
Introduction to General, Organic and Biochemistry
12th Edition
ISBN: 9780357119303
Author: Bettelheim, Frederick A., Brown, William H., Campbell, Mary K., FARRELL, Shawn O., Torres, Omar
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16, Problem 45P
Interpretation Introduction
Interpretation:
For the formation of an acetal from any carbonyl compound; is the oxygen-18 atom in carbonyl stays in the acetal or it eliminates as water, we need to find that by the mechanism.
Concept Introduction:
An acetal is the type of compound where two −OR group attached to a single carbon where −R is any alkyl group. Acetal forms when an alcohol reacts with a hemi acetal in acidic condition. A hemi-acetal formed when an alcohol reacts with an
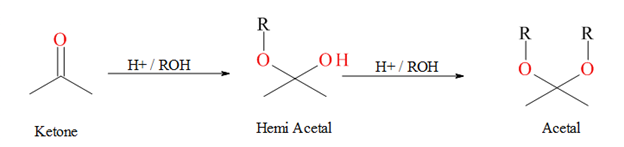
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Complete the mechanism
V
Biological Macromolecules
Drawing the Haworth projection of an aldose from its Fischer projection
Draw a Haworth projection of a common cyclic form of this monosaccharide:
H
C=O
HO
H
HO
H
H
OH
CH₂OH
Explanation
Check
Click and drag to start drawing a
structure.
X
Complete the mechanism
Chapter 16 Solutions
Introduction to General, Organic and Biochemistry
Ch. 16.2 - Problem 17-1 Wrtie the IUPAC name for each...Ch. 16.2 - Prob. 16.2QCCh. 16.2 - Prob. 16.3QCCh. 16.4 - Prob. 16.4QCCh. 16.4 - Prob. 16.5QCCh. 16.4 - Problem 17-6 Show the reaction of benzaldehyde...Ch. 16.4 - Problem 17-7 Identify all hemiacetals and acetals...Ch. 16.5 - Prob. 16.8QCCh. 16 - 17-9 Answer true or false. (a) The one aldehyde...Ch. 16 - Prob. 2P
Ch. 16 - 17-11 What is the difference in structure between...Ch. 16 - 17-12 Is it possible for the carbon atom of a...Ch. 16 - 17-13 Which compounds contain carbonyl groups?Ch. 16 - 17-14 Following are structural formulas for two...Ch. 16 - 17-15 Draw structural formulas for the four...Ch. 16 - Prob. 8PCh. 16 - Prob. 9PCh. 16 - 17-18 Draw structural formulas for these ketones....Ch. 16 - 17-19 Write the JUPAC names for these compounds.Ch. 16 - Prob. 12PCh. 16 - 17-2 1 Explain why each name is incorrect. Write...Ch. 16 - Prob. 14PCh. 16 - Prob. 15PCh. 16 - 17-24 In each pair of compounds, select the one...Ch. 16 - Prob. 17PCh. 16 - 17-26 Account for the fact that acetone has a...Ch. 16 - 17-27 Pentane, 1-butanol, and butanal all have...Ch. 16 - 17-28 Show how acetaldehyde can form hydrogen...Ch. 16 - 17-29 Why can’t two molecules of acetone form a...Ch. 16 - 17-30 Answer true or false. (a) The reduction of...Ch. 16 - 17-3 1 Draw a structural formula for the principal...Ch. 16 - Prob. 24PCh. 16 - 17-33 What simple chemical test could you use to...Ch. 16 - 17-34 Explain why liquid aldehydes are often...Ch. 16 - 17-35 Suppose that you take a bottle of...Ch. 16 - 17-36 Explain why the reduction of an aldehyde...Ch. 16 - Prob. 29PCh. 16 - Prob. 30PCh. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Prob. 33PCh. 16 - Prob. 34PCh. 16 - Prob. 35PCh. 16 - Prob. 36PCh. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - 17-47 What is the characteristic structural...Ch. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - Prob. 43PCh. 16 - Prob. 44PCh. 16 - Prob. 45PCh. 16 - 17-54 Following is the structure of...Ch. 16 - Prob. 47PCh. 16 - Prob. 48PCh. 16 - Prob. 49PCh. 16 - Prob. 50PCh. 16 - Prob. 51PCh. 16 - 17-60 1-Propanol can be prepared by the reduction...Ch. 16 - Prob. 53PCh. 16 - 17-62 Show how to bring about these conversions....Ch. 16 - Prob. 55PCh. 16 - Prob. 56PCh. 16 - Prob. 57PCh. 16 - Prob. 58PCh. 16 - 17-67 Draw structural formulas for these...Ch. 16 - Prob. 60PCh. 16 - 17-69 Propanal (bp 49°C) and 1-propanol (bp 97°C)...Ch. 16 - 17-70 What simple chemical test could you use to...Ch. 16 - Prob. 63PCh. 16 - Prob. 64PCh. 16 - Prob. 65PCh. 16 - 17-72 The following molecule is an enediol; each...Ch. 16 - 17-73 Alcohols can be prepared by the...Ch. 16 - 17-74 Glucose, C6H12O6, contains an aldehyde group...Ch. 16 - Prob. 69PCh. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - 17-78 Complete the following equation for these...Ch. 16 - 17-79 Write an equation for each conversion. (a)...Ch. 16 - Prob. 74P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete the mechanismarrow_forward8 00 6 = 10 10 Decide whether each of the molecules in the table below is stable, in the exact form in which it is drawn, at pH = 11. If you decide at least one molecule is not stable, then redraw one of the unstable molecules in its stable form below the table. (If more than unstable, you can pick any of them to redraw.) Check OH stable HO stable Ounstable unstable O OH stable unstable OH 80 F6 F5 stable Ounstable X Save For Later Sub 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C ཀྭ་ A F7 매 F8 F9 4 F10arrow_forwardJust try completing it and it should be straightforward according to the professor and TAs.arrow_forward
- The grading is not on correctness, so if you can just get to the correct answers without perfectionism that would be great. They care about the steps and reasoning and that you did something. I asked for an extension, but was denied the extension.arrow_forwardShow your work and do something that is reasonable. It does not have to be 100% correct. Just show something that looks good or pretty good as acceptable answers. Something that looks reasonable or correct would be sufficient. If you can get many of them correct that would be great!arrow_forwardShow your work and do something that is reasonable. It does not have to be 100% correct. Just show something that looks good or pretty good as acceptable answers. Something that looks reasonable or correct would be sufficient. If you can get many of them correct that would be great!arrow_forward
- Take a look at the following molecule, and then answer the questions in the table below it. (You can click the other tab to see the molecule without the colored regions.) with colored region plain 0= CH2-0-C-(CH2)16-CH3 =0 CH-O-C (CH2)7-CH=CH-(CH2)5-CH3 D CH3 | + OMPLO CH3-N-CH2-CH2-0-P-O-CH2 B CH3 A Try again * 000 Ar 8 0 ?arrow_forwardShow your work and do something that is reasonable. It does not have to be 100% correct. Just show something that looks good or pretty good as acceptable answers.arrow_forwardShow your work and do something that is reasonable. It does not have to be 100% correct. Just show something that looks good or pretty good as acceptable answers.arrow_forward
- = 1 = 2 3 4 5 6 ✓ 7 8 ✓ 9 =10 Devise a synthesis to prepare the product from the given starting material. Complete the following reaction scheme. Part 1 of 3 -Br Draw the structure for compound A. Check Step 1 Step 2 A Click and drag to start drawing a structure. × ↓m + OH Save For Later S 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privaarrow_forwardPredict the products of this organic reduction: 田 Check AP + + H2 Lindlar catalyst Click an drawing 2025 McGraw Hill LLC. All Rigarrow_forward70 Suppose the molecule below is in acidic aqueous solution. Is keto-enol tautomerization possible? • If a keto-enol tautomerization is possible, draw the mechanism for it. Be sure any extra reagents you add to the left-hand sid available in this solution. • If a keto-enol tautomerization is not possible, check the box under the drawing area. : ☐ Add/Remove step Click and drag to st drawing a structure Check Save For Late. 2025 McGraw Hill LLC. All Rights Reserved. Terms of Usearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY