prob. 17.33ECh. 17 - Prob. 17.34ECh. 17 - Prob. 17.35ECh. 17 - Prob. 17.36ECh. 17 - Prob. 17.37ECh. 17 - Prob. 17.38ECh. 17 - Prob. 17.39ECh. 17 - Prob. 17.40ECh. 17 - Prob. 17.41ECh. 17 - Prob. 17.42ECh. 17 - Prob. 17.43ECh. 17 - Prob. 17.44ECh. 17 - Prob. 17.45ECh. 17 - Prob. 17.46ECh. 17 - Prob. 17.47ECh. 17 - Sucrose and honey are commonly used sweeteners....Ch. 17 - Prob. 17.49ECh. 17 - Prob. 17.50ECh. 17 - Prob. 17.51ECh. 17 - Prob. 17.52ECh. 17 - Prob. 17.53ECh. 17 - Prob. 17.54ECh. 17 - Prob. 17.55ECh. 17 - Prob. 17.56ECh. 17 - Prob. 17.57ECh. 17 - Prob. 17.58ECh. 17 - Prob. 17.59ECh. 17 - Prob. 17.60ECh. 17 - Prob. 17.61ECh. 17 - Prob. 17.62ECh. 17 - Prob. 17.63ECh. 17 - Prob. 17.64ECh. 17 - Prob. 17.65ECh. 17 - Prob. 17.66ECh. 17 - Prob. 17.67ECh. 17 - Prob. 17.68ECh. 17 - Prob. 17.69ECh. 17 - Prob. 17.70ECh. 17 - Prob. 17.71ECh. 17 - Prob. 17.72ECh. 17 - Prob. 17.73ECh. 17 - Glucose is a reducing sugar, which if boiled in...Ch. 17 - Prob. 17.75E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What are isopolianions? Describe the structural unit of isopolianions.arrow_forwardJustify the polymerization of vanadates VO43-, as a function of concentration and pH.arrow_forwardWhat is the preparation of 500 mL of 100mM MOPS buffer (pH=7.5) starting with 1 M MOPS and 1 M NaOH? How would I calculate the math?arrow_forward
- Indicate the correct option.a) Isopolianions are formed around metallic atoms in a low oxidation state.b) Non-metals such as N, S, C, Cl, ... give rise to polyacids (oxygenated).c) Both are incorrect.arrow_forward14. Which one of the compounds below is the major organic product obtained from the following series of reactions? Br OH OH CH3O™ Na+ H*, H₂O SN2 HO OH A B C D 0 Earrow_forwardWavelength (nm) I'm not sure what equation I can come up with other than the one generated with my graph. Can you please show me the calculations that were used to find this equation? Give an equation that relates energy to wavelength. Explain how you arrived at your equation. Wavelength Energy (kJ/mol) (nm) 350 341.8 420 284.8 470 254.5 530 225.7 580 206.3 620 192.9 700 170.9 750 159.5 Energy vs. Wavelength (Graph 1) 400 350 y=-0.4367x+470.82 300 250 200 150 100 50 O 0 100 200 300 400 500 600 700 800 Energy (kJ/mol)arrow_forward
- 5. Draw molecular orbital diagrams for superoxide (O2¯), and peroxide (O2²-). A good starting point would be MO diagram for O2 given in your textbook. Then: a) calculate bond orders in superoxide and in peroxide; indicate which species would have a stronger oxygen-oxygen bond; b) indicate which species would be a radical. (4 points)arrow_forward16. Which one of the compunds below is the final product of the reaction sequence shown here? عملاء .OH Br. (CH3)2CH-C=C H+,H,O 2 mol H2, Pt A OH B OH D OH E OH C OHarrow_forwardIndicate whether any of the two options is correct.a) The most common coordination structure for isopolianions is the prismb) Heteropolianions incorporate alkaline cations into their structuresarrow_forward
- Please correct answer and don't use hand ratingarrow_forwardWavelength (nm) I'm not sure what equation I can come up with other than the one generated with my graph. Can you please show me the calculations that were used to find this equation? Give an equation that relates energy to wavelength. Explain how you arrived at your equation. Wavelength Energy (kJ/mol) (nm) 350 341.8 420 284.8 470 254.5 530 225.7 580 206.3 620 192.9 700 170.9 750 159.5 Energy vs. Wavelength (Graph 1) 400 350 y=-0.4367x+470.82 300 250 200 150 100 50 O 0 100 200 300 400 500 600 700 800 Energy (kJ/mol)arrow_forward6. For the following molecules: draw Lewis dot-structures; use VSEPR method to determine geometries of the following molecules/ions. Are the central atoms in these molecules/ions considered of normal valency, or are they hypervalent? (please read paragraph 2.6) a) BrF3 (6 points) b) BrF4 c) IF₂ 4arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY