
Chemistry: Atoms First
2nd Edition
ISBN: 9780073511184
Author: Julia Burdge, Jason Overby Professor
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.12, Problem 21PPC
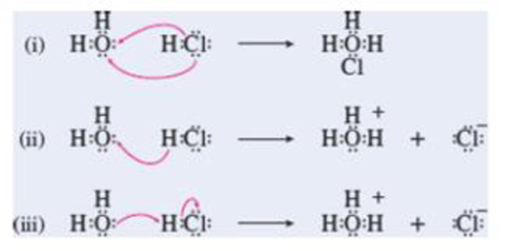
Which of the diagrams best depicts the combination of HCl and water as a Lewis acid-base reaction? Which best depicts the combination as a Brønsted acid-base reaction?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
hybridization of nitrogen of complex molecules
Using reaction free energy to predict equilibrium composition
Consider the following equilibrium:
2NO2 (g) = N2O4(g)
AGº = -5.4 kJ
Now suppose a reaction vessel is filled with 4.53 atm of dinitrogen tetroxide (N2O4) at 279. °C. Answer the following questions about this system:
Under these conditions, will the pressure of N2O4 tend to rise or fall?
Is it possible to reverse this tendency by adding NO2?
In other words, if you said the pressure of N2O4 will tend to rise, can that
be changed to a tendency to fall by adding NO2? Similarly, if you said the
pressure of N2O4 will tend to fall, can that be changed to a tendency to
'2'
rise by adding NO2?
If you said the tendency can be reversed in the second question, calculate
the minimum pressure of NO 2 needed to reverse it.
Round your answer to 2 significant digits.
00
rise
☐ x10
fall
yes
no
☐ atm
G
Ar
1
Why do we analyse salt?
Chapter 16 Solutions
Chemistry: Atoms First
Ch. 16.1 - What is (a) the conjugate base of HNO3, (b) the...Ch. 16.1 - What is (a) the conjugate acid of ClO4, (b) the...Ch. 16.1 - HSO3 is the conjugate acid of what species? HSO3...Ch. 16.1 - Which of the models represents a species that has...Ch. 16.1 - Prob. 16.2WECh. 16.1 - Identify and label the species in each reaction....Ch. 16.1 - Prob. 2PPBCh. 16.1 - Write the formula and charge for each species in...Ch. 16.1 - Prob. 16.1.1SRCh. 16.1 - Prob. 16.1.2SR
Ch. 16.2 - Predict the relative strengths of the oxoacids in...Ch. 16.2 - Prob. 3PPACh. 16.2 - Based on the information in this section, which is...Ch. 16.2 - Prob. 3PPCCh. 16.2 - Prob. 16.2.1SRCh. 16.2 - Prob. 16.2.2SRCh. 16.2 - Prob. 16.2.3SRCh. 16.3 - Prob. 16.4WECh. 16.3 - The concentration of hydroxide ions in the antacid...Ch. 16.3 - The value of Kw at normal body temperature (37C)...Ch. 16.3 - Prob. 4PPCCh. 16.3 - Prob. 16.3.1SRCh. 16.3 - Prob. 16.3.2SRCh. 16.4 - Determine the pOH of a solution at 25C in which...Ch. 16.4 - Determine the pOH of a solution at 25C in which...Ch. 16.4 - Determine the pOH of a solution at 25C in which...Ch. 16.4 - Prob. 5PPCCh. 16.4 - Calculate the hydroxide ion concentration in a...Ch. 16.4 - Prob. 6PPACh. 16.4 - Prob. 6PPBCh. 16.4 - Prob. 6PPCCh. 16.4 - Prob. 16.4.1SRCh. 16.4 - Prob. 16.4.2SRCh. 16.4 - Prob. 16.4.3SRCh. 16.4 - Prob. 16.4.4SRCh. 16.5 - Calculate the pH of an aqueous solution at 25C...Ch. 16.5 - Prob. 7PPACh. 16.5 - Prob. 7PPBCh. 16.5 - Prob. 7PPCCh. 16.5 - Prob. 16.8WECh. 16.5 - Calculate the concentration of HNO3 in a solution...Ch. 16.5 - Prob. 8PPBCh. 16.5 - Which of the plots [(i)(iv)] best approximates the...Ch. 16.5 - Prob. 16.9WECh. 16.5 - Prob. 9PPACh. 16.5 - Prob. 9PPBCh. 16.5 - Prob. 9PPCCh. 16.5 - Prob. 16.10WECh. 16.5 - Prob. 10PPACh. 16.5 - Prob. 10PPBCh. 16.5 - Prob. 10PPCCh. 16.5 - Prob. 16.5.1SRCh. 16.5 - Prob. 16.5.2SRCh. 16.5 - Prob. 16.5.3SRCh. 16.5 - Prob. 16.5.4SRCh. 16.5 - Prob. 16.5.5SRCh. 16.5 - Prob. 16.5.6SRCh. 16.5 - Prob. 16.5.7SRCh. 16.6 - The Ka of hypochlorous acid (HClO) is 3.5 108....Ch. 16.6 - Calculate the pH at 25C of a 0.18-M solution of a...Ch. 16.6 - Prob. 11PPBCh. 16.6 - The diagrams show solutions of four different weak...Ch. 16.6 - Determine the pH and percent ionization for acetic...Ch. 16.6 - Determine the pH and percent ionization for...Ch. 16.6 - At what concentration does hydrocyanic acid...Ch. 16.6 - Prob. 12PPCCh. 16.6 - Aspirin (acetylsalicylie acid, HC9H7O4) is a weak...Ch. 16.6 - Prob. 13PPACh. 16.6 - Prob. 13PPBCh. 16.6 - Calculate Ka values (to two significant figures)...Ch. 16.6 - Prob. 16.6.1SRCh. 16.6 - Prob. 16.6.2SRCh. 16.6 - Prob. 16.6.3SRCh. 16.7 - Prob. 16.14WECh. 16.7 - Calculate the pH at 25C of a 0.0028-M solution of...Ch. 16.7 - Prob. 14PPBCh. 16.7 - The diagrams represent solutions of three...Ch. 16.7 - Caffeine, the stimulant in coffee and tea, is a...Ch. 16.7 - Prob. 15PPACh. 16.7 - Prob. 15PPBCh. 16.7 - Prob. 15PPCCh. 16.7 - Prob. 16.7.1SRCh. 16.7 - Prob. 16.7.2SRCh. 16.7 - Prob. 16.7.3SRCh. 16.8 - Prob. 16.16WECh. 16.8 - Prob. 16PPACh. 16.8 - Prob. 16PPBCh. 16.8 - Prob. 16PPCCh. 16.8 - Prob. 16.8.1SRCh. 16.8 - Prob. 16.8.2SRCh. 16.8 - Prob. 16.8.3SRCh. 16.9 - Oxalic acid (H2C2O4) is a poisonous substance used...Ch. 16.9 - Calculate the concentrations of H2C2O4, HC2O4,...Ch. 16.9 - Calculate the concentrations of H2SO4, HSO4, SO42,...Ch. 16.9 - Prob. 17PPCCh. 16.9 - Prob. 16.9.1SRCh. 16.9 - Prob. 16.9.2SRCh. 16.9 - Prob. 16.9.3SRCh. 16.10 - Prob. 16.18WECh. 16.10 - Determine the pH of a 0.15-M solution of sodium...Ch. 16.10 - Prob. 18PPBCh. 16.10 - Winch of the graphs [(i)(iv)] best represents the...Ch. 16.10 - Calculate the pH of a 0.10-M solution of ammonium...Ch. 16.10 - Determine the pH of a 0.25-M solution of...Ch. 16.10 - Prob. 19PPBCh. 16.10 - Prob. 19PPCCh. 16.10 - Predict whether a 0.10-M solution of each of the...Ch. 16.10 - Predict whether a 0.10-M solution of each of the...Ch. 16.10 - Prob. 20PPBCh. 16.10 - Prob. 20PPCCh. 16.10 - Prob. 16.10.1SRCh. 16.10 - Prob. 16.10.2SRCh. 16.10 - Prob. 16.10.3SRCh. 16.10 - Prob. 16.10.4SRCh. 16.10 - Prob. 16.10.5SRCh. 16.12 - Identify the Lewis acid and Lewis base in each of...Ch. 16.12 - Prob. 21PPACh. 16.12 - Prob. 21PPBCh. 16.12 - Which of the diagrams best depicts the combination...Ch. 16.12 - Prob. 16.12.1SRCh. 16.12 - Prob. 16.12.2SRCh. 16 - F or a species to act as a Brnsted base, an atom...Ch. 16 - Identify the acid-base conjugate pairs in each of...Ch. 16 - Prob. 16.3QPCh. 16 - Prob. 16.4QPCh. 16 - Write the formulas of the conjugate bases of the...Ch. 16 - Prob. 16.6QPCh. 16 - Prob. 16.7QPCh. 16 - List four factors that affect the strength of an...Ch. 16 - Prob. 16.9QPCh. 16 - Prob. 16.10QPCh. 16 - Prob. 16.11QPCh. 16 - Prob. 16.12QPCh. 16 - Prob. 16.13QPCh. 16 - Write the equilibrium expression for the...Ch. 16 - Write an equation relating [H+] and [OH] in...Ch. 16 - Write an equation relating [H+] and [OH] in...Ch. 16 - Prob. 16.17QPCh. 16 - Prob. 16.18QPCh. 16 - Prob. 16.19QPCh. 16 - Prob. 16.20QPCh. 16 - Prob. 16.21QPCh. 16 - Prob. 16.22QPCh. 16 - Prob. 16.23QPCh. 16 - Calculate the concentration of H+ ions in a 0.62 M...Ch. 16 - Calculate the concentration of OH ions in a 1.4 ...Ch. 16 - Calculate the pH of each of the following...Ch. 16 - Calculate the pH of each of the following...Ch. 16 - Prob. 16.28QPCh. 16 - Prob. 16.29QPCh. 16 - Prob. 16.30QPCh. 16 - How much NaOH (in grams) is needed to prepare 546...Ch. 16 - Prob. 16.32QPCh. 16 - Why are ionizations of strong acids and strong...Ch. 16 - Prob. 16.34QPCh. 16 - Prob. 16.35QPCh. 16 - Prob. 16.36QPCh. 16 - Prob. 16.37QPCh. 16 - Prob. 16.38QPCh. 16 - Prob. 16.39QPCh. 16 - Prob. 16.40QPCh. 16 - Prob. 16.41QPCh. 16 - Prob. 16.42QPCh. 16 - Prob. 16.43QPCh. 16 - Prob. 16.1VCCh. 16 - Prob. 16.2VCCh. 16 - Prob. 16.3VCCh. 16 - Prob. 16.4VCCh. 16 - Prob. 16.44QPCh. 16 - Prob. 16.45QPCh. 16 - Prob. 16.46QPCh. 16 - Why do we normally not quote Ka values for strong...Ch. 16 - Why is it necessary to specify temperature when...Ch. 16 - Which of the following solutions has the highest...Ch. 16 - Prob. 16.50QPCh. 16 - The Ka for benzoic acid is 6.5 105. Calculate the...Ch. 16 - Calculate the pH of an aqueous solution at 25C...Ch. 16 - Calculate the pH of an aqueous solution at 25C...Ch. 16 - Determine the percent ionization of the following...Ch. 16 - Determine the percent ionization of the following...Ch. 16 - Prob. 16.56QPCh. 16 - A 0.015-M solution of a monoprotic acid is 0.92%...Ch. 16 - Prob. 16.58QPCh. 16 - Prob. 16.59QPCh. 16 - Prob. 16.60QPCh. 16 - Prob. 16.61QPCh. 16 - Prob. 16.62QPCh. 16 - In biological and medical applications, it is...Ch. 16 - Classify each of the following species as a weak...Ch. 16 - Prob. 16.65QPCh. 16 - Prob. 16.66QPCh. 16 - Prob. 16.67QPCh. 16 - Which of the following has a higher pH: (a) 1.0 M...Ch. 16 - Prob. 16.69QPCh. 16 - Prob. 16.70QPCh. 16 - Prob. 16.71QPCh. 16 - What is the original molarity of an aqueous...Ch. 16 - Prob. 16.73QPCh. 16 - Prob. 16.74QPCh. 16 - Prob. 16.75QPCh. 16 - Prob. 16.76QPCh. 16 - Prob. 16.77QPCh. 16 - Calculate Ka for each of the following ions: NH4+,...Ch. 16 - The following diagrams represent aqueous solutions...Ch. 16 - Prob. 16.80QPCh. 16 - Write all the species (except water) that are...Ch. 16 - Write the Ka1 and Ka2 expressions for sulfurous...Ch. 16 - Prob. 16.83QPCh. 16 - Prob. 16.84QPCh. 16 - Prob. 16.85QPCh. 16 - Prob. 16.86QPCh. 16 - Calculate the pH at 25C of a 0.25-M aqueous...Ch. 16 - The first and second ionization constants of a...Ch. 16 - Prob. 16.89QPCh. 16 - Prob. 16.90QPCh. 16 - Explain why small, highly charged metal ions are...Ch. 16 - Prob. 16.92QPCh. 16 - Specify which of the following salts will undergo...Ch. 16 - Prob. 16.94QPCh. 16 - Calculate the pH of a 0.42 M NH4Cl solution. (Kb...Ch. 16 - Calculate the pH of a 0.082 M NaF solution. (Ka...Ch. 16 - Calculate the pH of a 0.91 M C2H5NH3I solution....Ch. 16 - Prob. 16.98QPCh. 16 - Predict whether the following solutions are...Ch. 16 - Prob. 16.100QPCh. 16 - In a certain experiment, a student finds that the...Ch. 16 - Prob. 16.102QPCh. 16 - Prob. 16.103QPCh. 16 - Classify the following oxides as acidic, basic,...Ch. 16 - Prob. 16.105QPCh. 16 - Explain why metal oxides tend to be basic if the...Ch. 16 - Arrange the oxides in each of the following groups...Ch. 16 - Prob. 16.108QPCh. 16 - Prob. 16.109QPCh. 16 - Prob. 16.110QPCh. 16 - Prob. 16.111QPCh. 16 - Prob. 16.112QPCh. 16 - In terms of orbitals and electron arrangements,...Ch. 16 - Prob. 16.114QPCh. 16 - Prob. 16.115QPCh. 16 - Which would be considered a stronger Lewis acid:...Ch. 16 - Prob. 16.117QPCh. 16 - Identify the Lewis acid and the Lewis base in the...Ch. 16 - Identify the Lewis acid and the Lewis base in the...Ch. 16 - Prob. 16.120QPCh. 16 - Prob. 16.121QPCh. 16 - Prob. 16.122QPCh. 16 - Prob. 16.123QPCh. 16 - Prob. 16.124QPCh. 16 - Calculate the pH and percent ionization of a 0.88...Ch. 16 - Prob. 16.126QPCh. 16 - Prob. 16.127QPCh. 16 - The pH of a 0.0642-M solution of a monoprotic acid...Ch. 16 - Prob. 16.129QPCh. 16 - HA and HB are both weak acids although HB is the...Ch. 16 - Prob. 16.131QPCh. 16 - Prob. 16.132QPCh. 16 - Use the data in Table 16.5 to calculate the...Ch. 16 - Prob. 16.134QPCh. 16 - Most of the hydrides of Group 1A and Group 2 A...Ch. 16 - Prob. 16.136QPCh. 16 - Novocaine, used as a local anesthetic by dentists,...Ch. 16 - Which of the following is the stronger base: NF3...Ch. 16 - Prob. 16.139QPCh. 16 - The ion product of D20 is 1.35 1015 at 25C. (a)...Ch. 16 - Prob. 16.141QPCh. 16 - Prob. 16.142QPCh. 16 - Prob. 16.143QPCh. 16 - Prob. 16.144QPCh. 16 - Prob. 16.145QPCh. 16 - When the concentration of a strong acid is not...Ch. 16 - Calculate the pH of a 2.00 M NH4CN solution.Ch. 16 - Prob. 16.148QPCh. 16 - Prob. 16.149QPCh. 16 - Prob. 16.150QPCh. 16 - Prob. 16.151QPCh. 16 - Hydrocyanic acid (HCN) is a weak acid and a deadly...Ch. 16 - How many grams of NaCN would you need to dissolve...Ch. 16 - Prob. 16.154QPCh. 16 - Calculate the pH of a 1-L solution containing...Ch. 16 - Prob. 16.156QPCh. 16 - You are given two beakers, one containing an...Ch. 16 - Use Le Chteliers principle to predict the effect...Ch. 16 - A 0.400 M formic acid (HCOOH) solution freezes at...Ch. 16 - The disagreeable odor of fish is mainly due to...Ch. 16 - Prob. 16.161QPCh. 16 - Prob. 16.162QPCh. 16 - Both the amide ion (NH2) and the nitride ion (N3)...Ch. 16 - When carbon dioxide is bubbled through a clear...Ch. 16 - Explain the action of smelling salt, which is...Ch. 16 - About half of the hydrochloric acid produced...Ch. 16 - Which of the following does not represent a Lewis...Ch. 16 - Determine whether each of the following statements...Ch. 16 - How many milliliters of a strong monoprotic acid...Ch. 16 - Hemoglobin (Hb) is a blood protein that is...Ch. 16 - Prob. 16.171QPCh. 16 - Calculate the pH of a solution that is 1.00 M HCN...Ch. 16 - Tooth enamel is largely hydroxyapatite...Ch. 16 - Prob. 16.174QPCh. 16 - Prob. 16.175QPCh. 16 - Prob. 16.176QPCh. 16 - Sulfuric acid (H2SO4) accounts for as much as 80...Ch. 16 - A 1-87-g sample of Mg reacts with 80.0 mL of a HCl...Ch. 16 - Calculate the pH of a solution that is 0.22 M in...Ch. 16 - Determine pH at the equivalence point in the...Ch. 16 - Calculate the pH of a solution that is 0.22 M in...Ch. 16 - Determine pH at the equivalence point in the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. H H CH3OH, H+ H Select to Add Arrows H° 0:0 'H + Q HH ■ Select to Add Arrows CH3OH, H* H. H CH3OH, H+ HH ■ Select to Add Arrows i Please select a drawing or reagent from the question areaarrow_forwardWhat are examples of analytical methods that can be used to analyse salt in tomato sauce?arrow_forwardA common alkene starting material is shown below. Predict the major product for each reaction. Use a dash or wedge bond to indicate the relative stereochemistry of substituents on asymmetric centers, where applicable. Ignore any inorganic byproducts H Šali OH H OH Select to Edit Select to Draw 1. BH3-THF 1. Hg(OAc)2, H2O =U= 2. H2O2, NaOH 2. NaBH4, NaOH + Please select a drawing or reagent from the question areaarrow_forward
- What is the MOHR titration & AOAC method? What is it and how does it work? How can it be used to quantify salt in a sample?arrow_forwardPredict the major products of this reaction. Cl₂ hv ? Draw only the major product or products in the drawing area below. If there's more than one major product, you can draw them in any arrangement you like. Be sure you use wedge and dash bonds if necessary, for example to distinguish between major products with different stereochemistry. If there will be no products because there will be no significant reaction, just check the box under the drawing area and leave it blank. Note for advanced students: you can ignore any products of repeated addition. Explanation Check Click and drag to start drawing a structure. 80 10 m 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility DII A F1 F2 F3 F4 F5 F6 F7 F8 EO F11arrow_forwardGiven a system with an anodic overpotential, the variation of η as a function of current density- at low fields is linear.- at higher fields, it follows Tafel's law.Calculate the range of current densities for which the overpotential has the same value when calculated for both cases (the maximum relative difference will be 5%, compared to the behavior for higher fields).arrow_forward
- Using reaction free energy to predict equilibrium composition Consider the following equilibrium: N2 (g) + 3H2 (g) = 2NH3 (g) AGº = -34. KJ Now suppose a reaction vessel is filled with 8.06 atm of nitrogen (N2) and 2.58 atm of ammonia (NH3) at 106. °C. Answer the following questions about this system: rise Under these conditions, will the pressure of N2 tend to rise or fall? ☐ x10 fall Is it possible to reverse this tendency by adding H₂? In other words, if you said the pressure of N2 will tend to rise, can that be changed to a tendency to fall by adding H2? Similarly, if you said the pressure of N will tend to fall, can that be changed to a tendency to rise by adding H₂? If you said the tendency can be reversed in the second question, calculate the minimum pressure of H₂ needed to reverse it. Round your answer to 2 significant digits. yes no ☐ atm Х ด ? olo 18 Ararrow_forwardFour liters of an aqueous solution containing 6.98 mg of acetic acid were prepared. At 25°C, the measured conductivity was 5.89x10-3 mS cm-1. Calculate the degree of dissociation of the acid and its ionization constant.Molecular weights: O (15.999), C (12.011), H (1.008).Limiting molar ionic conductivities (λ+0 and λ-0) of Ac-(aq) and H+(aq): 40.9 and 349.8 S cm-2 mol-1.arrow_forwardDetermine the change in Gibbs energy, entropy, and enthalpy at 25°C for the battery from which the data in the table were obtained.T (°C) 15 20 25 30 35Eo (mV) 227.13 224.38 221.87 219.37 216.59Data: n = 1, F = 96485 C mol–1arrow_forward
- Indicate the correct options.1. The units of the transport number are Siemens per mole.2. The Siemens and the ohm are not equivalent.3. The Van't Hoff factor is dimensionless.4. Molar conductivity does not depend on the electrolyte concentration.arrow_forwardIdeally nonpolarizable electrodes can1. participate as reducers in reactions.2. be formed only with hydrogen.3. participate as oxidizers in reactions.4. form open and closed electrochemical systems.arrow_forwardIndicate the options for an electrified interface:1. Temperature has no influence on it.2. Not all theories that describe it include a well-defined electrical double layer.3. Under favorable conditions, its differential capacitance can be determined with the help of experimental measurements.4. A component with high electronic conductivity is involved in its formation.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY