
Concept explainers
(a)
Interpretation:
Whether table sugar is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
Table sugar is a disaccharide.
Explanation of Solution
Table sugar is also known as sucrose. Sucrose on hydrolysis gives two monosaccharides, glucose and fructose which are linked through a glycosidic linkage. This shows that sucrose or table sugar is a disaccharide.
Table sugar is a disaccharide.
(b)
Interpretation:
Whether the given carbohydrate is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy aldehydes or ketones containing only one aldehyde or ketone unit. For example: glucose and fructose.
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
The given carbohydrate is an example of monosaccharide.
Explanation of Solution
The structural formula of the given carbohydrate is shown below.
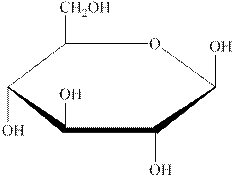
Figure 1
The above structure of carbohydrate shows that there is no glycosidic linkage through which another carbohydrate is linked. Therefore, the above carbohydrate is a monosaccharide.
The carbohydrate shown in Figure 1 is an example of monosaccharide.
(c)
Interpretation:
Whether starch is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy aldehydes or ketones containing only one aldehyde or ketone unit. For example: glucose and fructose.
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
Starch is a polysaccharide.
Explanation of Solution
Starch is the
Starch is an example of polysaccharide.
(d)
Interpretation:
Whether fructose is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy aldehydes or ketones containing only one aldehyde or ketone unit. For example: glucose and fructose.
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
Fructose is a monosaccharide.
Explanation of Solution
Hydrolysis of sucrose produces glucose and fructose which do not undergo further hydrolysis. Therefore, fructose comes under the category of monosaccharide.
Fructose is an example of monosaccharide.
(e)
Interpretation:
Whether cellulose is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy aldehydes or ketones containing only one aldehyde or ketone unit. For example: glucose and fructose.
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
Cellulose is a polysaccharide.
Explanation of Solution
Cellulose is a polymeric form of D-glucose because cellulose on hydrolysis gives a large number of glucose as monosaccharide. Each glucose is linked with
Cellulose on hydrolysis gives a large number of molecules of glucose. As a result, cellulose is a polysaccharide.
(f)
Interpretation:
Whether the given carbohydrate is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy aldehydes or ketones containing only one aldehyde or ketone unit. For example: glucose and fructose.
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
The given carbohydrate is a disaccharide.
Explanation of Solution
The structural formula of the given carbohydrate is shown below.
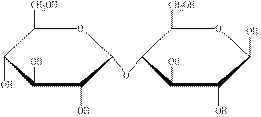
Figure 2
The above structure of carbohydrate shows that two monosaccharides are linked to each other through glycosidic linkage and results in the formation of a disaccharide. Therefore, the above carbohydrate is a disaccharide.
The carbohydrate shown in Figure 2 is an example of disaccharide.
(g)
Interpretation:
Whether glycogen is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy aldehydes or ketones containing only one aldehyde or ketone unit. For example: glucose and fructose.
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
Glycogen is a polysaccharide.
Explanation of Solution
Glycogen is a polymeric form of D-glucose which on hydrolysis gives glucose units. Each glucose unit is linked by
Glycogen on hydrolysis gives a large number of molecules of glucose. As a result, glycogen is a polysaccharide.
(h)
Interpretation:
Whether amulose is a monosaccharide, disaccharide, or polysaccharide is to be stated.
Concept introduction:
On the basis of the structural differences carbohydrates are classified into three parts.
Monosaccharide: They are the polyhydroxy aldehydes or ketones containing only one aldehyde or ketone unit. For example: glucose and fructose.
Disaccharide: They are the polyhydroxy aldehydes or ketones containing two aldehyde or ketone units. For example: Sucrose, lactose.
Polysaccharide: They are the polyhydroxy aldehydes or ketones containing more than two aldehyde or ketone units. For example: Cellulose, starch.
Answer to Problem 17.4E
Amylose is a polysaccharide.
Explanation of Solution
Amylose is a polymeric form of
Amylose on hydrolysis gives a large number of glucose units. As a result, amylose is a polysaccharide.
Want to see more full solutions like this?
Chapter 17 Solutions
Chemistry for Today: General, Organic, and Biochemistry
- X 5 Check the box under each molecule that has a total of five ẞ hydrogens. If none of the molecules fit this description, check the box underneath the table. CI Br Br Br 0 None of these molecules have a total of five ẞ hydrogens. Explanation Check esc F1 F2 tab caps lock fn Q @2 A W # 3 OH O OH HO © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility IK F7 F7 F8 TA F9 F10 & 6 28 * ( > 7 8 9 0 80 F3 O F4 KKO F5 F6 S 64 $ D % 25 R T Y U பட F G H O J K L Z X C V B N M H control option command P H F11 F12 + || { [ command optionarrow_forwardAn open vessel containing water stands in a laboratory measuring 5.0 m x 5.0 m x 3.0 m at 25 °C ; the vapor pressure (vp) of water at this temperature is 3.2 kPa. When the system has come to equilibrium, what mass of water will be found in the air if there is no ventilation? Repeat the calculation for open vessels containing benzene (vp = 13.1 kPa) and mercury (vp = 0.23 Pa)arrow_forwardEvery chemist knows to ‘add acid to water with constant stirring’ when diluting a concentrated acid in order to keep the solution from spewing boiling acid all over the place. Explain how this one fact is enough to prove that strong acids and water do not form ideal solutions.arrow_forward
- The predominant components of our atmosphere are N₂, O₂, and Ar in the following mole fractions: χN2 = 0.780, χO2 = 0.21, χAr = 0.01. Assuming that these molecules act as ideal gases, calculate ΔGmix, ΔSmix, and ΔHmix when the total pressure is 1 bar and the temperature is 300 K.arrow_forwarddG = Vdp - SdT + μA dnA + μB dnB + ... so that under constant pressure and temperature conditions, the chemical potential of a component is the rate of change of the Gibbs energy of the system with respect to changing composition, μJ = (∂G / ∂nJ)p,T,n' Using first principles prove that under conditions of constant volume and temperature, the chemical potential is a measure of the partial molar Helmholtz energy (μJ = (∂A / ∂nJ)V,T,n')arrow_forwardThe vapor pressure of dichloromethane at 20.0 °C is 58.0 kPa and its enthalpy of vaporization is 32.7 kJ/mol. Estimate the temperature at which its vapor pressure is 66.0 kPa.arrow_forward
- Draw the structure of A, the minor E1 product of the reaction. Cl Skip Part Check F1 esc CH_CH OH, D 3 2 Click and drag to start drawing a structure. 80 R3 F4 F2 F3 @ 2 # $ 4 3 Q W 95 % KO 5 F6 A F7 × G ☐ Save For Later Sub 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C ►II A A F8 F9 F10 FL 6 7 88 & * 8 9 LLI E R T Y U A S D lock LL F G H 0 P J K L Z X C V B N M 9 Harrow_forwardFrom the choices given, which two substances have the same crystal structure? (Select both) Group of answer choices ZnS (zincblende) Diamond TiO2 (rutile) ZnS (wurtzite)arrow_forwardPotassium (K) blends with germanium (Ge) to form a Zintl phase with a chemical formula of K4Ge4. Which of the following elements would you expect potassium to blend with to form an alloy? Electronegativities: As (2.0), Cl (3.0), Ge (1.8), K (0.8), S (2.5), Ti (1.5) Group of answer choices Arsenic (As) Sulfur (S) Chlorine (Cl) Titanium (Ti)arrow_forward
- Consider two elements, X and Z. Both have cubic-based unit cells with the same edge lengths. X has a bcc unit cell while Z has a fcc unit cell. Which of the following statements is TRUE? Group of answer choices Z has a larger density than X X has more particles in its unit cell than Z does X has a larger density than Z Z has a larger unit cell volume than Xarrow_forwardHow many particles does a face-centered cubic (fcc) unit cell contain? Group of answer choices 2 14 8 4arrow_forwardV Highlight all of the carbon atoms that have at least one beta (B) hydrogen, using red for one ẞ hydrogen, blue for two ẞ hydrogens, and green for three ẞ hydrogens. If none of the carbon atoms have ẞ hydrogens, check the box underneath the molecule. ED X None of the carbon atoms have ẞ hydrogens. Explanation esc 2 Check * F1 F2 1 2 80 # 3 Q W tab A caps lock shift fn control F3 N S option O 694 $ F4 F5 F6 005 % E R D F LL 6 olo 18 Ar B © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility A DII F7 F8 87 & * 8 T Y U G H 4 F9 F10 ( 9 0 E F11 F12 உ J K L + || X C V B N M H H command option commandarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





