
Concept explainers
(a)
Interpretation:
The name of the given ketone is to be determined.
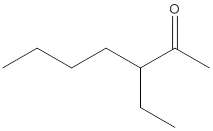
Concept Introduction:
While naming the
(b)
Interpretation:
The name of the given ketone is to be determined.
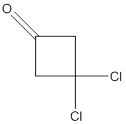
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
(c)
Interpretation:
The name of the given ketone is to be determined.
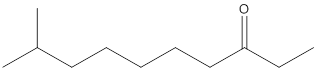
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
(d)
Interpretation:
The name of the given ketone is to be determined.
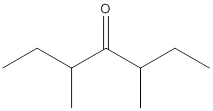
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
(e)
Interpretation:
The name of the given ketone is to be determined.
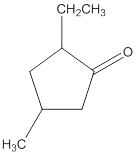
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
- Draw the complete mechanism for the acid-catalyzed hydration of this alkene. esc 田 Explanation Check 1 888 Q A slock Add/Remove step Q F4 F5 F6 A བྲA F7 $ % 5 @ 4 2 3 & 6 87 Click and drag to start drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Ce W E R T Y U S D LL G H IK DD 요 F8 F9 F10 F1 * ( 8 9 0 O P J K L Z X C V B N M H He commandarrow_forwardExplanation Check F1 H₂O H₂ Pd 1) MCPBA 2) H3O+ 1) Hg(OAc)2, H₂O 2) NaBH4 OH CI OH OH OH hydration halohydrin formation addition halogenation hydrogenation inhalation hydrogenation hydration ☐ halohydrin formation addition halogenation formation chelation hydrogenation halohydrin formation substitution hydration halogenation addition Ohalohydrin formation subtraction halogenation addition hydrogenation hydration F2 80 F3 σ F4 F5 F6 1 ! 2 # 3 $ 4 % 05 Q W & Å © 2025 McGraw Hill LLC. All Rights Reserved. F7 F8 ( 6 7 8 9 LU E R T Y U A F9arrow_forwardShow the mechanism steps to obtain the lowerenergy intermediate: *see imagearrow_forward
- Soap is made by the previous reaction *see image. The main difference between one soap and another soap isthe length (number of carbons) of the carboxylic acid. However, if a soap irritates your skin, they mostlikely used too much lye.Detergents have the same chemical structure as soaps except for the functional group. Detergentshave sulfate (R-SO4H) and phosphate (R-PO4H2) functional groups. Draw the above carboxylic acidcarbon chain but as the two variants of detergents. *see imagearrow_forwardWhat are the reactions or reagents used? *see imagearrow_forwardWhat are the reactions or reagents used? *see imagearrow_forward
- Provide the mechanism for this transformation: *see imagearrow_forwardAssign all the signals individually (please assign the red, green and blue)arrow_forwardThe two pKa values of oxalic acid are 1.25 and 3.81. Why are they not the same value? Show the protontransfer as part of your explanation. *see imagearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




